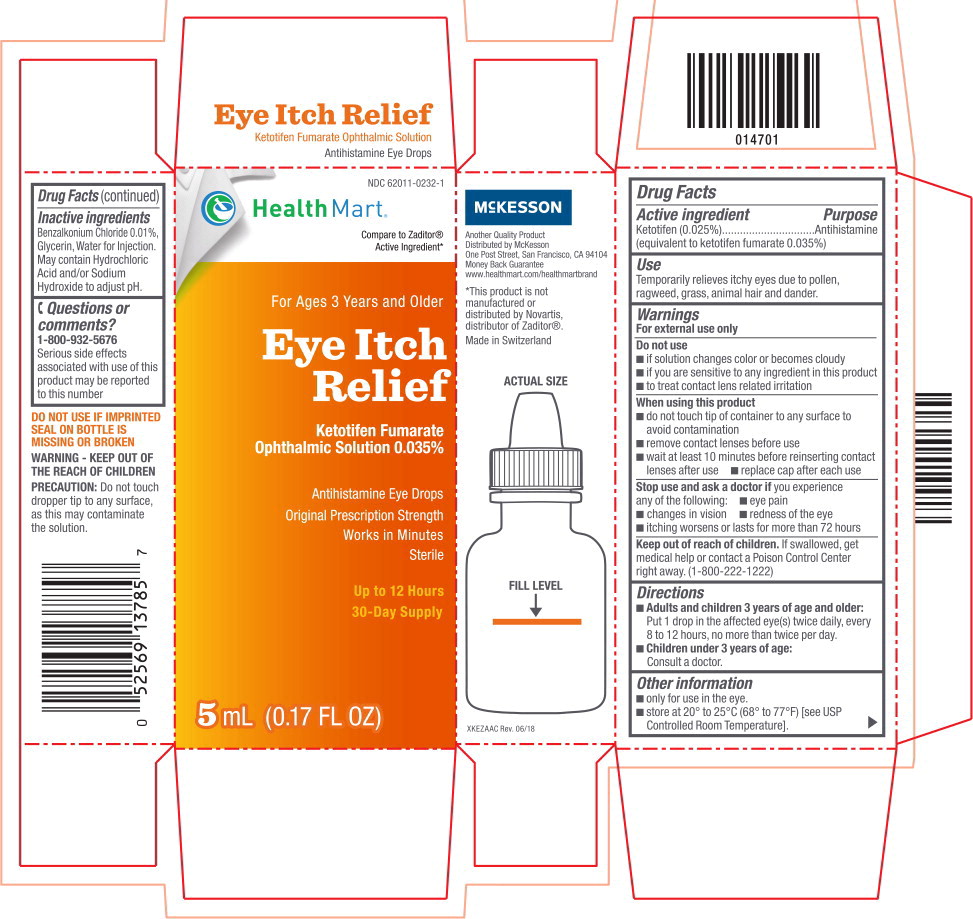
Warnings
For external use only
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
- to treat contact lens related irritation
When using this product
- do not touch tip of container to any surface to avoid contamination
- remove contact lenses before use
- wait at least 10 minutes before reinserting contact lenses after use
- replace cap after each use
Directions
-
Adults and children 3 years of age and older:
Put 1 drop in the affected eye(s) twice daily, every 8 to 12 hours, no more than twice per day. -
Children under 3 years of age:
Consult a doctor.
Other information
- only for use in the eye.
- store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Inactive ingredients
Benzalkonium Chloride 0.01%, Glycerin, Water for Injection. May contain Hydrochloric Acid and/or Sodium Hydroxide to adjust pH.
1-800-932-5676
Serious side effects associated with use of this product may be reported to this number
Principal Display Panel Text for Container Label:
Health Mart®
NDC 62011-0232-1
Eye Itch Relief
Ketotifen Fumarate
Ophthalmic Solution 0.035%
Antihistamine Eye Drops
Sterile
5 mL (0.17 FL OZ)
Principal Display Panel Text for Carton Label:
NDC 62011-0232-1
Health Mart®
Compare to Zaditor® Active Ingredient*
For Ages 3 Years and Older
Eye Itch Relief
Ketotifen Fumarate
Ophthalmic Solution 0.035%
Antihistamine Eye Drops
Original Prescription Strength
Works in Minutes
Sterile
Up to 12 Hours
30-Day Supply
5 mL (0.17 FL OZ)