NAFCILLIN- nafcillin injection, powder, for solution
Sandoz Inc
----------
Nafcillin for Injection, USP
Rx Only
For Intramuscular or Intravenous Injection
To reduce the development of drug-resistant bacteria and maintain the effectiveness of nafcillin for injection and other antibacterial drugs, nafcillin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
DESCRIPTION
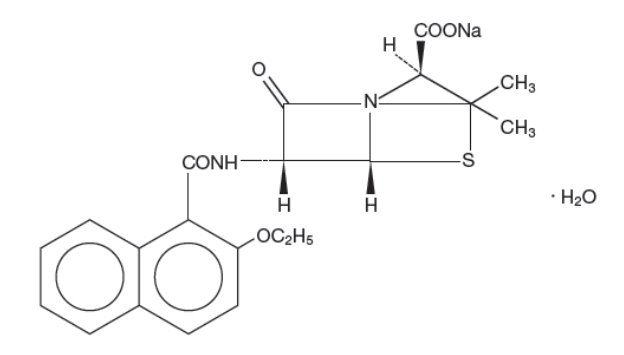
Nafcillin for injection, USP is a sterile semisynthetic penicillin derived from the penicillin nucleus, 6- aminopenicillanic acid. The chemical name of nafcillin sodium is Monosodium (2S,5R,6R)-6-(2-ethoxy- 1-naphthamido)-3,3 dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate monohydrate. It is resistant to inactivation by the enzyme penicillinase (beta-lactamase). The structural formula is as follows:
Nafcillin Sodium
Molecular Formula: C21H21N2NaO5S • H2O
Molecular Weight: 454.47
Nafcillin for injection, USP for the intramuscular or intravenous route of administration, contains nafcillin sodium as a sterile white to slightly yellowish powder for reconstitution. The pH of the reconstituted solution is 6 - 8.5. Nafcillin for injection, USP contains nafcillin sodium as the monohydrate equivalent to 1 gram or 2 grams of nafcillin per vial and is buffered with 40 mg sodium citrate per gram of nafcillin. The sodium content is 66.1 mg [2.9 mEq] per gram of nafcillin.
CLINICAL PHARMACOLOGY
In a study of five healthy adults administered a single 500 mg dose of nafcillin by intravenous injection over seven minutes, the mean plasma concentration of the drug was approximately 30 mcg/mL at 5 minutes after injection. The mean area under the plasma concentration-versus-time curve (AUC) for nafcillin in this study was 18.06 mcg•h/mL.
The serum half-life of nafcillin administered by the intravenous route ranged from 33 to 61 minutes as measured in three separate studies.
In contrast to the other penicillinase-resistant penicillins, only about 30% of nafcillin is excreted as unchanged drug in the urine of normal volunteers, and most within the first six hours. Nafcillin is primarily eliminated by nonrenal routes, namely hepatic inactivation and excretion in the bile.
Nafcillin binds to serum proteins, mainly albumin. The degree of protein binding reported for nafcillin is 89.9 ± 1.5%. Reported values vary with the method of study and the investigator.
The concurrent administration of probenecid with nafcillin increases and prolongs plasma concentrations of nafcillin. Probenecid significantly reduces the total body clearance of nafcillin with renal clearance being decreased to a greater extent than nonrenal clearance.
The penicillinase-resistant penicillins are widely distributed in various body fluids, including bile, pleural, amniotic and synovial fluids. With normal doses insignificant concentrations are found in the aqueous humor of the eye. High nafcillin CSF levels have been obtained in the presence of inflamed meninges.
Renal failure does not appreciably affect the serum half-life of nafcillin; therefore, no modification of the usual nafcillin dosage is necessary in renal failure with or without hemodialysis. Hemodialysis does not accelerate the rate of clearance of nafcillin from the blood.
A study which assessed the effects of cirrhosis and extrahepatic biliary obstruction in man demonstrated that the plasma clearance of nafcillin was significantly decreased in patients with hepatic dysfunction. In these patients with cirrhosis and extrahepatic obstruction, nafcillin excretion in the urine was significantly increased from about 30 to 50% of the administered dose, suggesting that renal disease superimposed on hepatic disease could further decrease nafcillin clearance.
PHARMACOKINETICS
Intramuscular injections of nafcillin sodium 1 gram produced peak serum levels in 0.5 to 1 hour of 7.61 mcg/mL. The degree of protein binding reported has been 89.9 +/-1.5%. With normal doses nafcillin is found in therapeutic concentrations in the pleural, bile, and amniotic fluids. Insignificant concentrations are found in the cerebrospinal fluid and aqueous humor. Blood concentrations may be tripled by the concurrent use of probenecid. Clinical studies with nafcillin sodium in infants under three days of age and prematures have revealed higher blood levels and slower rates of urinary excretion than in older children and adults. A high concentration of nafcillin sodium is excreted via the bile. About 30% of an intramuscular dose is excreted in the urine.
MICROBIOLOGY
Penicillinase-resistant penicillins exert a bactericidal action against penicillin-susceptible microorganisms during the state of active multiplication. All penicillins inhibit the biosynthesis of the bacterial cell wall. Nafcillin sodium has been shown to be active against most isolates of the following microorganism, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Gram-Positive Bacteria
Staphylococcus aureus (Methicillin-susceptible isolates only)
INDICATIONS AND USAGE
Nafcillin is indicated in the treatment of infections caused by penicillinase-producing staphylococci which have demonstrated susceptibility to the drug. Culture and susceptibility tests should be performed initially to determine the causative organism and its susceptibility to the drug (see CLINICAL PHARMACOLOGY:Susceptibility Testing).
Nafcillin should not be used in infections caused by organisms susceptible to penicillin G. If the susceptibility tests indicate that the infection is due to methicillin-resistant Staphylococcus sp., therapy with Nafcillin for Injection, USP should be discontinued and alternative therapy provided.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of nafcillin for injection and other antibacterial drugs, nafcillin for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
CONTRAINDICATIONS
A history of a hypersensitivity (anaphylactic) reaction to any penicillin is a contraindication.
WARNINGS
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients on penicillin therapy. These reactions are more likely to occur in individuals with a history of penicillin hypersensitivity and/or a history of sensitivity to multiple allergens. Before initiating therapy with nafcillin, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, or other allergens. If an allergic reaction occurs, Nafcillin should be discontinued and appropriate therapy instituted.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including nafcillin for injection and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C.difficile.
C.difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C.difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C.difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C.difficile, and surgical evaluation should be instituted as clinically indicated.
PRECAUTIONS
General
Nafcillin should generally not be administered to patients with a history of sensitivity to any penicillin.
Penicillin should be used with caution in individuals with histories of significant allergies and/or asthma. Whenever allergic reactions occur, penicillin should be withdrawn unless, in the opinion of the physician, the condition being treated is life-threatening and amenable only to penicillin therapy.
The use of antibiotics may result in overgrowth of nonsusceptible organisms. If new infections due to bacteria or fungi occur, the drug should be discontinued and appropriate measures taken.
The liver/biliary tract is the primary route of nafcillin clearance. Caution should be exercised when patients with concomitant hepatic insufficiency and renal dysfunction are treated with nafcillin.
Prescribing nafcillin for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Laboratory Tests
Bacteriologic studies to determine the causative organisms and their susceptibility to the penicillinase-resistant penicillins should be performed (see CLINICAL PHARMACOLOGY: Microbiology). In the treatment of suspected staphylococcal infections, therapy should be changed to another active agent if culture tests fail to demonstrate the presence of staphylococci.
Periodic assessment of organ system function including renal, hepatic, and hematopoietic should be made during prolonged therapy with nafcillin. White blood cell and differential cell counts should be obtained prior to initiation of therapy and periodically during therapy with nafcillin. Urinalysis, serum blood urea nitrogen, and creatinine determinations should be performed at baseline and periodically during therapy with nafcillin. Serum bilirubin, SGOT, SGPT, alkaline phosphatase, and gamma glutamyl transferase should be obtained at baseline and periodically during therapy, especially when using high nafcillin doses. In patients with worsening hepatic function, the risk versus benefit of continued nafcillin use should be re-evaluated.
Drug Interactions
Tetracycline, a bacteriostatic antibiotic, may antagonize the bactericidal effect of penicillin, and concurrent use of these drugs should be avoided.
Nafcillin in high dosage regimens, i.e., 2 grams every 4 hours, has been reported to decrease the effects of warfarin. When nafcillin and warfarin are used concomitantly, the prothrombin time should be closely monitored and the dose of warfarin adjusted as necessary. This effect may persist for up to 30 days after nafcillin has been discontinued.
Nafcillin when administered concomitantly with cyclosporine has been reported to result in subtherapeutic cyclosporine levels. The nafcillin-cyclosporine interaction was documented in a patient during two separate courses of therapy. When cyclosporine and nafcillin are used concomitantly in organ transplant patients, the cyclosporine levels should be monitored.
Drug/Laboratory Test Interactions
Nafcillin in the urine can cause a false-positive urine reaction for protein when the sulfosalicyclic acid test is used, but not with the dipstick.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long term animal studies have been conducted with these drugs.
Studies on reproduction (nafcillin) in rats and mice reveal no fetal or maternal abnormalities before conception and continuously through weaning (one generation).
Pregnancy
Teratogenic Effects
Reproduction studies have been performed in the mouse with oral doses up to 20 times the human dose and orally in the rat at doses up to 40 times the human dose and have revealed no evidence of impaired fertility or harm to the rodent fetus due to nafcillin. There are, however, no adequate or well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, nafcillin should be used during pregnancy only if clearly needed.
Nursing Mothers
Penicillins are excreted in human milk. Caution should be exercised when penicillins are administered to a nursing woman.
Pediatric Use
The liver/biliary tract is the principal route of nafcillin elimination. Because of immature hepatic and renal function in pediatric patients, nafcillin excretion may be impaired. Safety and effectiveness in pediatric patients have not been established for the use of intravenous nafcillin. Safety and effectiveness in pediatric patients have been established for the use of intramuscular nafcillin.
Geriatric Use
Clinical studies of nafcillin for injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Nafcillin for injection contains 66.1 mg [2.9 mEq] of sodium per gram of nafcillin. At the usual recommended doses, patients would receive between 132.2 and 396.6 mg/day [5.8 and 17.4 mEq] of sodium. The geriatric population may respond with a blunted natriuresis to salt loading. This may be clinically important with regard to such diseases as congestive heart failure.
Information for Patients
Patients should be counseled that antibacterial drugs including nafcillin for injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When nafcillin for injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by nafcillin for injection or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
ADVERSE REACTIONS
Body as a Whole
The reported incidence of allergic reactions to penicillin ranges from 0.7 to 10 percent (see WARNINGS). Sensitization is usually the result of treatment, but some individuals have had immediate reactions to penicillin when first treated. In such cases, it is thought that the patients may have had prior exposure to the drug via trace amounts present in milk or vaccines. Two types of allergic reactions to penicillins are noted clinically, immediate and delayed.
Immediate reactions usually occur within 20 minutes of administration and range in severity from urticaria and pruritus to angioedema, laryngospasm, bronchospasm, hypotension, vascular collapse, and death. Such immediate anaphylactic reactions are very rare (see WARNINGS) and usually occur after parenteral therapy but have occurred in patients receiving oral therapy. Another type of immediate reaction, an accelerated reaction, may occur between 20 minutes and 48 hours after administration and may include urticaria, pruritus, and fever.
Although laryngeal edema, laryngospasm, and hypotension occasionally occur, fatality is uncommon. Delayed allergic reactions to penicillin therapy usually occur after 48 hours and sometimes as late as 2 to 4 weeks after initiation of therapy. Manifestations of this type of reaction include serum sickness-like symptoms (i.e., fever, malaise, urticaria, myalgia, arthralgia, abdominal pain) and various skin rashes. Nausea, vomiting, diarrhea, stomatitis, black or hairy tongue, and other symptoms of gastrointestinal irritation may occur, especially during oral penicillin therapy.
Local Reactions
Pain, swelling, inflammation, phlebitis, thrombophlebitis, and occasional skin sloughing at the injection site have occurred with intravenous administration of nafcillin (see DOSAGE AND ADMINISTRATION). Severe tissue necrosis with sloughing secondary to subcutaneous extravasation of nafcillin has been reported.
Nervous System Reactions
Neurotoxic reactions similar to those observed with penicillin G could occur with large intravenous or intraventricular doses of nafcillin especially in patients with concomitant hepatic insufficiency and renal dysfunction (see PRECAUTIONS).
Urogenital Reactions
Renal tubular damage and interstitial nephritis have been associated with the administration of nafcillin. Manifestations of this reaction may include rash, fever, eosinophilia, hematuria, proteinuria, and renal insufficiency.
Hepatic Reactions
Elevation of liver transaminases and/or cholestasis may occur, especially with administration of high doses of nafcillin.
Gastrointestinal Reactions
Pseudomembranous colitis has been reported with the use of nafcillin. The onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment (see WARNINGS).
OVERDOSAGE
Neurotoxic reactions similar to those observed with penicillin G may arise with intravenous doses of nafcillin especially in patients with concomitant hepatic insufficiency and renal dysfunction (see PRECAUTIONS).
In the case of overdosage, discontinue nafcillin, treat symptomatically and institute supportive measures as required. Hemodialysis does not increase the rate of clearance of nafcillin from the blood.
DOSAGE AND ADMINISTRATION
Nafcillin for injection is available for intramuscular and intravenous use.
The usual intravenous dosage for adults is 500 mg every 4 hours. For severe infections, 1 gram every 4 hours is recommended. Administer slowly over at least 30 to 60 minutes to minimize the risk of vein irritation and extravasation.
|
Drug |
Adults |
Infants and Children <40 kg (88 lbs) |
Other Recommendations |
|
Nafcillin |
500 mg Intramuscular every 4 to 6 hours Intravenous every 4 hours |
25 mg/kg Intramuscular twice daily |
Neonates 10 mg/kg Intramuscular twice daily |
|
Nafcillin |
1 gram Intramuscular or Intravenous every 4 hours (severe infections) |
Bacteriologic studies to determine the causative organisms and their susceptibility to nafcillin should always be performed. Duration of therapy varies with the type and severity of infection as well as the overall condition of the patient; therefore, it should be determined by the clinical and bacteriological response of the patient. In severe staphylococcal infections, therapy with nafcillin should be continued for at least 14 days. The treatment of endocarditis and osteomyelitis may require a longer duration of therapy.
No dosage alterations are necessary for patients with renal dysfunction, including those on hemodialysis. Hemodialysis does not accelerate nafcillin clearance from the blood.
With intravenous administration, particularly in elderly patients, care should be taken because of the possibility of thrombophlebitis.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Do not add supplementary medication to nafcillin.
DIRECTIONS FOR USE
For Intramuscular Use
Reconstitute with Sterile Water for Injection, USP, 0.9% Sodium Chloride Injection, USP or Bacteriostatic Water for Injection, USP (with benzyl alcohol or parabens); add 3.4 mL to the 1 g vial for 4 mL resulting solution and 6.5 mL to the 2 g vial for 8 mL resulting solution. All reconstituted vials have a concentration of 250 mg per mL.
The clear solution should be administered by deep intragluteal injection immediately after reconstitution.
Reconstituted Stability
Reconstitute with the required amount of Sterile Water for Injection, USP, 0.9% Sodium Chloride Injection, USP or Bacteriostatic Water for Injection, USP (with benzyl alcohol or parabens). The resulting solutions are stable for 3 days at room temperature or 7 days under refrigeration and 90 days frozen.
For Direct Intravenous Use
The required amount of drug should be diluted in 15 to 30 mL of Sterile Water for Injection, USP or Sodium Chloride Injection, USP and injected over a 5 to 10 minute period. This may be accomplished through the tubing of an intravenous infusion if desirable.
For Administration by Intravenous Drip
Reconstitute as directed above (For Intravenous Use) prior to diluting with intravenous solutions.
|
Concentration mg/mL |
Sterile Water for Injection, USP |
Sodium Chloride Injection, USP (0.9%) |
Sodium Lactate Solution, USP (M/6 Molar) |
Dextrose Injection, USP (5%) |
Dextrose and Sodium Chloride Injection, USP (5% Dextrose and 0.45% Sodium Chloride) |
Invert Sugar Injection, USP (10%) |
Lactated Ringer’s Injection, USP |
||
|
ROOM TEMPERATURE (25° C) |
|||||||||
|
10 to 200 |
24 Hours |
24 Hours | |||||||
|
30 |
24 Hours | ||||||||
|
2 to 30 |
24 Hours |
24 Hours | |||||||
|
10 to 30 |
24 Hours |
24 Hours |
|||||||
|
REFRIGERATION (4° C) |
|||||||||
|
10 to 200 |
7 Days |
7 Days | |||||||
|
10 to 30 |
7 Days |
7 Days |
7 Days |
7 Days |
7 Days |
||||
|
FROZEN (-15° C) |
|||||||||
|
250 |
90 Days |
90 Days | |||||||
|
10 to 250 |
90 Days |
90 Days |
90 Days |
90 Days |
90 Days |
||||
- * IMPORTANT: This chemical stability information in no way indicates that it would be acceptable practice to use this product well after the preparation time. Good professional practice suggests that a product should be used as soon after preparation as feasible.
Only those solutions listed above should be used for the intravenous infusion of nafcillin sodium, USP. The concentration of the antibiotic should fall within the range specified. The drug concentration and the rate and volume of the infusion should be adjusted so that the total dose of nafcillin is administered before the drug loses its stability in the solution in use.
There is no clinical experience available on the use of this agent in neonates or infants for this route of administration.
This route of administration should be used for relatively short-term therapy (24 to 48 hours) because of the occasional occurrence of thrombophlebitis particularly in elderly patients.
If another agent is used in conjunction with nafcillin therapy, it should not be physically mixed with nafcillin but should be administered separately.
HOW SUPPLIED
Nafcillin for Injection, USP contains nafcillin sodium as the monohydrate equivalent to 1 gram or 2 grams nafcillin per vial.
NDC 0781-3124-95 1 gram vial packaged in 10s
NDC 0781-3125-95 2 gram vial packaged in 10s
Other package sizes available:
NDC 0781-3126-95 Pharmacy Bulk Package bottle containing nafcillin sodium as the monohydrate equivalent to 10 grams of nafcillin, supplied in cartons of ten
Store dry powder at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Revised: December 2020
46275725
Manufactured by Sandoz GmbH for Sandoz Inc., Princeton, NJ 08540
| NAFCILLIN
nafcillin injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| NAFCILLIN
nafcillin injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Sandoz Inc (005387188) |