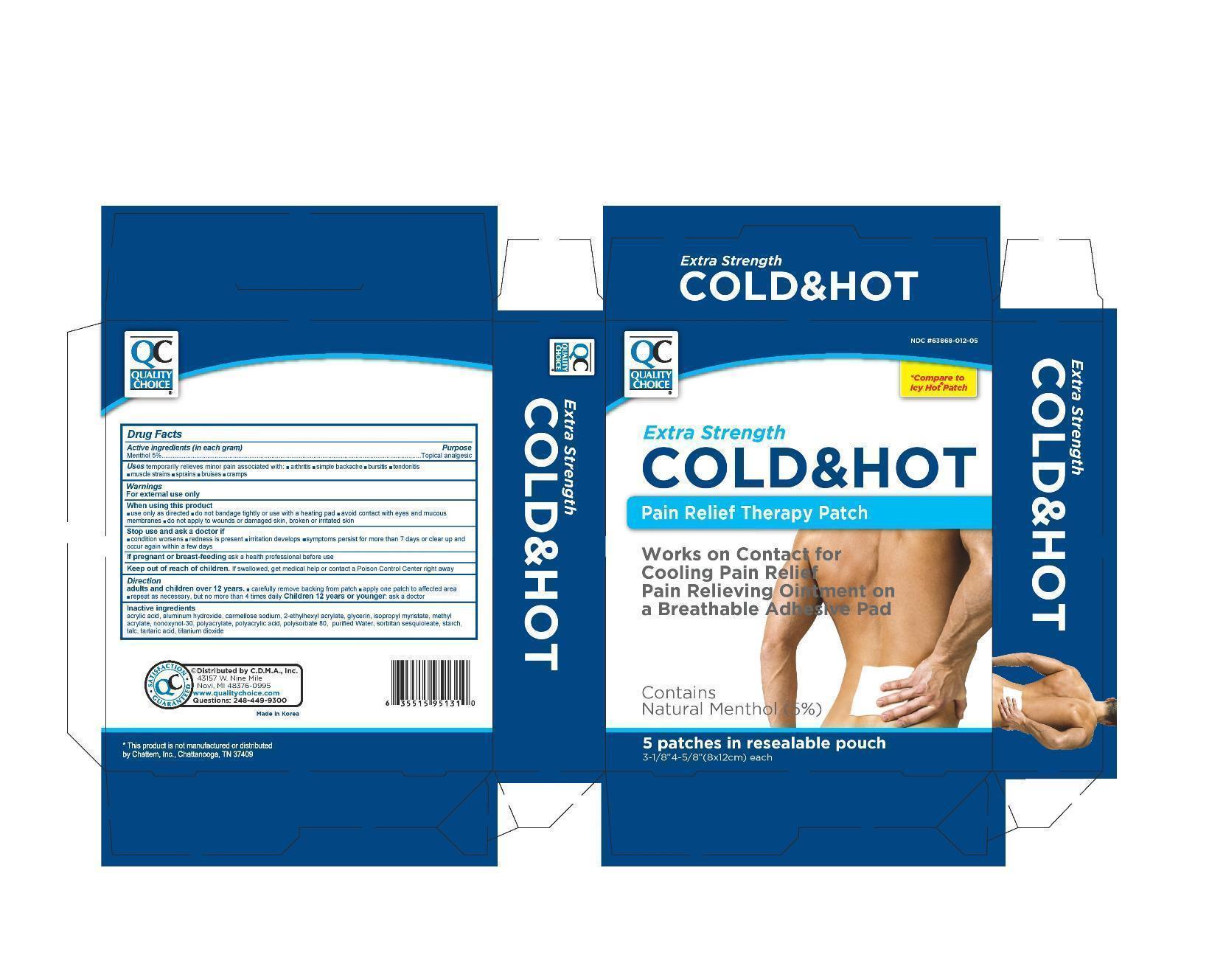
QC COLD AND HOT PAIN RELIEF- menthol patch
CHAIN DRUG MARKETING ASSOCIATION INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredient
Menthol 5.0%
Purpose
Topical Analgesic
Uses
temporarily relieves minor pain associated with
- arthritis
- simple backache
- bursitis
- tendonitis
- muscle strains
- sprains
- bruises
- cramps
Warnings
For external use only
When using this product
- use only as directed
- avoid contact with eyes and mucous membranes
- do not bandage tightly or use with a heating pad
- do not apply to wounds or damaged skin, broken or irritated skin
Stop use and ask a doctor if
- condition worsens
- redness is present
- irritation develops
- symptoms persist for more than 7 days or clear up and occur again within a few days
If pregnant or breast-feeding,
ask a health professional before use
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children over 12 years:
- carefully remove backing from patch
- apply one patch to affected area
- repeat as necessary, but no more than 4 times daily
Children 12 years or younger: ask a doctor
Inactive ingredients
Acrylic Acid, Aluminum Hydroxide, Carmellose sodium, 2-Ethylhexyl Acrylate, Glycerin, Isopropyl Myristate, Methyl Acrylate, Nonoxymol-30, Sodium Polyacrylate, Polyacrylic Acid, Polysorbate 80, Purified Water, Sorbitan Sesquioleate, Starch, Talc, Tartaric acid, Titanium Dioxide
package label
QC Cold and Hot Patch