TRAZODONE HCL- trazodone hydrochloride tablet
Mylan Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use trazodone hydrochloride tablets safely and effectively. See full prescribing information for trazodone hydrochloride tablets.
Trazodone Hydrochloride Tablets USP, for oral use Initial U.S. Approval: 1981 WARNING: SUICIDALITY AND ANTIDEPRESSANT DRUGSSee full prescribing information for complete boxed warning.
INDICATIONS AND USAGEDOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSBisectable tablets of 50 mg, 100 mg, 150 mg or 300 mg (3). CONTRAINDICATIONSNone (4). WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (incidence ≥ 5% and twice that of placebo) are: somnolence/sedation, dizziness, constipation, vision blurred (6). To report SUSPECTED ADVERSE REACTIONS, contact Mylan Pharmaceuticals Inc. at 1-877-4-INFO-RX (1-877-446-3679) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 07/2012 |
FULL PRESCRIBING INFORMATION
WARNING: SUICIDALITY AND ANTIDEPRESSANT DRUGS
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of trazodone hydrochloride tablets or any other antidepressant in a child, adolescent or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Trazodone hydrochloride tablets are not approved for use in pediatric patients [see Warnings and Precautions (5.1) and Patient Counseling Information (17.1)].
1 INDICATIONS AND USAGE
Trazodone hydrochloride tablets are indicated for the treatment of major depressive disorder (MDD) in adults. The efficacy of trazodone hydrochloride tablets has been established in trials with the immediate release formulation of trazodone hydrochloride [see Clinical Studies (14)].
2 DOSAGE AND ADMINISTRATION
The dosage should be initiated at a low-dose and increased gradually, noting the clinical response and any evidence of intolerance. Occurrence of drowsiness may require the administration of a major portion of the daily dose at bedtime or a reduction of dosage. Trazodone hydrochloride tablets should be taken shortly after a meal or light snack.
Dose Selection: An initial dose of 150 mg/day in divided doses is suggested. The dose may be increased by 50 mg/day every 3 to 4 days. The maximum dose for outpatients usually should not exceed 400 mg/day in divided doses. Inpatients (i.e., more severely depressed patients) may be given up to but not in excess of 600 mg/day in divided doses
- •
- Once an adequate response has been achieved, dosage may be gradually reduced, with subsequent adjustment depending on therapeutic response.
- •
- Patients should be monitored for withdrawal symptoms when discontinuing treatment with trazodone hydrochloride tablets. The dose should be gradually reduced whenever possible [see Warnings and Precautions (5.12)].
Maintenance Treatment: The efficacy of trazodone hydrochloride tablets for the maintenance treatment of MDD has not been evaluated. While there is no body of evidence available to answer the question of how long a patient treated with trazodone hydrochloride tablets should continue the drug, it is generally recommended that treatment be continued for several months after an initial response. Patients should be maintained on the lowest effective dose and be periodically reassessed to determine the continued need for maintenance treatment.
Important Administration Instructions: Trazodone hydrochloride tablets are scored to provide flexibility in dosing.
Trazodone hydrochloride tablets can be swallowed whole or administered as a half tablet by breaking the tablet along the score line.
3 DOSAGE FORMS AND STRENGTHS
Trazodone hydrochloride tablets, USP are available in the following strengths: 50 mg, 100 mg, 150 mg or 300 mg.
5 WARNINGS AND PRECAUTIONS
5.1 Clinical Worsening and Suicide Risk
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders and these disorders themselves are the strongest predictors of suicide. There has been a longstanding concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents and young adults (ages 18 to 24) with MDD and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD) or other psychiatric disorders included a total of 24 short-term trials of nine antidepressant drugs in over 4,400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs. placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1,000 patients treated) are provided in Table 1.
|
Age Range |
Drug-placebo Difference in Number of Cases of Suicidality per 1,000 Patients Treated |
|
Increases Compared to Placebo |
|
|
< 18 |
14 additional cases |
|
18 to 24 |
5 additional cases |
|
Decreases Compared to Placebo |
|
|
25 to 64 |
1 fewer case |
|
≥ 65 |
6 fewer cases |
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality and unusual changes in behavior, especially during the initial few months of a course of drug therapy or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset or were not part of the patient's presenting symptoms.
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to health care providers. Such monitoring should include daily observation by families and caregivers. Prescriptions for trazodone hydrochloride tablets should be written for the smallest quantity of tablets consistent with good patient management, in order to reduce the risk of overdose.
5.2 Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions
The development of a potentially life threatening serotonin syndrome or neuroleptic malignant syndrome (NMS)-like reactions have been reported with antidepressants alone and may occur with trazodone hydrochloride treatment, but particularly with concomitant use of other serotoninergic drugs (including SSRIs, SNRIs and triptans) and with drugs that impair metabolism of serotonin (including monoamine oxidase inhibitors [MAOIs]) or with antipsychotics or other dopamine antagonists. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations and coma), autonomic instability (e.g., tachycardia, labile blood pressure and hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination) and/or gastrointestinal symptoms (e.g., nausea, vomiting and diarrhea). Serotonin syndrome, in its most severe form, can resemble neuroleptic malignant syndrome, which includes hyperthermia, muscle rigidity, autonomic instability with possible rapid fluctuation of vital signs and mental status changes.
Treatment with trazodone hydrochloride tablets and any concomitant serotonergic or antidopaminergic agents, including antipsychotics, should be discontinued immediately if the above reactions occur and supportive symptomatic treatment should be initiated.
Trazodone hydrochloride tablets should not be used within 14 days of an MAOI [see Warnings and Precautions (5.8) and Drug Interactions (7)].
If concomitant treatment with trazodone hydrochloride tablets and an SSRI, SNRI or a 5- hydroxytryptamine receptor agonist (triptan) is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases.
The concomitant use of trazodone hydrochloride tablets with serotonin precursors (such as tryptophan) is not recommended.
5.3 Screening Patients for Bipolar Disorder and Monitoring for Mania/Hypomania
A major depressive episode may be the initial presentation of bipolar disorder. It is generally believed (though not established in controlled trials) that treating such an episode with an antidepressant alone may increase the likelihood of precipitation of a mixed/manic episode in patients at risk for bipolar disorder. Whether any of the symptoms described for clinical worsening and suicide risk represent such a conversion is unknown. However, prior to initiating treatment with an antidepressant, patients with depressive symptoms should be adequately screened to determine if they are at risk for bipolar disorder; such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder and depression. It should be noted that trazodone hydrochloride tablets are not approved for use in treating bipolar depression.
5.4 QT Prolongation and Risk of Sudden Death
Trazodone hydrochloride is known to prolong the QT/QTc interval. Some drugs that prolong the QT/QTc interval can cause Torsades de Pointes with sudden, unexplained death. The relationship of QT prolongation is clearest for larger increases (20 msec and greater), but it is possible that smaller QT/QTc prolongations may also increase risk, especially in susceptible individuals, such as those with hypokalemia, hypomagnesemia or a genetic predisposition to prolonged QT/QTc.
Although Torsades de Pointes has not been observed with the use of trazodone hydrochloride tablets at recommended doses in premarketing trials, experience is too limited to rule out an increased risk. However, there have been postmarketing reports of Torsades de Pointes with the immediate-release form of trazodone hydrochloride (in the presence of multiple confounding factors), even at doses of 100 mg per day or less.
5.5 Use in Patients with Heart Disease
Trazodone hydrochloride is not recommended for use during the initial recovery phase of myocardial infarction.
Caution should be used when administering trazodone hydrochloride tablets to patients with cardiac disease and such patients should be closely monitored, since antidepressant drugs (including trazodone hydrochloride) may cause cardiac arrhythmias.
QT prolongation has been reported with trazodone therapy [see Warnings and Precautions (5.4)]. Clinical studies in patients with preexisting cardiac disease indicate that trazodone hydrochloride may be arrhythmogenic in some patients in that population. Arrhythmias identified include isolated PVCs, ventricular couplets, tachycardia with syncope and Torsades de Pointes. Post-marketing events have been reported at doses of 100 mg or less with the immediate-release form of trazodone hydrochloride.
Concomitant administration of drugs that prolong the QT interval or that are inhibitors of CYP3A4 may increase the risk of cardiac arrhythmia.
5.6 Orthostatic Hypotension and Syncope
Hypotension, including orthostatic hypotension and syncope has been reported in patients receiving trazodone hydrochloride. Concomitant use with an antihypertensive may require a reduction in the dose of the antihypertensive drug.
5.7 Abnormal Bleeding
Post-marketing data have shown an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal (GI) bleeding. While no association between trazodone hydrochloride and bleeding events, in particular GI bleeding, was shown, patients should be cautioned about potential risk of bleeding associated with the concomitant use of trazodone and NSAIDs, aspirin or other drugs that affect coagulation or bleeding. Other bleeding events related to SSRIs and SNRIs have ranged from ecchymosis, hematoma, epistaxis and petechiae to life threatening hemorrhages.
5.8 Interaction with MAOIs
In patients receiving serotonergic drugs in combination with a monoamine oxidase inhibitor (MAOI), there have been reports of serious, sometimes fatal reactions including hyperthermia, rigidity, myoclonus, autonomic instability with rapid fluctuation in vital signs and mental status changes that include extreme agitation progressing to delirium and coma. These reactions have also been reported in patients who have recently discontinued antidepressant treatment and have been started on an MAOI. Some cases presented with features resembling neuroleptic malignant syndrome. Furthermore, limited animal data on the effects of combined use of serotonergic antidepressants and MAOIs suggest that these drugs may act synergistically to elevate blood pressure and evoke behavioral excitation. Therefore, it is recommended that trazodone hydrochloride tablets should not be used in combination with an MAOI or within 14 days of discontinuing treatment with an MAOI. Similarly, at least 14 days should be allowed after stopping trazodone hydrochloride tablets before starting an MAOI.
5.9 Priapism
Rare cases of priapism (painful erections greater than 6 hours in duration) were reported in men receiving trazodone hydrochloride. Priapism, if not treated promptly, can result in irreversible damage to the erectile tissue. Men who have an erection lasting greater than 6 hours, whether painful or not, should immediately discontinue the drug and seek emergency medical attention [see Adverse Reactions (6.2) and Overdosage (10)].
Trazodone hydrochloride should be used with caution in men who have conditions that might predispose them to priapism (e.g., sickle cell anemia, multiple myeloma or leukemia) or in men with anatomical deformation of the penis (e.g., angulation, cavernosal fibrosis or Peyronie's disease).
5.10 Hyponatremia
Hyponatremia may occur as a result of treatment with antidepressants. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH). Cases with serum sodium lower than 110 mmol/L have been reported. Elderly patients may be at greater risk of developing hyponatremia with antidepressants. Also, patients taking diuretics or who are otherwise volume-depleted can be at greater risk. Discontinuation of trazodone hydrochloride tablets should be considered in patients with symptomatic hyponatremia and appropriate medical intervention should be instituted.
Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness and unsteadiness, which can lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest and death.
5.11 Potential for Cognitive and Motor Impairment
Trazodone hydrochloride tablets may cause somnolence or sedation and may impair the mental and/or physical ability required for the performance of potentially hazardous tasks. Patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that the drug treatment does not affect them adversely.
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- •
- Clinical Worsening and Suicide Risk [see Boxed Warning and Warnings and Precautions (5.1)]
- •
- Serotonin Syndrome or NMS-like Reactions [see Warnings and Precautions (5.2)]
- •
- QT Prolongation and Risk of Sudden Death [see Warnings and Precautions (5.4)]
- •
- Orthostatic Hypotension [see Warnings and Precautions (5.6)]
- •
- Abnormal bleeding events [see Warnings and Precautions (5.7)]
- •
- Priapism [see Warnings and Precautions (5.9)]
- •
- Hyponatremia [see Warnings and Precautions (5.10)]
- •
- Cognitive and Motor Impairment [see Warnings and Precautions (5.11)]
- •
- Discontinuation symptoms [see Warnings and Precautions (5.12)]
The most common adverse reactions (reported in ≥ 5% and at twice the rate of placebo) are: Somnolence/sedation, dizziness, constipation, vision blurred.
Table 2 presents the summary of adverse events (AEs) leading to discontinuation of trazodone hydrochloride tablets treatment with an incidence of at least 1% and at least twice that for placebo.
|
Trazodone Hydrochloride N = 202 |
|
|
Somnolence/Sedation |
8 (4%) |
|
Dizziness |
7 (3.5%) |
|
Confusional state |
2 (1%) |
|
Coordination abnormal |
2 (1%) |
|
Headache |
2 (1%) |
|
Nausea |
2 (1%) |
|
Balance disorder / Gait disturbance |
2 (1%) |
6.1 Clinical Studies Experience
The table below is presented solely to indicate the relative frequency of adverse events reported in representative controlled clinical studies conducted to evaluate the safety and efficacy of trazodone hydrochloride.
The figures cited cannot be used to predict concisely the incidence of untoward events in the course of usual medical practice where patient characteristics and other factors often differ from those which prevailed in the clinical trials. These incidence figures, also, cannot be compared with those obtained from other clinical studies involving related drug products and placebo as each group of drug trials is conducted under a different set of conditions.
|
||||
|
Inpatients |
Outpatients |
|||
|
Trazodone Hydrochloride |
Placebo |
Trazodone Hydrochloride |
Placebo |
|
|
Number of Patients |
142 |
95 |
157 |
158 |
|
% of Patients Reporting | ||||
|
Allergic | ||||
|
Skin Condition/Edema |
2.8 |
1.1 |
7 |
1.3 |
|
Autonomic | ||||
|
Blurred Vision |
6.3 |
4.2 |
14.7 |
3.8 |
|
Constipation |
7 |
4.2 |
7.6 |
5.7 |
|
Dry Mouth |
14.8 |
8.4 |
33.8 |
20.3 |
|
Cardiovascular | ||||
|
Hypertension |
2.1 |
1.1 |
1.3 | |
|
Hypotension |
7 |
1.1 |
3.8 |
0 |
|
Shortness of Breath |
1.1 |
1.3 |
0 |
|
|
Syncope |
2.8 |
2.1 |
4.5 |
1.3 |
|
Tachycardia/ Palpitations |
0 |
0 |
7 |
7 |
|
CNS | ||||
|
Anger/Hostility |
3.5 |
6.3 |
1.3 |
2.5 |
|
Confusion |
4.9 |
0 |
5.7 |
7.6 |
|
Decreased Concentration |
2.8 |
2.1 |
1.3 |
0 |
|
Disorientation |
2.1 |
0 |
0 |
|
|
Dizziness/Light-headedness |
19.7 |
5.3 |
28 |
15.2 |
|
Drowsiness |
23.9 |
6.3 |
40.8 |
19.6 |
|
Excitement |
1.4 |
1.1 |
5.1 |
5.7 |
|
Fatigue |
11.3 |
4.2 |
5.7 |
2.5 |
|
Headache |
9.9 |
5.3 |
19.8 |
15.8 |
|
Insomnia |
9.9 |
10.5 |
6.4 |
12 |
|
Impaired Memory |
1.4 |
0 | ||
|
Nervousness |
14.8 |
10.5 |
6.4 |
8.2 |
|
Gastrointestinal | ||||
|
Abdominal/Gastric Disorder |
3.5 |
4.2 |
5.7 |
4.4 |
|
Bad Taste in Mouth |
1.4 |
0 |
0 |
0 |
|
Diarrhea |
0 |
1.1 |
4.5 |
1.9 |
|
Nausea/Vomiting |
9.9 |
1.1 |
12.7 |
9.5 |
|
Musculoskeletal | ||||
|
Musculoskeletal Aches/Pains |
5.6 |
3.2 |
5.1 |
2.5 |
|
Neurological | ||||
|
Incoordination |
4.9 |
0 |
1.9 |
0 |
|
Paresthesia |
1.4 |
0 |
0 | |
|
Tremors |
2.8 |
1.1 |
5.1 |
3.8 |
|
Sexual Function | ||||
|
Decreased Libido |
1.1 |
1.3 | ||
|
Other | ||||
|
Decreased Appetite |
3.5 |
5.3 |
0 | |
|
Eyes Red/Tired/Itching |
2.8 |
0 |
0 |
0 |
|
Head Full-Heavy |
2.8 |
0 |
0 |
0 |
|
Malaise |
2.8 |
0 |
0 |
0 |
|
Nasal/Sinus Congestion |
2.8 |
0 |
5.7 |
3.2 |
|
Nightmares/Vivid Dreams |
1.1 |
5.1 |
5.7 |
|
|
Sweating/Clamminess |
1.4 |
1.1 | ||
|
Tinnitus |
1.4 |
0 |
0 | |
|
Weight Gain |
1.4 |
0 |
4.5 |
1.9 |
|
Weight Loss |
3.2 |
5.7 |
2.5 |
|
Occasional sinus bradycardia has occurred in long-term studies.
In addition to the relatively common (i.e., greater than 1%) untoward events enumerated above, the following adverse events have been reported to occur in association with the use of trazodone hydrochloride in the controlled clinical studies: akathisia, allergic reaction, anemia, chest pain, delayed urine flow, early menses, flatulence, hallucinations/delusions, hematuria, hyper-salivation, hypomania, impaired speech, impotence, increased appetite, increased libido, increased urinary frequency, missed periods, muscle twitches, numbness and retrograde ejaculation.
6.2 Post-marketing Experience
Spontaneous reports regarding trazodone hydrochloride received from post-marketing experience include the following: abnormal dreams, agitation, alopecia, anxiety, aphasia, apnea, ataxia, breast enlargement or engorgement, cardiospasm, cerebrovascular accident, chills, cholestasis, clitorism, congestive heart failure, diplopia, edema, extrapyramidal symptoms, grand mal seizures, hallucinations, hemolytic anemia, hirsutism, hyperbilirubinemia, increased amylase, increased salivation, insomnia, leukocytosis, leukonychia, jaundice, lactation, liver enzyme alterations, methemoglobinemia, nausea/vomiting (most frequently), paresthesia, paranoid reaction, priapism [see Warnings and Precautions (5.9) and Patient Counseling Information (17.1)], pruritus, psoriasis, psychosis, rash, stupor, inappropriate ADH syndrome, tardive dyskinesia, unexplained death, urinary incontinence, urinary retention, urticaria, vasodilation, vertigo and weakness.
Cardiovascular system effects which have been reported include the following: conduction block, orthostatic hypotension and syncope, palpitations, bradycardia, atrial fibrillation, myocardial infarction, cardiac arrest, arrhythmia, ventricular ectopic activity, including ventricular tachycardia and QT prolongation. In post-marketing surveillance, prolonged QT interval, Torsades de Pointes and ventricular tachycardia have been reported with the immediate-release form of trazodone hydrochloride at doses of 100 mg per day or less [see Warnings and Precautions (5.4)].
7 DRUG INTERACTIONS
MAOIs: MAOIs should not be used within 14 days of trazodone hydrochloride [see Warnings and Precautions (5.8)].
Central Nervous System (CNS) Depressants: Trazodone hydrochloride may enhance the response to alcohol, barbiturates and other CNS depressants.
Cytochrome P450 3A4 Inhibitors: In vitro drug metabolism studies suggest that there is a potential for drug interactions when trazodone hydrochloride is given with cytochrome P450 3A4 (CYP3A4) inhibitors. The effect of short-term administration of ritonavir (200 mg twice daily, four doses) on the pharmacokinetics of a single dose of trazodone hydrochloride (50 mg) has been studied in 10 healthy subjects. The Cmax of trazodone hydrochloride increased by 34%, the AUC increased 2.4-fold, the half-life increased by 2.2-fold and the clearance decreased by 52%. Adverse effects including nausea, hypotension and syncope were observed when ritonavir and trazodone were coadministered. It is likely that ketoconazole, indinavir and other CYP3A4 inhibitors such as itraconazole may lead to substantial increases in trazodone hydrochloride plasma concentrations with the potential for adverse effects. If trazodone hydrochloride is used with a potent CYP3A4 inhibitor, the risk of cardiac arrhythmia may be increased [see Warnings and Precautions (5.4)] and a lower dose of trazodone hydrochloride should be considered.
Cytochrome P450 Inducers (e.g., Carbamazepine): Carbamazepine induces CYP3A4. Following coadministration of carbamazepine 400 mg per day with trazodone hydrochloride 100 mg to 300 mg daily, carbamazepine reduced plasma concentrations of trazodone hydrochloride and m-chlorophenlypiperazine (an active metabolite) by 76% and 60% respectively, compared to pre-carbamazepine values. Patients should be closely monitored to see if there is a need for an increased dose of trazodone hydrochloride when taking both drugs.
Digoxin and Phenytoin: Increased serum digoxin or phenytoin levels have been reported in patients receiving trazodone hydrochloride concurrently with either of these drugs. Monitor serum levels and adjust dosages as needed.
Serotonergic Drugs: Based on the mechanism of action of trazodone hydrochloride and the potential for serotonin syndrome, caution is advised when trazodone hydrochloride is coadministered with other drugs that may affect the neurotransmitter systems [see Warnings and Precautions (5.2)].
NSAIDs, Aspirin, or Other Drugs Affecting Coagulation or Bleeding: Due to a possible association between serotonin modulating drugs and gastrointestinal bleeding, patients should be monitored for and cautioned about the potential risk of bleeding associated with the concomitant use of trazodone hydrochloride and NSAIDs, aspirin or other drugs that affect coagulation or bleeding [see Warnings and Precautions (5.7)].
Warfarin: There have been reports of altered (either increased or decreased) prothrombin times in taking both warfarin and trazodone hydrochloride.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects
Pregnancy Category C
Trazodone hydrochloride has been shown to cause increased fetal resorption and other adverse effects on the fetus in two studies using the rat when given at dose levels approximately 30 to 50 times the proposed maximum human dose. There was also an increase in congenital anomalies in one of three rabbit studies at approximately 15 to 50 times the maximum human dose. There are no adequate and well controlled studies in pregnant women. Trazodone hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
8.3 Nursing Mothers
Trazodone hydrochloride and/or its metabolites have been found in the milk of lactating rats, suggesting that the drug may be secreted in human milk. Caution should be exercised when trazodone hydrochloride is administered to a nursing woman.
8.4 Pediatric Use
Safety and effectiveness in the pediatric population have not been established [see Boxed Warning and Warnings and Precautions (5.1)]. Trazodone hydrochloride should not be used in children or adolescents.
8.5 Geriatric Use
Reported clinical literature and experience with trazodone hydrochloride has not identified differences in responses between elderly and younger patients. However, as experience in the elderly with trazodone hydrochloride is limited, it should be used with caution in geriatric patients.
Antidepressants have been associated with cases of clinically significant hyponatremia in elderly patients who may be at greater risk for this adverse reaction [see Warnings and Precautions (5.10)].
9 DRUG ABUSE AND DEPENDENCE
9.2 Abuse
Although trazodone hydrochloride has not been systematically studied in preclinical or clinical studies for its potential for abuse, no indication of drug seeking behavior was seen in the clinical studies with trazodone hydrochloride. However, it is difficult to predict the extent to which a CNS-active drug will be misused, diverted and abused. Consequently, physicians should carefully evaluate patients for a history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of trazodone hydrochloride (e.g., development of tolerance, incrementation of dose, drug seeking behavior).
10 OVERDOSAGE
10.1 Human Experience
Death from overdose has occurred in patients ingesting trazodone hydrochloride and other CNS depressant drugs concurrently (alcohol; alcohol and chloral hydrate and diazepam; amobarbital; chlordiazepoxide; or meprobamate).
The most severe reactions reported to have occurred with overdose of trazodone hydrochloride alone have been priapism, respiratory arrest, seizures and ECG changes, including QT prolongation. The reactions reported most frequently have been drowsiness and vomiting. Overdosage may cause an increase in incidence or severity of any of the reported adverse reactions.
10.2 Management of Overdose
There is no specific antidote for trazodone hydrochloride overdose.
Treatment should consist of those general measures employed in the management of overdosage with any drug effective in the treatment of major depressive disorder.
Ensure an adequate airway, oxygenation and ventilation. Monitor cardiac rhythm and vital signs.
General supportive and symptomatic measures are also recommended. Induction of emesis is not recommended. Gastric lavage with a large bore orogastric tube with appropriate airway protection, if needed, may be indicated if performed soon after ingestion or in symptomatic patients. Activated charcoal should be administered. Forced diuresis may be useful in facilitating elimination of the drug.
In managing overdosage, consider the possibility of multiple drug involvement. The physician should consider contacting a poison control center for additional information on the treatment of any overdose.
11 DESCRIPTION
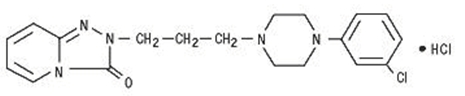
Trazodone hydrochloride, USP is a triazolopyridine. It is a white odorless crystalline powder which is freely soluble in water.
Chemical Name: 2-[3-[4-(3-chlorophenyl)-1-piperazinyl]propyl]-1,2,4-triazolo[4,3-a]pyridin-3(2H)-one hydrochloride
Structural Formula:
Molecular Formula: C19H22 IN5O·HCl
Molecular Weight: 408.33
Each tablet, for oral administration, contains 50 mg, 100 mg, 150 mg or 300 mg of trazodone hydrochloride, USP. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide magnesium stearate, microcrystalline cellulose, pregelatinized starch, sodium lauryl sulfate, sodium starch glycolate and yellow iron oxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of trazodone hydrochloride’s antidepressant action is not fully understood, but is thought to be related to its potentiation of serotonergic activity in the CNS.
12.2 Pharmacodynamics
Preclinical studies have shown that trazodone hydrochloride selectively inhibits neuronal reuptake of serotonin and acts as an antagonist at 5-HT-2A/2C serotonin receptors.
Trazodone hydrochloride is not a monoamine oxidase inhibitor and, unlike amphetamine-type drugs, does not stimulate the central nervous system.
Trazodone hydrochloride antagonizes alpha 1-adrenergic receptors, a property which may be associated with postural hypotension.
12.3 Pharmacokinetics
Absorption: In humans, trazodone hydrochloride is well absorbed after oral administration without selective localization in any tissue. When trazodone hydrochloride is taken shortly after ingestion of food, there may be an increase in the amount of drug absorbed, a decrease in maximum concentration and a lengthening in the time to maximum concentration. Peak plasma levels occur approximately one hour after dosing when trazodone hydrochloride is taken on an empty stomach or 2 hours after dosing when taken with food.
Metabolism: In vitro studies in human liver microsomes show that trazodone hydrochloride is metabolized, via oxidative cleavage, to an active metabolite, m-chlorophenylpiperazine (mCPP) by CYP3A4. Other metabolic pathways that may be involved in the metabolism of trazodone hydrochloride have not been well characterized. Trazodone hydrochloride is extensively metabolized; less than 1% of an oral dose is excreted unchanged in the urine.
Elimination: In some patients trazodone hydrochloride may accumulate in the plasma.
Protein Binding: Trazodone is 89% to 95% protein bound in vitro at concentrations attained with therapeutic doses in humans.
14 CLINICAL STUDIES
The efficacy and safety of trazodone hydrochloride was established from both inpatient and outpatient trials of the trazodone hydrochloride immediate release formulation in the treatment of major depressive disorder.
16 HOW SUPPLIED/STORAGE AND HANDLING
Trazodone Hydrochloride Tablets, USP are available containing 50 mg, 100 mg, 150 mg or 300 mg of trazodone hydrochloride, USP.
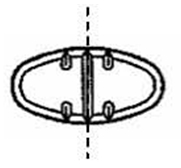
The 50 mg tablets are light yellow round, scored tablets debossed with MX above the score and 71 below the score on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-3471-01
bottles of 100 tablets
NDC 0378-3471-10
bottles of 1000 tablets
The 100 mg tablets are light yellow round, scored tablets debossed with MX above the score and 72 below the score on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-3472-01
bottles of 100 tablets
NDC 0378-3472-10
bottles of 1000 tablets
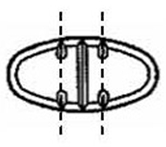
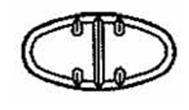
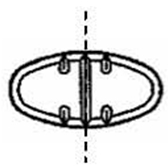
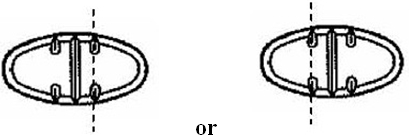
The 150 mg tablets are light yellow oval, scored tablets debossed with MX 73 on one side of the tablet and one full bisect and two partial trisects on the other side. They are available as follows:
NDC 0378-3473-01
bottles of 100 tablets
NDC 0378-3473-10
bottles of 1000 tablets
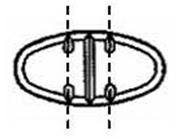
The 300 mg tablets are light yellow oval, scored tablets debossed with MX 74 on one side of the tablet and one full bisect and two partial trisects on the other side. They are available as follows:
NDC 0378-3474-01
bottles of 100 tablets
NDC 0378-3474-05
bottles of 500 tablets
Directions for using the correct score when breaking the 150 mg tablet, please refer to the following:
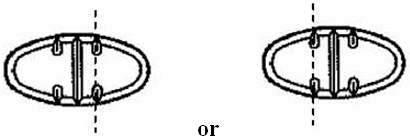
For 50 mg, break the score on either the left or right side of the tablet (one-third of a tablet).
For 75 mg, break the score down the middle of the tablet (one-half of a tablet).
For 100 mg, break the score on either the left or right side of the tablet (two-thirds of a tablet).
For 150 mg, use the entire tablet.
Directions for using the correct score when breaking the 300 mg tablet, please refer to the following:
For 100 mg, break the score on either the left or right side of the tablet (one-third of a tablet).
For 150 mg, break the score down the middle of the tablet (one-half of a tablet).
For 200 mg, break the score on either the left or right side of the tablet (two-thirds of a tablet).
For 300 mg, use the entire tablet.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
17 PATIENT COUNSELING INFORMATION
See Medication Guide (17.2).
17.1 Information for Patients
Prescribers or other health professionals should inform patients, their families and their caregivers about the benefits and risks associated with treatment with trazodone hydrochloride tablets and should counsel them in its appropriate use.
Patients should be warned that:
- •
- There is a potential for increased risk of suicidal thoughts especially in children, teenagers and young adults.
- •
- The following symptoms should be reported to the physician: anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia, hypomania and mania.
- •
- They should inform their physician if they have a history of bipolar disorder, cardiac disease or myocardial infarction.
- •
- Serotonin syndrome could occur and symptoms may include changes in mental status (e.g., agitation, hallucinations and coma), autonomic instability (e.g., tachycardia, labile blood pressure and hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination) and/or gastrointestinal symptoms (e.g., nausea, vomiting and diarrhea).
- •
- Trazodone hydrochloride has been associated with the occurrence of priapism.
- •
- There is a potential for hypotension, including orthostatic hypotension and syncope.
- •
- There is a potential risk of bleeding (including life threatening hemorrhages) and bleeding related events (including ecchymosis, hematoma, epistaxis, and petechiae) with the concomitant use of trazodone hydrochloride and NSAIDs, aspirin or other drugs that affect coagulation or bleeding.
- •
- Withdrawal symptoms including anxiety, agitation and sleep disturbances, have been reported with trazodone hydrochloride. Clinical experience suggests that the dose should be gradually reduced.
Patients should be counseled that:
- •
- Trazodone hydrochloride tablets may cause somnolence or sedation and may impair the mental and/or physical ability required for the performance of potentially hazardous tasks. Patients should be cautioned about operating hazardous machinery, including automobiles until they are reasonably certain that the drug treatment does not affect them.
- •
- Trazodone hydrochloride tablets may enhance the response to alcohol, barbiturates and other CNS depressants.
- •
- Women who intend to become pregnant or who are breast-feeding should discuss with a physician whether they should continue to use trazodone hydrochloride tablets, since use in pregnant and nursing women is not recommended.
Important Administration Instructions:
- •
- Trazodone hydrochloride tablets should be swallowed whole or broken in half along the score line.
- •
- Trazodone hydrochloride tablets should be taken shortly after a meal or light snack.
MEDICATION GUIDE
TRAZODONE HYDROCHLORIDE TABLETS, USP
50 mg, 100 mg, 150 mg and 300 mg
(traz′ oh done hye″ droe klor′ ide)
Read the Medication Guide that comes with trazodone hydrochloride tablets before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk to your healthcare provider or pharmacist if there is something you do not understand or you want to learn about trazodone hydrochloride tablets.
What is the most important information I should know about trazodone hydrochloride tablets?
Antidepressant medicines, depression or other serious mental illnesses, and suicidal thoughts or actions: Talk to your healthcare provider about:
- •
- All risks and benefits of treatment with antidepressant medicines
- •
- All treatment choices for depression or other serious mental illnesses
1. Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers and young adults within the first few months of treatment.
2. Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a higher risk of having suicidal thoughts or actions. These include people who have or have a family history of bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
3. How can I watch for and try to prevent suicidal thoughts and actions?
- •
- Pay close attention to any changes, especially sudden changes in mood, behaviors, thoughts or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
- •
- Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts or feelings.
- •
- Keep all follow-up visits with your healthcare provider as scheduled. Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
Call a healthcare provider right away if you have any of the following symptoms, especially if they are new, worse or worry you:
- •
- Thoughts about suicide or dying
- •
- Attempts to commit suicide
- •
- New or worse depression
- •
- New or worse anxiety
- •
- Feeling very agitated or restless
- •
- Panic attacks
- •
- Trouble sleeping (insomnia)
- •
- New or worse irritability
- •
- Acting aggressive, being angry or violent
- •
- Acting on dangerous impulses
- •
- An extreme increase in activity and talking (mania)
- •
- Other unusual changes in behavior or mood
What else do I need to know about antidepressant medicines?
Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. You should discuss all treatment choices with your healthcare provider, not just the use of antidepressants.
Antidepressant medicines have other side effects. Talk to your healthcare provider about the side effects of your medicines.
Antidepressant medicines can interact with other medicines. Know all of the medicines that you take. Keep a list of all medicines to show your healthcare provider. Do not start new medicines without first checking with your healthcare provider.
4. Trazodone hydrochloride tablets are not approved for use in children. Talk to your healthcare provider for more information.
What are trazodone hydrochloride tablets?
Trazodone hydrochloride tablets are a prescription medicine used to treat major depressive disorder in adults.
What should I tell my healthcare provider before taking trazodone hydrochloride tablets?
Before you take trazodone hydrochloride tablets tell your healthcare provider if you:
- •
- have heart problems, including QT prolongation or a family history of it
- •
- have ever had a heart attack
- •
- have bipolar disorder
- •
- have liver or kidney problems
- •
- have other serious medical conditions
- •
- are pregnant or plan to become pregnant. Trazodone hydrochloride tablets may harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- •
- are breast-feeding or plan to breast-feed. It is not known if trazodone hydrochloride passes into your breast milk. You and your healthcare provider should decide if you will take trazodone hydrochloride tablets or breast-feed.
- •
- have taken a Monoamine Oxidase Inhibitor (MAOI) or if you have stopped taking an MAOI in the last 2 weeks.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements.
Using trazodone hydrochloride tablets with certain other medicines can affect each other causing serious side effects.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take trazodone hydrochloride tablets?
- •
- Take trazodone hydrochloride tablets exactly as your healthcare provider tells you.
- •
- Trazodone hydrochloride tablets should be taken shortly after a meal or light snack.
- •
- If you feel drowsy after taking trazodone hydrochloride tablets, talk to your healthcare provider. Your healthcare provider may change your dose or the time of day you take your trazodone hydrochloride tablets.
- •
- Do not stop taking trazodone hydrochloride tablets without talking to your healthcare provider.
- •
- Trazodone hydrochloride tablets should be swallowed whole or broken in half along the score line. Do not chew or crush trazodone hydrochloride tablets. Tell your healthcare provider if you cannot swallow trazodone either whole or as a half tablet.
- •
- If you take too much trazodone hydrochloride, call your doctor or go to the nearest emergency room right away.
What should I avoid while taking trazodone hydrochloride tablets?
- •
- Do not drive, operate heavy machinery or do other dangerous activities until you know how trazodone hydrochloride tablets affect you. Trazodone hydrochloride tablets can slow your thinking and motor skills.
- •
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking trazodone hydrochloride tablets until you talk with your healthcare provider. Trazodone hydrochloride tablets may make your sleepiness or dizziness worse if you take it with alcohol or other medicines that cause sleepiness or dizziness.
What are the possible side effects of trazodone hydrochloride tablets?
Trazodone hydrochloride tablets can cause serious side effects or death. See “What is the most important information I should know about trazodone hydrochloride tablets?”
Serious side effects include:
- •
- Serotonin syndrome. Symptoms of serotonin syndrome include: agitation, hallucinations, problems with coordination, fast heartbeat, tight muscles, trouble walking, nausea, vomiting, diarrhea.
- •
- Feeling high or in a very good mood, then becoming irritable or having too much energy, feeling like you have to keep talking or do not sleep (Mania).
- •
- Irregular or fast heartbeat or faint (QT prolongation).
- •
- Low blood pressure. You feel dizzy or faint when you change positions (go from sitting to standing).
- •
- Unusual bruising or bleeding.
- •
- Erection lasting for more than 6 hours (Priapism).
- •
- Low sodium in your blood (Hyponatremia). Symptoms of hyponatremia include: headache, feeling weak, feeling confused, trouble concentrating, memory problems and feeling unsteady when you walk.
- •
- Withdrawal symptoms. Symptoms of withdrawal can include anxiety, agitation and sleep problems. Do not stop taking trazodone hydrochloride tablets without talking to your healthcare provider.
Get medical help right away, if you have any of the symptoms listed above.
The most common side effects of trazodone hydrochloride tablets include:
- •
- Sleepiness
- •
- Dizziness
- •
- Constipation
- •
- Blurry vision
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of trazodone hydrochloride tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800- FDA-1088.
How should I store trazodone hydrochloride tablets?
- •
- Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.]
- •
- Keep in tight container
- •
- Keep out of the light
- •
- Safely throw away medicine that is out of date or no longer needed.
Keep trazodone hydrochloride tablets and all medicines out of the reach of children.
General information about the safe and effective use of trazodone hydrochloride tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use trazodone hydrochloride tablets for a condition for which it was not prescribed. Do not give trazodone hydrochloride tablets to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about trazodone hydrochloride tablets. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about trazodone hydrochloride tablets that is written for health professionals.
For more information, call Mylan Pharmaceuticals Inc. at 1-877-4-INFO-RX (1-877-446-3679).
What are the ingredients in trazodone hydrochloride tablets?
Active ingredient: trazodone hydrochloride, USP
Inactive ingredients: colloidal silicon dioxide magnesium stearate, microcrystalline cellulose, pregelatinized starch, sodium lauryl sulfate, sodium starch glycolate and yellow iron oxide.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Manufactured in India by:
Mylan Laboratories Limited
Hyderabad—500 034, India
Code No. MH/DRUGS/25/NKD/89
75050221
REVISED JULY 2012
MX:TRZN:R3mmt
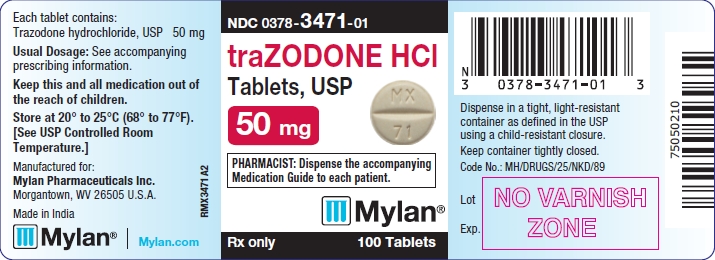
PRINCIPAL DISPLAY PANEL - 50 mg
NDC 0378-3471-01
traZODONE HCl
Tablets, USP
50 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Rx only 100 Tablets
Each tablet contains:
Trazodone hydrochloride, USP 50 mg
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMX3471A2
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/25/NKD/89
PRINCIPAL DISPLAY PANEL - 100 mg
NDC 0378-3472-01
traZODONE HCl
Tablets, USP
100 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Rx only 100 Tablets
Each tablet contains:
Trazodone hydrochloride, USP 100 mg
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMX3472A2
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/25/NKD/89
PRINCIPAL DISPLAY PANEL - 150 mg
NDC 0378-3473-01
traZODONE HCl
Tablets, USP
150 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Rx only 100 Tablets
Each tablet contains:
Trazodone hydrochloride, USP 150 mg
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of the
reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room Temperature.]
Note to Pharmacist: If other than a
whole tablet is to be taken, instruct
patient as to the correct score to
use when breaking the tablet.
See HOW SUPPLIED section.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMX3473A2
Dispense in a tight, light-resistant container
as defined in the USP using a child-resistant
closure.
Keep container tightly closed.
Code No.: MH/DRUGS/25/NKD/89
PRINCIPAL DISPLAY PANEL - 300 mg
NDC 0378-3474-01
traZODONE HCl
Tablets, USP
300 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Rx only 100 Tablets
Each tablet contains:
Trazodone hydrochloride, USP 300 mg
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of the
reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room Temperature.]
Note to Pharmacist: If other than a
whole tablet is to be taken, instruct
patient as to the correct score to
use when breaking the tablet.
See HOW SUPPLIED section.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMX3474A2
This container is not intended for dispensing for
household use.
Dispense in a tight, light-resistant container as
defined in the USP using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/25/NKD/89
| TRAZODONE HCL
trazodone hydrochloride tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| TRAZODONE HCL
trazodone hydrochloride tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| TRAZODONE HCL
trazodone hydrochloride tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| TRAZODONE HCL
trazodone hydrochloride tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Mylan Pharmaceuticals Inc. (059295980) |