GALLIUM CITRATE GA-67- gallium chloride ga-67 injection, solution
Mallinckrodt Inc.
----------
Gallium Citrate Ga 67 Injection
Rx Only
Diagnostic – For Intravenous Use
DESCRIPTION
Gallium Citrate Ga 67 Injection is supplied in a 10 milliliter vial as an isotonic, sterile, non-pyrogenic solution. Each milliliter of the isotonic solution contains 74 megabecquerels (2 millicuries) of Gallium Ga 67 on the calibration date as a complex formed from 8.3 nanograms gallium chloride Ga 67, 1.9 milligrams of sodium citrate dihydrate, 7.8 milligrams of sodium chloride and 0.9 percent benzyl alcohol (v/v) as a preservative. The pH is adjusted to between 5.5 to 8.0 with hydrochloric acid and/or sodium hydroxide solution.
Gallium Ga 67, with a half-life of 78.26 hours, is cyclotron produced by the proton irradiation of enriched zinc. At the time of calibration the drug contains no more than 0.02% Gallium Ga 66 and no more than 0.2% Zinc Zn 65. The concentration of each radionuclidic impurity changes with time. At expiration, the drug contains no more than 0.001% Gallium Ga 66 and no more than 1.0% Zinc Zn 65. No carrier has been added.
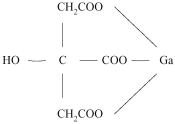
Gallium Citrate has the following chemical structure:
Physical Characteristics
Gallium Ga 67 with a physical half-life of 78.26 hours1 decays by electron capture to stable Zinc Zn 67. Photons that are useful for imaging studies are listed in Table 1.
| Radiation | Mean Percent Per Disintegration | Energy (keV) |
| Gamma-2 | 2.9 | 91.3 |
| Gamma-3 | 35.7 | 93.3 |
| Gamma-4 | 19.7 | 184.6 |
| Gamma-5 | 2.2 | 209.0 |
| Gamma-6 | 16.0 | 300.2 |
| Gamma-7 | 4.5 | 393.5 |
- 1
- Kocher, D.C., Radioactive Decay Data Tables, Health and Safety Research Division, National Technical Information Service, DOE/TIC-11026, pg. 80, 1981.
CLINICAL PHARMACOLOGY
Gallium Citrate Ga 67, with no carrier added, has been found to concentrate in certain viable primary and metastatic tumors as well as focal sites of infection. The mechanism of concentration is unknown, but investigational studies have shown that Gallium Ga 67 accumulates in lysosomes and is bound to a soluble intracellular protein.
It has been reported in the scientific literature that following intravenous injection, the highest tissue concentration of Gallium Ga 67 - other than tumors and sites of infection - is the renal cortex. After the first day, the maximum concentration shifts to bone and lymph nodes and after the first week, to liver and spleen. Gallium Ga 67 is excreted relatively slowly from the body. The average whole body retention is 65 percent after seven days, with 26 percent having been excreted in the urine and 9 percent in the stools.
INDICATIONS AND USAGE
Gallium Citrate Ga 67 Injection may be useful to demonstrate the presence and extent of Hodgkin's disease, lymphoma, and bronchogenic carcinoma. Positive Gallium Ga 67 uptake in the absence of prior symptoms warrants follow-up as an indication of a potential disease state. Gallium Citrate Ga 67 Injection may be useful as an aid in detecting some acute inflammatory lesions.
PRECAUTIONS
General
A thorough knowledge of the normal distribution of intravenously administered Gallium Citrate Ga 67 Injection is essential in order to accurately interpret pathologic states. The finding of an abnormal Gallium Ga 67 concentration usually implies the existence of underlying pathology, but further diagnostic studies should be done to distinguish benign from malignant lesions. Gallium Citrate Ga 67 injection is intended for use as an adjunct in the diagnosis of certain neoplasms as well as focal areas of infection. Certain pathologic conditions may yield up to 40 percent false negative Gallium Ga 67 studies. Therefore, a negative study cannot be definitely interpreted as ruling out the presence of disease.
Lymphocytic lymphoma frequently does not accumulate Gallium Ga 67 sufficiently for unequivocal imaging and the use of gallium with this histologic type of lymphoma is not recommended at this time.
Gallium Ga 67 localization cannot differentiate between tumor and acute inflammation, and other diagnostic studies must be added to define the underlying pathology.
As in the use of any radioactive material, care should be taken to minimize radiation exposure to the patient consistent with proper management and to insure minimum radiation exposure to occupational workers.
The vial contents are sterile and non-pyrogenic. It is essential that the user follow the directions carefully and adhere to strict aseptic procedures.
Radiopharmaceuticals should be used only by physicians who are qualified by training and experience in the safe use and handling of radionuclides produced by nuclear reactor or particle accelerator and whose experience and training have been approved by the appropriate government agency authorized to license the use of radionuclides.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic or mutagenic potential or whether this drug affects fertility in males or females.
Pregnancy Category C
Animal reproductive studies have not been conducted with Gallium Citrate Ga 67. It is also not known whether Gallium Citrate Ga 67 can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Gallium Citrate Ga 67 should be given to a pregnant woman only if clearly needed.
Ideally, examinations using radiopharmaceuticals, especially those elective in nature of women of childbearing capability, should be performed during the first few (approximately ten) days following the onset of menses.
ADVERSE REACTIONS
Rare occurrences of allergic reactions, skin rash and nausea have been reported in association with Gallium Citrate Ga 67 use.
DOSAGE AND ADMINISTRATION
The recommended adult (70 kg) dose of Gallium Citrate Ga 67 Injection is 74 to 185 megabecquerels (2 to 5 millicuries). Gallium Citrate Ga 67 Injection is intended for intravenous administration only.
Approximately 10 percent of the administered dose is excreted in the feces during the first week after injection. Daily laxatives and/or enemas are recommended from the day of injection until the final images are obtained in order to cleanse the bowel of radioactive material and minimize the possibility of false positive studies.
Studies indicate the optimal tumor to background concentration ratios are often obtained 48 hours post injection. However, considerable biological variability may occur in individuals and acceptable images may be obtained as early as 6 hours and as late as 120 hours after injection.
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use if contents are turbid.
Instructions for the handling of Gallium Citrate Ga 67:
- Waterproof gloves should be used during the entire handling and administration procedure.
- Using proper shielding, the vial containing the Gallium Citrate Ga 67 should be visually inspected to insure that it is free of particulate matter and discoloration prior to use.
- Maintain adequate shielding during the life of the product and use a sterile, shielded syringe for withdrawing and injecting the preparation.
Radiation Dosimetry
The estimated absorbed radiation doses2 from an intravenous injection of 185 megabecquerels (5 millicuries) of Gallium Citrate Ga 67 are shown in Table 4.
| Tissue | mGy/ 185MBq | rads/ 5mCi |
| Whole Body | 13.0 | 1.30 |
| Skeleton | 22.0 | 2.20 |
| Liver | 23.0 | 2.30 |
| Bone Marrow | 29.0 | 2.90 |
| Spleen | 26.5 | 2.65 |
| Kidney | 20.5 | 2.05 |
| Ovaries | 14.0 | 1.40 |
| Testes | 12.0 | 1.20 |
| Gastrointestinal Tract | ||
| Stomach | 11.0 | 1.10 |
| Small Intestine | 18.0 | 1.80 |
| Upper Large Intestine | 28.0 | 2.80 |
| Lower Large Intestine | 45.0 | 4.50 |
- 2
- MIRD Dose Estimate Report No. 2, J. Nucl. Med. 14; 755-6 (1973).
HOW SUPPLIED
Catalog Number 180.
Gallium Citrate Ga 67 Injection is supplied sterile and non-pyrogenic for intravenous use. Each milliliter contains 74 megabecquerels (2 millicuries) of Gallium Ga 67 on the calibration date, as a complex formed from 8.3 nanograms gallium chloride Ga 67, 1.9 milligrams of sodium citrate dihydrate, 7.8 milligrams of sodium chloride, and 0.9 percent benzyl alcohol (v/v) as a preservative. The pH is adjusted to between 5.5 to 8.0 with hydrochloric acid and/or sodium hydroxide solution.
Gallium Citrate Ga 67 Injection is available in vials containing the following amounts on the calibration date.
Catalog No.
N180G0 222 megabecquerels (6 mCi)
NDC 0019-N180-G0
N180M0 444 megabecquerels (12 mCi)
NDC 0019-N180-M0
Storage and Handling
The contents of the vial are radioactive, and adequate shielding and handling precautions must be maintained. Store at controlled room temperature 20° to 25°C
(68° to 77°F) [see USP].
Storage and disposal of Gallium Citrate Ga 67 Injection should be controlled in a manner that is in compliance with the appropriate regulations of the government agency authorized to license the use of this radionuclide.
Manufactured by:
Mallinckrodt Inc.
St. Louis, MO 63134 USA
Distributed in Canada by:
tyco Healthcare
Pointe-Claire, QC, Canada H9R 5H8
Estb. Lic. No.: 100689-A
A180I0
Rev 04/2012
tyco
Healthcare
Mallinckrodt
PRINCIPAL DISPLAY PANEL - A180C0
Gallium Citrate Ga 67 Injection
DIAGNOSTIC
Sterile, Non-Pyrogenic Solution
For Intravenous Administration
Store at Controlled Room Temperature 20-25°C (68-77°F).
Each mililiter contains 74 MBq (2 mCi) Gallium Citrate Ga 67 (essentially carrier free) at date and time of calibration, as a complex formed from 8.3 ng gallium chloride Ga 67, 1.9 mg sodium citrate dihydrate, 7.8 mg sodium chloride, and 0.9% (v/v), benzyl alcohol as a preservative. Sodium hydroxide or hydrochloric acid are added for pH adjustment. The pH is between 5.5 and 8.0.
For information on dosage, administration and indications see package insert.
Rx Only.
WARNING: Radioactive drugs must be handled only by qualified personnel in conformity with regulations of the U.S. Nuclear Regulatory Commission or state regulatory agencies where applicable. Bottle containing drug should be kept in this container or within heavier shield.
Distributed in Canada by:
tyco Healthcare
Pointe-Claire, QC, Canada H9R 5H8
Estb. Lic. No.: 100689-A
Manufactured by:
Mallinckrodt Inc.
St. Louis, MO 63134 USA
MALLINCKRODT
CAUTION RADIOACTIVE MATERIAL
A180C0
R12/2011

| GALLIUM CITRATE GA-67
gallium citrate ga-67 injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Mallinckrodt Inc. (047021092) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Mallinckrodt Inc. | 557570652 | ANALYSIS(0019-N180), API MANUFACTURE(0019-N180), MANUFACTURE(0019-N180) | |