HEARTSHIELD PLUS- ivermectin and pyrantel pamoate tablet, chewable
TruRX LLC
----------
HEARTSHIELD™ plus
(ivermectin/pyrantel)
Caution
Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian.
INDICATIONS
For use in dogs to prevent canine heartworm disease by eliminating the tissue stage of heartworm larvae (Dirofilaria immitis) for a month (30 days) after infection and for the treatment and control of ascarids (Toxocara canis, Toxascaris leonina) and hookworms (Ancylostoma caninum, Uncinaria stenocephala, Ancylostoma braziliense).
DOSAGE
HEARTSHIELD PLUS™ (ivermectin/pyrantel) Flavored Chewables should be administered orally at monthly intervals at the recommended minimum dose level of 6 mcg of ivermectin per kilogram (2.72 mcg/lb) and 5 mg of pyrantel (as pamoate salt) per kg (2.27 mg/lb) of body weight. The recommended dosing schedule for prevention of canine heartworm disease and for the treatment and control of ascarids and hookworms is as follows:
| Dog Weight | Flavored Chewable Per Month | Ivermectin Content | Pyrantel Content |
|---|---|---|---|
| Up to 25 lbs | 1 | 68 mcg | 57 mg |
| 26 to 50 lbs | 1 | 136 mcg | 114 mg |
| 51 to 100 lbs | 1 | 272 mcg | 227 mg |
HEARTSHIELD PLUS Flavored Chewables are recommended for dogs 6 weeks of age and older. For dogs over 100 lbs use the appropriate combination of these flavored chewables.
ADMINISTRATION
Remove only one flavored chewable at a time from the foil-backed blister card. Because most dogs find HEARTSHIELD PLUS Flavored Chewables palatable, the product can be offered to the dog by hand. Alternatively, it may be added intact to a small amount of dog food or placed in the back of the dog's mouth for forced swallowing.
Care should be taken that the dog consumes the complete dose, and treated animals should be observed for a few minutes after administration to ensure that part of the dose is not lost or rejected. If it is suspected that any of the dose has been lost, redosing is recommended.
HEARTSHIELD PLUS Flavored Chewables should be given at monthly intervals during the period of the year when mosquitoes (vectors), potentially carrying infective heartworm larvae, are active. The initial dose must be given within a month (30 days) after the dog's first exposure to mosquitoes. The final dose must be given within a month (30 days) after the dog's last exposure to mosquitoes.
When replacing another heartworm preventive product in a heartworm disease preventive program, the first dose of HEARTSHIELD PLUS Flavored Chewables must be given within a month (30 days) of the last dose of the former medication.
If the interval between doses exceeds a month (30 days), the efficacy of ivermectin can be reduced. Therefore, for optimal performance, the flavored chewable must be given once a month on or about the same day of the month. If treatment is delayed, whether by a few days or many, immediate treatment with HEARTSHIELD PLUS Flavored Chewables and resumption of the recommended dosing regimen minimizes the opportunity for the development of adult heartworms.
Monthly treatment with HEARTSHIELD PLUS Flavored Chewables also provides effective treatment and control of ascarids (T. canis, T. leonina) and hookworms (A. caninum, U. stenocephala, A. braziliense). Clients should be advised of measures to be taken to prevent reinfection with intestinal parasites.
EFFICACY
HEARTSHIELD PLUS Flavored Chewables, given orally using the recommended dose and regimen, are effective against the tissue larval stage of D. immitis for a month (30 days) after infection and, as a result, prevent the development of the adult stage. HEARTSHIELD PLUS Flavored Chewables are also effective against canine ascarids (T. canis, T. leonina) and hookworms (A. caninum, U. stenocephala, A. braziliense).
ACCEPTABILITY
In a trial in client–owned dogs, HEARTSHIELD PLUS Flavored Chewables were shown to be a palatable oral dosage form consumed at first offering by the majority of dogs.
PRECAUTIONS
All dogs should be tested for existing heartworm infection before starting treatment with HEARTSHIELD PLUS Flavored Chewables, which are not effective against adult D. immitis. Infected dogs must be treated to remove adult heartworms and microfilariae before initiating a program with HEARTSHIELD PLUS Flavored Chewables.
While some microfilariae may be killed by the ivermectin in HEARTSHIELD PLUS Flavored Chewables at the recommended dose level, HEARTSHIELD PLUS Flavored Chewables are not effective for microfilariae clearance. A mild hypersensitivity-type reaction, presumably due to dead or dying microfilariae and particularly involving a transient diarrhea, has been observed in clinical trials with ivermectin alone after treatment of some dogs that have circulating microfilariae.
Keep this and all drugs out of the reach of children. In case of ingestion by humans, clients should be advised to contact a physician immediately. Physicians may contact a Poison Control Center for advice concerning cases of ingestion by humans.
Store at 20°C – 25°C (68°F – 77°F), excursions permitted between 15°C – 30°C (59°F – 86°F).
Protect product from light.
ADVERSE REACTIONS
In clinical trials with ivermectin/pyrantel, vomiting or diarrhea within 24 hours of dosing was rarely observed (1.1% of administered doses). The following adverse reactions have been reported following the use of ivermectin: Depression/lethargy, vomiting, anorexia, diarrhea, mydriasis, ataxia, staggering, convulsions and hypersalivation.
SAFETY
Studies with ivermectin indicate that certain dogs of the Collie breed are more sensitive to the effects of ivermectin administered at elevated dose levels (more than 16 times the target use level of 6 mcg/kg) than dogs of other breeds. At elevated doses, sensitive dogs showed adverse reactions which included mydriasis, depression, ataxia, tremors, drooling, paresis, recumbency, excitability, stupor, coma and death. Ivermectin demonstrated no signs of toxicity at 10 times the recommended dose (60 mcg/kg) in sensitive Collies. Results of these trials and bioequivalency studies, support the safety of ivermectin products in dogs, including Collies, when used as recommended.
Ivermectin/pyrantel has shown a wide margin of safety at the recommended dose level in dogs, including pregnant or breeding bitches, stud dogs and puppies aged 6 or more weeks. In clinical trials, many commonly used flea collars, dips, shampoos, anthelmintics, antibiotics, vaccines and steroid preparations have been administered with ivermectin/pyrantel in a heartworm disease preventive program.
In one trial, where some pups had parvovirus, there was a marginal reduction in efficacy against intestinal nematodes, possibly due to a change in intestinal transit time.
HOW SUPPLIED
HEARTSHIELD PLUS Flavored Chewables are available in three dosage strengths (See DOSAGE section) for dogs of different weights. Each strength comes in a box of 6 tablets, packed 10 boxes per display box.
ANADA 200-302, Approved by FDA.
Manufactured for: TruRx, LLC - Eagle, Idaho 83616 USA
For technical assistance or to report adverse drug reactions,
please call 1-855-631-7928.
49994-HS
06/12
302096-01
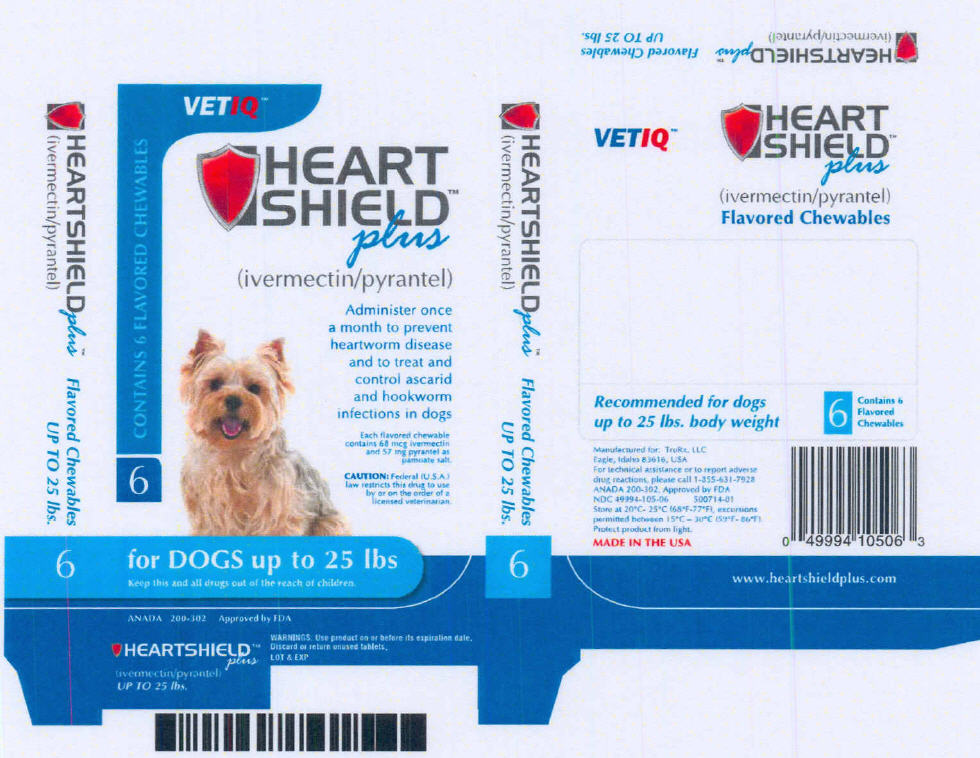
PRINCIPAL DISPLAY PANEL - up to 25 lbs Carton
VETIQ™
CONTAINS 6 FLAVORED CHEWABLES
6
HEART
SHIELD™
plus
(ivermectin/pyrantel)
Administer once
a month to prevent
heartworm disease
and to treat and
control ascarid
and hookworm
infections in dogs
Each flavored chewable
contains 68 mcg ivermectin
and 57 mg pyrantel as
pamoate salt.
CAUTION: Federal (U.S.A.)
law restricts this drug to use
by or on the order of a
licensed veterinarian.
for DOGS up to 25 lbs
Keep this and all drugs out of the reach of children.
ANADA 200-302 Approved by FDA
PRINCIPAL DISPLAY PANEL - 26 - 50 lbs Carton
VETIQ™
CONTAINS 6 FLAVORED CHEWABLES
6
HEART
SHIELD™
plus
(ivermectin/pyrantel)
Administer once
a month to prevent
heartworm disease
and to treat and
control ascarid
and hookworm
infections in dogs
Each flavored chewable
contains 136 mcg ivermectin
and 114 mg pyrantel as
pamoate salt.
CAUTION: Federal (U.S.A.)
law restricts this drug to use
by or on the order of a
licensed veterinarian.
for DOGS 26 - 50 lbs
Keep this and all drugs out of the reach of children.
ANADA 200-302 Approved by FDA

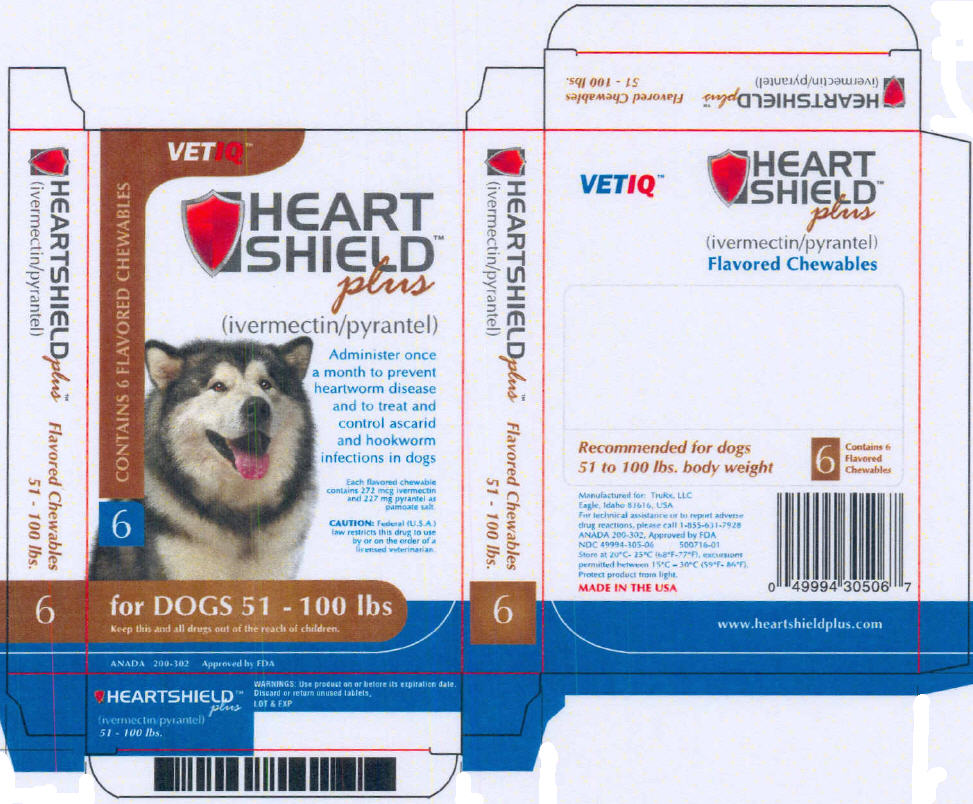
PRINCIPAL DISPLAY PANEL - 51 - 100 lbs Carton
VETIQ™
CONTAINS 6 FLAVORED CHEWABLES
6
HEART
SHIELD™
plus
(ivermectin/pyrantel)
Administer once
a month to prevent
heartworm disease
and to treat and
control ascarid
and hookworm
infections in dogs
Each flavored chewable
contains 272 mcg ivermectin
and 227 mg pyrantel as
pamoate salt.
CAUTION: Federal (U.S.A.)
law restricts this drug to use
by or on the order of a
licensed veterinarian.
for DOGS 51 - 100 lbs
Keep this and all drugs out of the reach of children.
ANADA 200-302 Approved by FDA
| HEARTSHIELD PLUS
ivermectin and pyrantel pamoate tablet, chewable |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| HEARTSHIELD PLUS
ivermectin and pyrantel pamoate tablet, chewable |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| HEARTSHIELD PLUS
ivermectin and pyrantel pamoate tablet, chewable |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - TruRX LLC (078454338) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Virbac Corporation | 829166276 | manufacture | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| COSMA Spa | 428655732 | api manufacture | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Shandong Qilu King-phar Pharmaceutical Co., Ltd. | 421524323 | api manufacture | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hangzhou Minsheng Binjiang Pharmaceutical Co.,Ltd | 547688780 | api manufacture | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Zhejiang Hisun Pharmaceutical Co., Ltd. | 654211754 | api manufacture | |