TERAZOSIN HYDROCHLORIDE- terazosin hydrochloride capsule
REMEDYREPACK INC.
----------
DESCRIPTION
Terazosin hydrochloride, an alpha-1-selective adrenoceptor blocking agent, is a quinazoline derivative represented by the following chemical name and structural formula: (RS)-Piperazine, 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-[(tetrahydro-2-furanyl)carbonyl]-, monohydrochloride. It has the following structural formula:
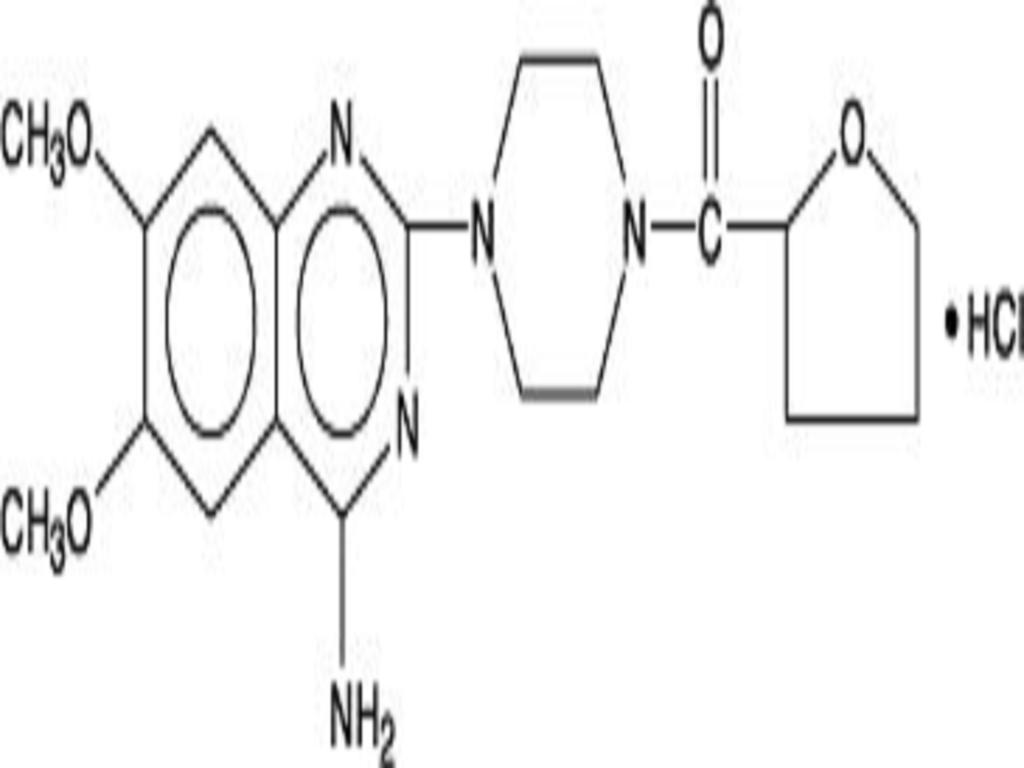
Terazosin hydrochloride is a white, crystalline substance, freely soluble in water and isotonic saline and has a molecular weight of 459.93. Terazosin hydrochloride capsules, for oral administration, are supplied in four dosage strengths containing terazosin hydrochloride equivalent to 1 mg, 2 mg, 5 mg, or 10 mg of terazosin. Inactive ingredients: Crospovidone, lactose (monohydrate), magnesium stearate, and microcrystalline cellulose. May contain corn starch. The capsule shells and imprinting inks contain: D & C Yellow #10 Aluminum Lake, FD & C Blue #1 Aluminum Lake, FD & C Blue #2 Aluminum Lake, FD & C Red #40 Aluminum Lake, gelatin, pharmaceutical glaze, propylene glycol, synthetic black iron oxide, and titanium dioxide. The 5 mg also contains: D & C Red #28.
PHARMACODYNAMICS
A. Benign Prostatic Hyperplasia (BPH)
The symptoms associated with BPH are related to bladder outlet obstruction, which is comprised of two underlying components: a static component and a dynamic component. The static component is a consequence of an increase in prostate size. Over time, the prostate will continue to enlarge. However, clinical studies have demonstrated that the size of the prostate does not correlate with the severity of BPH symptoms or the degree of urinary obstruction. The dynamic component is a function of an increase in smooth muscle tone in the prostate and bladder neck, leading to constriction of the bladder outlet. Smooth muscle tone is mediated by sympathetic nervous stimulation of alpha-1 adrenoceptors, which are abundant in the prostate, prostatic capsule and bladder neck. The reduction in symptoms and improvement in urine flow rates following administration of terazosin is related to relaxation of smooth muscle produced by blockade of alpha-1 adrenoceptors in the bladder neck and prostate. Because there are relatively few alpha-1 adrenoceptors in the bladder body, terazosin is able to reduce the bladder outlet obstruction without affecting bladder contractility.
Terazosin has been studied in 1222 men with symptomatic BPH. In three placebo-controlled studies, symptom evaluation and uroflowmetric measurements were performed approximately 24 hours following dosing. Symptoms were quantified using the Boyarsky Index. The questionnaire evaluated both obstructive (hesitancy, intermittency, terminal dribbling, impairment of size and force of stream, sensation of incomplete bladder emptying) and irritative (nocturia, daytime frequency, urgency, dysuria) symptoms by rating each of the 9 symptoms from 0 to 3, for a total score of 27 points. Results from these studies indicated that terazosin statistically significantly improved symptoms and peak urine flow rates over placebo as follows:
Symptom ScorePeak Flow Rate(Range 0-27)(mL/sec)NMeanMeanNMeanMeanBase-ChangeBase-Changeline%line%Study 1 (10 mg)
*
559.7-2. (24)5410.1+1.0 (10)Titration to fixed dose5410.1-4.5(45)
528.8+3.0 (34)
(12 weeks)PlaceboTerazosinStudy 2 (2, 5, 10, 208912.5-3.8(30)888.8+1.4 (16)mg)
8512.2-5.3(43)
848.4+2.9 (35)
Titration to response(24 weeks)PlaceboTerazosinStudy 3 (1, 2, 5, 107410.4-1.1(11) 748.8+1.2 (14)mg)
7310.9-4.6(42)
738.6+2.6 (30)
Titration to response(24 weeks)PlaceboTerazosin
*
Highest dose 10 mg shown.
Significantly (p0.05) more improvement than placebo.
23% of patients on 10 mg, 41% of patients on 20 mg.
67% of patients on 10 mg.
In all three studies, both symptom scores and peak urine flow rates showed statistically significant improvement from baseline in patients treated with terazosin capsules from week 2 (or the first clinic visit) and throughout the study duration.
Analysis of the effect of terazosin capsules on individual urinary symptoms demonstrated that compared to placebo, terazosin capsules significantly improved the symptoms of hesitancy, intermittency, impairment in size and force of urinary stream, sensation of incomplete emptying, terminal dribbling, daytime frequency and nocturia.
Global assessments of overall urinary function and symptoms were also performed by investigators who were blinded to patient treatment assignment. In studies 1 and 3, patients treated with terazosin capsules had a significantly (p0.001) greater overall improvement compared to placebo treated patients.
In a short term study (Study 1), patients were randomized to either 2, 5 or 10 mg of terazosin capsules or placebo. Patients randomized to the 10 mg group achieved a statistically significant response in both symptoms and peak flow rate compared to placebo (Figure 1).
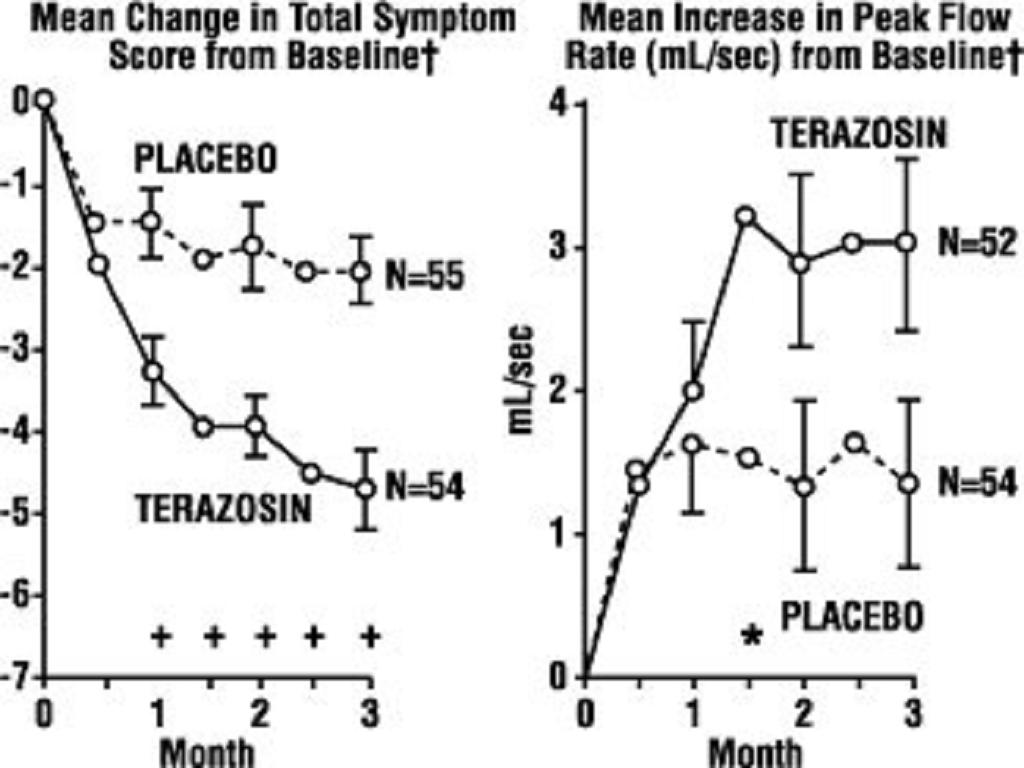
FIGURE 1, STUDY 1
for baseline values see above table
* p0.05, compared to placebo group
In a long-term, open-label, non-placebo controlled clinical trial, 181 men were followed for 2 years and 58 of these men were followed for 30 months. The effect of terazosin capsules on urinary symptom scores and peak flow rates was maintained throughout the study duration (Figures 2 and 3):
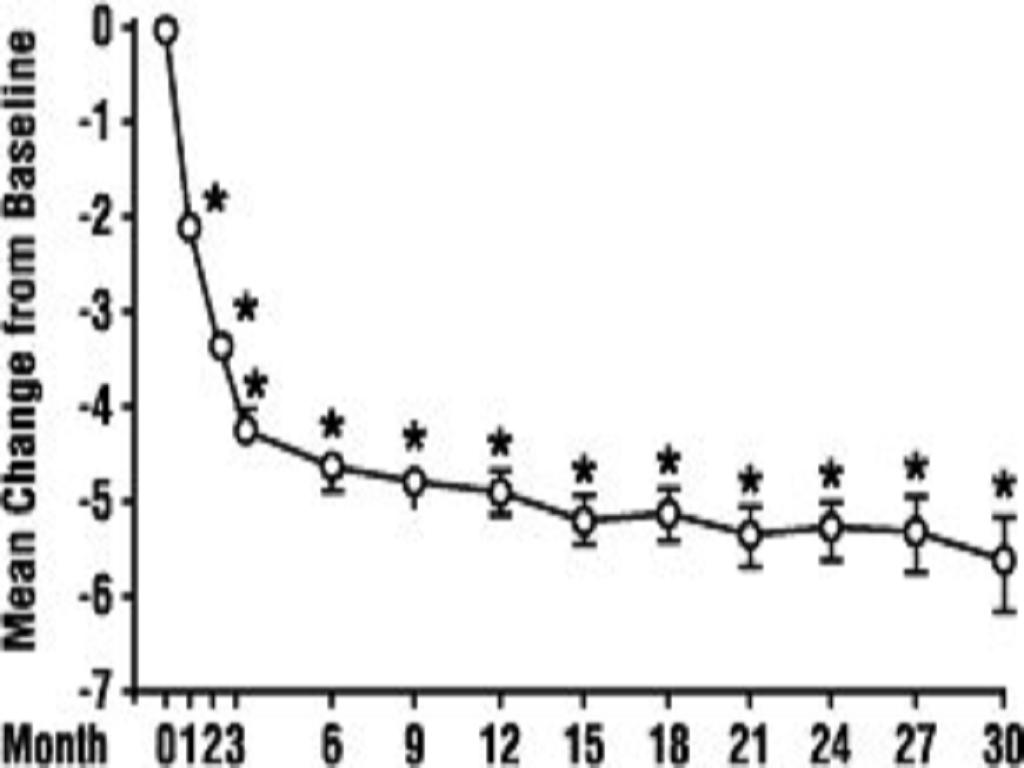
FIGURE 2. Mean Change in Total Symptom Score from Baseline Long-Term, Open-Label, Non-Placebo Controlled Study (N=494)
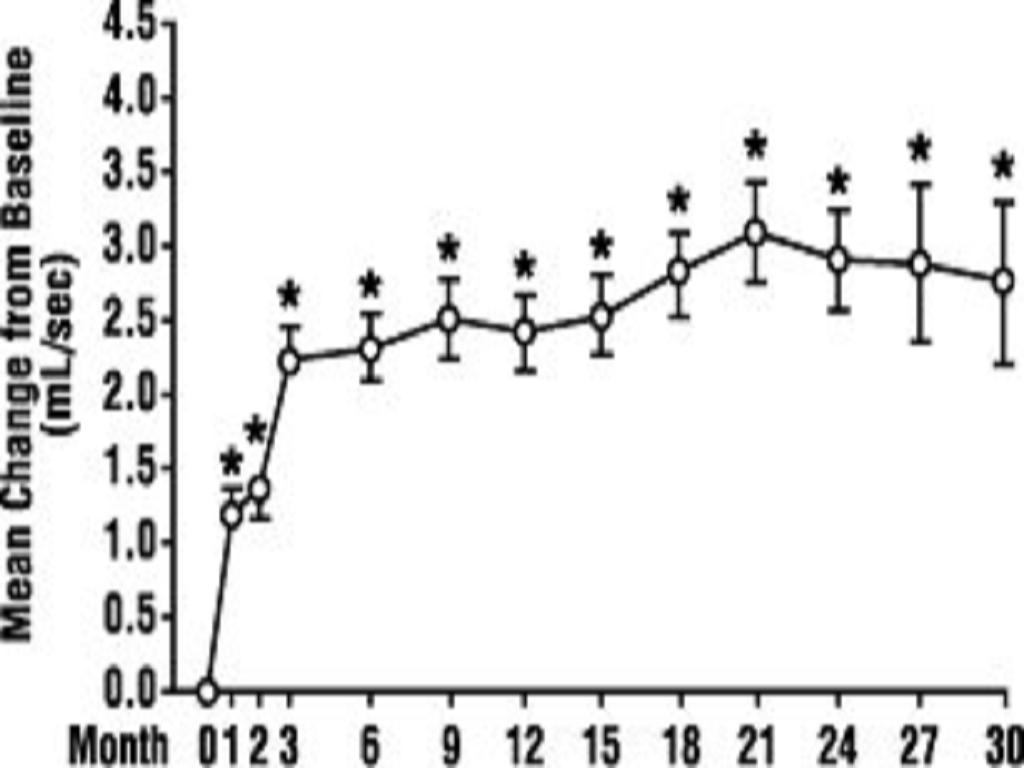
FIGURE 3. Mean Change in Peak Flow Rate from Baseline Long-Term, Open-Label, Non-Placebo Controlled Study (N=494)
In this long-term trial, both symptom scores and peak urinary flow rates showed statistically significant improvement suggesting a relaxation of smooth muscle cells.
Although blockade of alpha-1 adrenoceptors also lowers blood pressure in hypertensive patients with increased peripheral vascular resistance, terazosin treatment of normotensive men with BPH did not result in a clinically significant blood pressure lowering effect:
Mean Changes in Blood Pressure from Baseline to Final Visit in all Double-Blind, Placebo-Controlled Studies
GroupNormotensive Patients Hypertesive PatientsDBP
<
90 mm HgDBP > 90 mm HgMeanMeanNChangeNChangeSBPPlacebo293 519-0.145-5.8(mm Hg)Terazosin519-3.3
*
65-14.4
*
DBPPlacebo293+0.445-7.1(mm Hg)Terazosin519-2.2
*
65-15.1
*
*
p0.05 vs. placebo
B. Hypertension
In animals, terazosin causes a decrease in blood pressure by decreasing total peripheral vascular resistance. The vasodilatory hypotensive action of terazosin appears to be produced mainly by blockade of alpha-1 adrenoceptors. Terazosin decreases blood pressure gradually within 15 minutes following oral administration.
Patients in clinical trials of terazosin were administered once daily (the great majority) and twice daily regimens with total doses usually in the range of 5 to 20 mg/day, and had mild (about 77%, diastolic pressure 95 to 105 mmHg) or moderate (23%, diastolic pressure 105 to 115 mmHg) hypertension. Because terazosin, like all alpha antagonists, can cause unusually large falls in blood pressure after the first dose or first few doses, the initial dose was 1 mg in virtually all trials, with subsequent titration to a specified fixed dose or titration to some specified blood pressure end point (usually a supine diastolic pressure of 90 mmHg).
Blood pressure responses were measured at the end of the dosing interval (usually 24 hours) and effects were shown to persist throughout the interval, with the usual supine responses 5 to 10 mmHg systolic and 3.5 to 8 mmHg diastolic greater than placebo. The responses in the standing position tended to be somewhat larger, by 1 to 3 mmHg, although this was not true in all studies. The magnitude of the blood pressure responses was similar to prazosin and less than hydrochlorothiazide (in a single study of hypertensive patients). In measurements 24 hours after dosing, heart rate was unchanged.
Limited measurements of peak response (2 to 3 hours after dosing) during chronic terazosin administration indicate that it is greater than about twice the trough (24 hour) response, suggesting some attenuation of response at 24 hours, presumably due to a fall in blood terazosin concentrations at the end of the dose interval. This explanation is not established with certainty, however, and is not consistent with the similarity of blood pressure response to once daily and twice daily dosing and with the absence of an observed dose-response relationship over a range of 5 to 20 mg, i.e., if blood concentrations had fallen to the point of providing less than full effect at 24 hours, a shorter dosing interval or larger dose should have led to increased response.
Further dose response and dose duration studies are being carried out. Blood pressure should be measured at the end of the dose interval; if response is not satisfactory, patients may be tried on a larger dose or twice daily dosing regimen. The latter should also be considered if possibly blood pressure-related side effects, such as dizziness, palpitations, or orthostatic complaints, are seen within a few hours after dosing.
The greater blood pressure effect associated with peak plasma concentrations (first few hours after dosing) appears somewhat more position-dependent (greater in the erect position) than the effect of terazosin at 24 hours and in the erect position there is also a 6 to 10 beat per minute increase in heart rate in the first few hours after dosing. During the first 3 hours after dosing 12.5% of patients had a systolic pressure fall of 30 mmHg or more from supine to standing, or standing systolic pressure below 90 mmHg with a fall of at least 20 mmHg, compared to 4% of a placebo group.
There was a tendency for patients to gain weight during terazosin therapy. In placebo-controlled monotherapy trials, male and female patients receiving terazosin gained a mean of 1.7 and 2.2 pounds respectively, compared to losses of 0.2 and 1.2 pounds respectively in the placebo group. Both differences were statistically significant.
During controlled clinical trials, patients receiving terazosin monotherapy had a small but statistically significant decrease (a 3% fall) compared to placebo in total cholesterol and the combined low-density and very-low-density lipoprotein fractions. No significant changes were observed in high-density lipoprotein fraction and triglycerides compared to placebo.
Analysis of clinical laboratory data following administration of terazosin suggested the possibility of hemodilution based on decreases in hematocrit, hemoglobin, white blood cells, total protein and albumin. Decreases in hematocrit and total protein have been observed with alpha-blockade and are attributed to hemodilution.
INDICATIONS & USAGE
Terazosin capsules are indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH). There is a rapid response, with approximately 70% of patients experiencing an increase in urinary flow and improvement in symptoms of BPH when treated with terazosin capsules. The long-term effects of terazosin capsules on the incidence of surgery, acute urinary obstruction or other complications of BPH are yet to be determined.
Terazosin capsules are also indicated for the treatment of hypertension. Terazosin capsules can be used alone or in combination with other antihypertensive agents such as diuretics or beta-adrenergic blocking agents.
CONTRAINDICATIONS
Terazosin capsules are contraindicated in patients known to be hypersensitive to terazosin hydrochloride.
WARNINGS
Syncope andFirst-doseEffect
Terazosin capsules, like other alpha-adrenergic blocking agents, can cause marked lowering of blood pressure, especially postural hypotension, and syncope in association with the first dose or first few days of therapy. A similar effect can be anticipated if therapy is interrupted for several days and then restarted. Syncope has also been reported with other alpha-adrenergic blocking agents in association with rapid dosage increases or the introduction of another antihypertensive drug. Syncope is believed to be due to an excessive postural hypotensive effect, although occasionally the syncopal episode has been preceded by a bout of severe supraventricular tachycardia with heart rates of 120 to 160 beats per minute. Additionally, the possibility of the contribution of hemodilution to the symptoms of postural hypotension should be considered.
To decrease the likelihood of syncope or excessive hypotension, treatment should always be initiated with a 1 mg dose of terazosin capsules, given at bedtime. The 2 mg, 5 mg and 10 mg capsules are not indicated as initial therapy. Dosage should then be increased slowly, according to recommendations in the Dosage and Administration section and additional antihypertensive agents should be added with caution. The patient should be cautioned to avoid situations, such as driving or hazardous tasks, where injury could result should syncope occur during initiation of therapy.
In early investigational studies, where increasing single doses up to 7.5 mg were given at 3 day intervals, tolerance to the first dose phenomenon did not necessarily develop and thefirst-dose
In three placebo-controlled BPH studies 1, 2, and 3 (see
CLINICAL PHARMACOLOGY
), the incidence of postural hypotension in the terazosin treated patients was 5.1%, 5.2%, and 3.7% respectively.
In multiple dose clinical trials involving nearly 2000 hypertensive patients treated with terazosin capsules, syncope was reported in about 1% of patients. Syncope was not necessarily associated only with the first dose.
If syncope occurs, the patient should be placed in a recumbent position and treated supportively as necessary. There is evidence that the orthostatic effect of terazosin capsules is greater, even in chronic use, shortly after dosing. The risk of the events is greatest during the initial seven days of treatment, but continues at all time intervals.
Priapism
Rarely, (probably less than once in every several thousand patients), terazosin and otherhave been associated with priapism (painful penile erection, sustained for hours and unrelieved by sexual intercourse or masturbation). Two or three dozen cases have been reported. Because this condition can lead to permanent impotence if not promptly treated, patients must be advised about the seriousness of the condition (see
PRECAUTIONS: Information for Patients
).
PRECAUTIONS
General
Prostatic Cancer
Carcinoma of the prostate and BPH cause many of the same symptoms. These two diseases frequently co-exist. Therefore, patients thought to have BPH should be examined prior to starting terazosin capsule therapy to rule out the presence of carcinoma of the prostate.
Intraoperative Floppy Iris Syndrome (IFIS)
Intraoperative Floppy Iris Syndrome (IFIS) has been observed during cataract surgery in some patients on/or previously treated with alpha-1 blockers. This variant of small pupil syndrome is characterized by the combination of a flaccid iris that billows in response to intraoperative irrigation currents, progressive intraoperative miosis despite preoperative dilation with standard mydriatic drugs, and potential prolapse of the iris toward the phacoemulsification incisions. The patient's ophthalmologist should be prepared for possible modifications to their surgical technique, such as the utilization of iris hooks, iris dilator rings, or viscoelastic substances. There does not appear to be a benefit of stopping alpha-1 blocker therapy prior to cataract surgery.
Orthostatic Hypotension
While syncope is the most severe orthostatic effect of terazosin capsules (see
WARNINGS
), other symptoms of lowered blood pressure, such as dizziness, lightheadedness and palpitations, were more common and occurred in some 28% of patients in clinical trials of hypertension. In BPH clinical trials, 21% of the patients experienced one or more of the following: dizziness, hypotension, postural hypotension, syncope, and vertigo. Patients with occupations in which such events represent potential problems should be treated with particular caution.
INFORMATION FOR PATIENTS
(See
PATIENT INFORMATION
.)
Patients should be made aware of the possibility of syncopal and orthostatic symptoms, especially at the initiation of therapy, and to avoid driving or hazardous tasks for 12 hours after the first dose, after a dosage increase, and after interruption of therapy when treatment is resumed. They should be cautioned to avoid situations where injury could result should syncope occur during initiation of terazosin capsule therapy. They should also be advised of the need to sit or lie down when symptoms of lowered blood pressure occur, although these symptoms are not always orthostatic, and to be careful when rising from a sitting or lying position. If dizziness, lightheadedness, or palpitations are bothersome they should be reported to the physician, so that dose adjustment can be considered.
Patients should also be told that drowsiness or somnolence can occur with terazosin capsules, requiring caution in people who must drive or operate heavy machinery.
Patients should be advised about the possibility of priapism as a result of treatment with terazosin capsules and other similar medications. Patients should know that this reaction to terazosin capsules is extremely rare, but that if it is not brought to immediate medical attention, it can lead to permanent erectile dysfunction (impotence).
LABORATORY TESTS
Small but statistically significant decreases in hematocrit, hemoglobin, white blood cells, total protein and albumin were observed in controlled clinical trials. These laboratory findings suggested the possibility of hemodilution. Treatment with terazosin capsules for up to 24 months had no significant effect on prostate specific antigen (PSA) levels.
DRUG INTERACTIONS
2. antibiotics (e.g., erythromycin, trimethoprim and sulfamethoxazole);
3. anticholinergic/sympathomimetics (e.g., phenylephrine hydrochloride, phenylpropanolamine hydrochloride, pseudoephedrine hydrochloride);
4. antigout (e.g., allopurinol);
5. antihistamines (e.g., chlorpheniramine);
6. cardiovascular agents (e.g., atenolol, hydrochlorothiazide, methyclothiazide, propranolol);
7. corticosteroids;
8. gastrointestinal agents (e.g., antacids);
9. hypoglycemics
10. sedatives and tranquilizers (e.g., diazepam).
Use With Other Drugs
In a study (n=24) where terazosin and verapamil were administered concomitantly, terazosin's mean AUC0-24 increased 11% after the first verapamil dose and after 3 weeks of verapamil treatment it increased by 24% with associated increases in Cmax (25%) and Cmin (32%) means. Terazosin mean Tmax decreased from 1.3 hours to 0.8 hours after 3 weeks of verapamil treatment. Statistically significant differences were not found in the verapamil level with and without terazosin. In a study (n=6) where terazosin and captopril were administered concomitantly, plasma disposition of captopril was not influenced by concomitant administration of terazosin and terazosin maximum plasma concentrations increased linearly with dose at steady-state after administration of terazosin plus captopril (see
DOSAGE AND ADMINISTRATION
).
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
Terazosin capsules were devoid of mutagenic potential when evaluated in vivo and in vitro (the Ames test, in vivo cytogenetics, the dominant lethal test in mice, in vivo Chinese hamster chromosome aberration test and V79 forward mutation assay).
Terazosin capsules, administered in the feed to rats at doses of 8, 40, and 250 mg/kg/day (70, 350, and 2100 mg/M2/day), for two years, was associated with a statistically significant increase in benign adrenal medullary tumors of male rats exposed to the 250 mg/kg dose. This dose is 175 times the maximum recommended human dose of 20 mg (12 mg/M2). Female rats were unaffected. Terazosin capsules were not oncogenic in mice when administered in feed for 2 years at a maximum tolerated dose of 32 mg/kg/day (110 mg/M2; 9 times the maximum recommended human dose). The absence of mutagenicity in a battery of tests, of tumorigenicity of any cell type in the mouse carcinogenicity assay, of increased total tumor incidence in either species, and of proliferative adrenal lesions in female rats, suggests a male rat species-specific event. Numerous other diverse pharmaceutical and chemical compounds have also been associated with benign adrenal medullary tumors in male rats without supporting evidence for carcinogenicity in man.
The effect of terazosin capsules on fertility was assessed in a standard fertility/reproductive performance study in which male and female rats were administered oral doses of 8, 30 and 120 mg/kg/day. Four of 20 male rats given 30 mg/kg (240 mg/M2; 20 times the maximum recommended human dose) and five of 19 male rats given 120 mg/kg (960 mg/M2; 80 times the maximum recommended human dose) failed to sire a litter. Testicular weights and morphology were unaffected by treatment. Vaginal smears at 30 and 120 mg/kg/day, however, appeared to contain less sperm than smears from control matings and good correlation was reported between sperm count and subsequent pregnancy.
Oral administration of terazosin capsules for one or two years elicited a statistically significant increase in the incidence of testicular atrophy in rats exposed to 40 and 250 mg/kg/day (29 and 175 times the maximum recommended human dose), but not in rats exposed to 8 mg/kg/day (> 6 times the maximum recommended human dose). Testicular atrophy was also observed in dogs dosed with 300 mg/kg/day (> 500 times the maximum recommended human dose) for three months but not after one year when dosed with 20 mg/kg/day (38 times the maximum recommended human dose). This lesion has also been seen with prazosin hydrochloride, another (marketed) selective-alpha-1 blocking agent.
PREGNANCY
Teratogenic Effects
Pregnancy Category C
Terazosin capsules were not teratogenic in either rats or rabbits when administered at oral doses up to 280 and 60 times, respectively, the maximum recommended human dose. Fetal resorptions occurred in rats dosed with 480 mg/kg/day, approximately 280 times the maximum recommended human dose. Increased fetal resorptions, decreased fetal weight and an increased number of supernumerary ribs were observed in offspring of rabbits dosed with 60 times the maximum recommended human dose. These findings (in both species) were most likely secondary to maternal toxicity. There are no adequate and well-controlled studies in pregnant women and the safety of terazosin in pregnancy has not been established. Terazosin capsules are not recommended during pregnancy unless the potential benefit justifies the potential risk to the mother and fetus.
>
Nonteratogenic Effects
In a peri- and post-natal development study in rats, significantly more pups died in the group dosed with 120 mg/kg/day (> 75 times the maximum recommended human dose) than in the control group during the three-week postpartum period.
NURSING MOTHERS
It is not known whether terazosin is excreted in breast milk. Because many drugs are excreted in breast milk, caution should be exercised when terazosin capsules are administered to a nursing woman.
ADVERSE REACTIONS
Benign Prostatic Hyperplasia
The incidence of treatment-emergent adverse events has been ascertained from clinical trials conducted worldwide. All adverse events reported during these trials were recorded as adverse reactions. The incidence rates presented below are based on combined data from six placebo-controlled trials involving once-a-day administration of terazosin at doses ranging from 1 to 20 mg. Table 1 summarizes those adverse events reported for patients in these trials when the incidence rate in the terazosin group was at least 1%, and was greater than that for the placebo group, or where the reaction is of clinical interest. Asthenia, postural hypotension, dizziness, somnolence, nasal congestion/rhinitis, and impotence were the only events that were significantly (p0.05) more common in patients receiving terazosin than in patients receiving placebo. The incidence of urinary tract infection was significantly lower in the patients receiving terazosin than in patients receiving placebo. An analysis of the incidence rate of hypotensive adverse events (see
PRECAUTIONS
) adjusted for the length of drug treatment has shown that the risk of the events is greatest during the initial seven days of treatment, but continues at all time intervals.
TABLE 1. Adverse Reactions During Placebo-Controlled Trials Benign Prostatic Hyperplasia
Body SystemTerazosinPlacebo(N = 636)(N = 360)BODY AS A WHOLE
*
Asthenia7.4%
3.3%Flu Syndrome2.4%1.7%Headache4.9%5.8%
CARDIOVASCULAR SYSTEM
Hypotension0.6%0.6%Palpitations0.9%1.1%Postural Hypotension3.9%
0.8%Syncope0.6%0.0%
DIGESTIVE SYSTEM
Nausea1.7%1.1%
METABOLIC ANDNUTRITIONAL DISORDERS
Peripheral Edema0.9%0.3%Weight Gain0.5%0.0%
NERVOUS SYSTEM
Dizziness9.1%
4.2%Somnolence3.6%
1.9%Vertigo1.4%0.3%
RESPIRATORY SYSTEM
Dyspnea1.7%0.8%Nasal Congestion/Rhinitis1.9%
0.0%
SPECIAL SENSES
Blurred Vision/ Amblyopia1.3%0.6%
UROGENITAL SYSTEM
Impotence1.6%
0.6%Urinary Tract Infection1.3%3.9%
*
Includes weakness, tiredness, lassitude, and fatigue.
p0.05 comparison between groups.
Additional adverse events have been reported, but these are, in general, not distinguishable from symptoms that might have occurred in the absence of exposure to terazosin. The safety profile of patients treated in the long-term open-label study was similar to that observed in the controlled studies.
The adverse events were usually transient and mild or moderate in intensity, but sometimes were serious enough to interrupt treatment. In the placebo-controlled clinical trials, the rates of premature termination due to adverse events were not statistically different between the placebo and terazosin groups. The adverse events that were bothersome, as judged by their being reported as reasons for discontinuation of therapy by at least 0.5% of the terazosin group and being reported more often than in the placebo group, are shown in Table 2.
TABLE 2. Discontinuation During Placebo-Controlled Trials Benign Prostatic Hyperplasia
Body SystemTerazosinPlacebo(N = 636)(N = 360)BODY AS A WHOLE
Fever0.5%0.0%Headache1.1%0.8%
CARDIOVASCULAR SYSTEM
Postural Hypotension0.5%0.0%Syncope0.5%0.0%
DIGESTIVE SYSTEM
Nausea0.5%0.3%
NERVOUS SYSTEM
Dizziness2.0%1.1%Vertigo0.5%0.0%
RESPIRATORY SYSTEM
Dyspnea0.5%0.3%
SPECIAL SENSES
Blurred Vision/Amblyopia0.6%0.0%
UROGENITAL SYSTEM
Urinary Tract Infection0.5%0.3%
Hypertension
The prevalence of adverse reactions has been ascertained from clinical trials conducted primarily in the United States. All adverse experiences (events) reported during these trials were recorded as adverse reactions. The prevalence rates presented below are based on combined data from fourteen placebo-controlled trials involving once-a-day administration of terazosin, as monotherapy or in combination with other antihypertensive agents, at doses ranging from 1 to 40 mg. Table 3 summarizes those adverse experiences reported for patients in these trials where the prevalence rate in the terazosin group was at least 5%, where the prevalence rate for the terazosin group was at least 2% and was greater than the prevalence rate for the placebo group, or where the reaction is of particular interest. Asthenia, blurred vision, dizziness, nasal congestion, nausea, peripheral edema, palpitations and somnolence were the only symptoms that were significantly (p < 0.05) more common in patients receiving terazosin than in patients receiving placebo. Similar adverse reaction rates were observed in placebo-controlled monotherapy trials.
TABLE 3. Adverse Reactions During Placebo-Controlled Trials Hypertension
Body SystemTerazosinPlacebo(N = 859)(N = 506)BODY AS A WHOLE *
Asthenia11.3%
4.3%Back Pain2.4%1.2%Headache16.2%15.8%
CARDIOVASCULAR SYSTEM
Palpitations4.3%
1.2%Postural Hypotension1.3%0.4%Tachycardia1.9%1.2%
DIGESTIVE SYSTEM
Nausea4.4%
1.4%
METABOLIC ANDNUTRITIONAL DISORDERS
Edema0.9%0.6%Peripheral Adema5.5%
2.4%Weight Gain0.5%0.2%
MUSCULOSKETAL SYSTEM
PainExtremities3.5%3.0%
NERVOUS SYSTEM
Depression0.3%0.2%Dizziness19.3%
7.5%Libido Decreased0.6%0.2%Nervousness2.3%1.8%Paresthesia2.9%1.4%Somnolence5.4%
2.6%
RESPIRATORY SYSTEM
Dyspnea3.1%2.4%Nasal Congestion5.9%
3.4%Sinusitis2.6%1.4%
SPECIAL SENSES
Blurred Vision1.6%
0.0%
UROGENITAL SYSTEM
Impotence1.2%1.4%
*
Includes weakness, tiredness, lassitude, and fatigue.
Statistically significant at p=0.05 level.
Additional adverse reactions have been reported, but these are, in general, not distinguishable from symptoms that might have occurred in the absence of exposure to terazosin. The following additional adverse reactions were reported by at least 1% of 1987 patients who received terazosin in controlled or open, short- or long-term clinical trials studies or have been reported during marketing experience:
Body as a Whole
Chest pain, facial edema, fever, abdominal pain, neck pain, shoulder pain.
Cardiovascular System
Arrhythmia, vasodilation.
Digestive System
Constipation, diarrhea, dry mouth, dyspepsia, flatulence, vomiting.
Metabolic/Nutritional Disorders
Gout.
Musculoskeletal System
Arthralgia, arthritis, joint disorder, myalgia.
Nervous System
Anxiety, insomnia.
Respiratory System
Bronchitis, cold symptoms, epistaxis, flu symptoms, increased cough, pharyngitis, rhinitis.
Skin and Appendages
Pruritus, rash, sweating.
Special Senses
Abnormal vision, conjunctivitis, tinnitus.
Urogenital System
Urinary frequency, urinary incontinence primarily reported in postmenopausal women, urinary tract infection.
The adverse reactions were usually mild or moderate in intensity but sometimes were serious enough to interrupt treatment. The adverse reactions that were most bothersome, as judged by their being reported as reasons for discontinuation of therapy by at least 0.5% of the terazosin group and being reported more often than in the placebo group, are shown in Table 4.
TABLE 4. Discontinuation During Placebo-Controlled Trials Hypertension
Body SystemTerazosinPlacebo(N = 859)(N = 506)BODY AS A WHOLE
Asthenia1.6%0.0%Headache1.3%1.0%
CARDIOVASCULAR SYSTEM
Palpitations1.4%0.2%Postural Hypotension0.5%0.0%Syncope0.5%0.2%Tachycardia0.6%0.0%
DIGESTIVE SYSTEM
Nausea0.8%0.0%
METABOLIC AND NUTRITIONALDISORDERS
Peripheral Edema0.6%0.0%
NERVOUS SYSTEM
Dizziness3.1%0.4%Paresthesia0.8%0.2%Sonmolence0.6%0.2%
RESPIRATORY SYSTEM
Dyspnea0.9%0.6%Nasal Congestion0.6%0.0%
SPECIAL SENSES
Blurred Vision0.6%0.0%
Post-marketing Experience
Post-marketing experience indicates that in rare instances patients may develop allergic reactions, including anaphylaxis, following administration of terazosin hydrochloride. There have been reports of priapism and thrombocytopenia during post-marketing surveillance. Atrial fibrillation has been reported.
During cataract surgery, a variant of small pupil syndrome known as Intraoperative Floppy Iris Syndrome (IFIS) has been reported in association with alpha-1 blocker therapy (see
PRECAUTIONS
).
DOSAGE & ADMINISTRATION
If terazosin capsules administration is discontinued for several days, therapy should be reinstituted using the initial dosing regimen.
Benign Prostatic Hyperplasia
Initial Dose
1 mg at bedtime is the starting dose for all patients, and this dose should not be exceeded as an initial dose. Patients should be closely followed during initial administration in order to minimize the risk of severe hypotensive response.
Subsequent Doses
The dose should be increased in a stepwise fashion to 2 mg, 5 mg, or 10 mg once daily to achieve the desired improvement of symptoms and/or flow rates. Doses of 10 mg once daily are generally required for the clinical response. Therefore, treatment with 10 mg for a minimum of 4 to 6 weeks may be required to assess whether a beneficial response has been achieved. Some patients may not achieve a clinical response despite appropriate titration. Although some additional patients responded at a 20 mg daily dose, there was an insufficient number of patients studied to draw definitive conclusions about this dose. There are insufficient data to support the use of higher doses for those patients who show inadequate or no response to 20 mg daily. If terazosin administration is discontinued for several days or longer, therapy should be reinstituted using the initial dosing regimen.
Use With Other Drugs
Caution should be observed when terazosin capsules is administered concomitantly with other antihypertensive agents, especially the calcium channel blocker verapamil, to avoid the possibility of developing significant hypotension. When using terazosin capsules and other antihypertensive agents concomitantly, dosage reduction and retitration of either agent may be necessary (see
PRECAUTIONS
). Hypotension has been reported when terazosin capsules have been used with phosphodiesterase-5 (PDE-5) inhibitors.
Hypertension
The dose of terazosin capsules and the dose interval (12 or 24 hours) should be adjusted according to the patient's individual blood pressure response. The following is a guide to its administration:
Initial Dose
1 mg at bedtime is the starting dose for all patients, and this dose should not be exceeded. This initial dosing regimen should be strictly observed to minimize the potential for severe hypotensive effects.
Subsequent Doses
Use With Other Drugs
(See above.)
STORAGE AND HANDLING
Store at 20(68(see USP Controlled Room Temperature). Protect from light and moisture.
Dispense in a tight, light-resistant container.
| TERAZOSIN HYDROCHLORIDE
terazosin hydrochloride capsule |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - REMEDYREPACK INC. (829572556) |