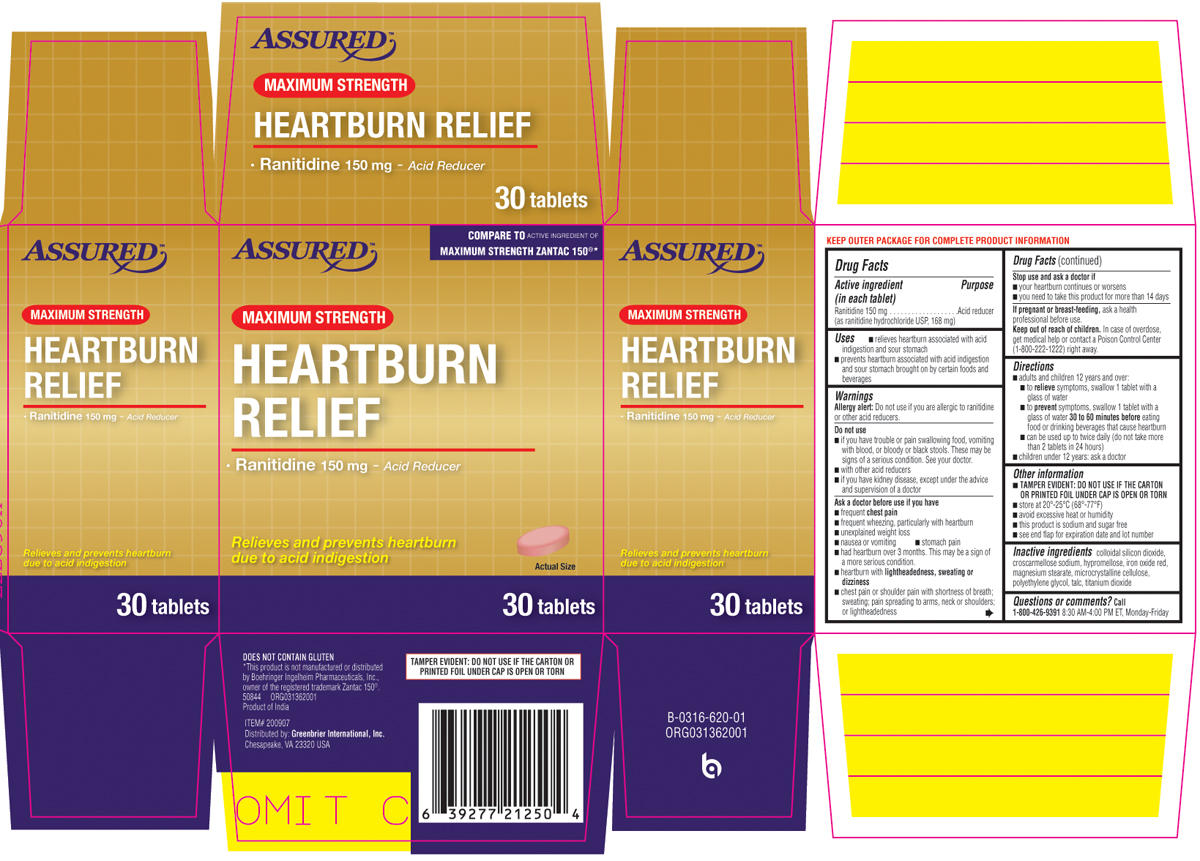
HEARTBURN RELIEF MAXIMUM STRENGTH- ranitidine tablet, film coated
GREENBRIER INTERNATIONAL, INC.
----------
Assured 44-620-Delisted
Uses
- relieves heartburn associated with acid indigestion and sour stomach
- prevents heartburn associated with acid indigestion and sour stomach brought on by certain foods and beverages
Warnings
Allergy alert: Do not use if you are allergic to ranitidine or any other acid reducers.
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor.
- with other acid reducers
- if you have kidney disease, except under the advice and supervision of a doctor
Ask a doctor before use if you have
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating, or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
Directions
- adults and children 12 years and over:
- to relieve symptoms, swallow 1 tablet with a glass of water
- to prevent symptoms, swallow 1 tablet with a glass of water 30 to 60 minutes before eating food or drinking beverages that cause heartburn
- can be used up to twice daily (do not take more than 2 tablets in 24 hours)
- children under 12 years: ask a doctor
Other information
-
TAMPER EVIDENT: DO NOT USE IF THE CARTON OR PRINTED FOIL UNDER CAP IS OPEN OR TORN
- store at 20º-25ºC (68º-77ºF)
- avoid excessive heat or humidity
- this product is sodium and sugar free
- see end flap for expiration date and lot number
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, hypromellose, iron oxide red, magnesium stearate, microcrystalline cellulose, polyethylene glycol, talc, titanium dioxide
Principal display panel
ASSURED™
COMPARE TO ACTIVE INGREDIENT OF MAXIMUM STRENGTH ZANTAC 150®*
MAXIMUM STRENGTH
HEARTBURN
RELIEF
• Ranitidine 150 mg - Acid Reducer
Relieves and prevents heartburn due to acid indigestion
30 tablets
TAMPER EVIDENT: DO NOT USE IF THE CARTON OR PRINTED FOIL UNDER CAP IS OPEN OR TORN
DOES NOT CONTAIN GLUTEN
*This product is not manufactured or distributed by Boehringer Ingelheim Pharmaceuticals, Inc., owner of the registered trademark Zantac 150®.
50844 ORG031362001
Product of India
ITEM #200907
Distributed by: Greenbrier International, Inc.
Chesapeake, VA 23320 USA
Assured 44-620
| HEARTBURN RELIEF
MAXIMUM STRENGTH
ranitidine tablet, film coated |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - GREENBRIER INTERNATIONAL, INC. (610322518) |