CHILDRENS IBUPROFEN- ibuprofen suspension
Fred's Inc.
----------
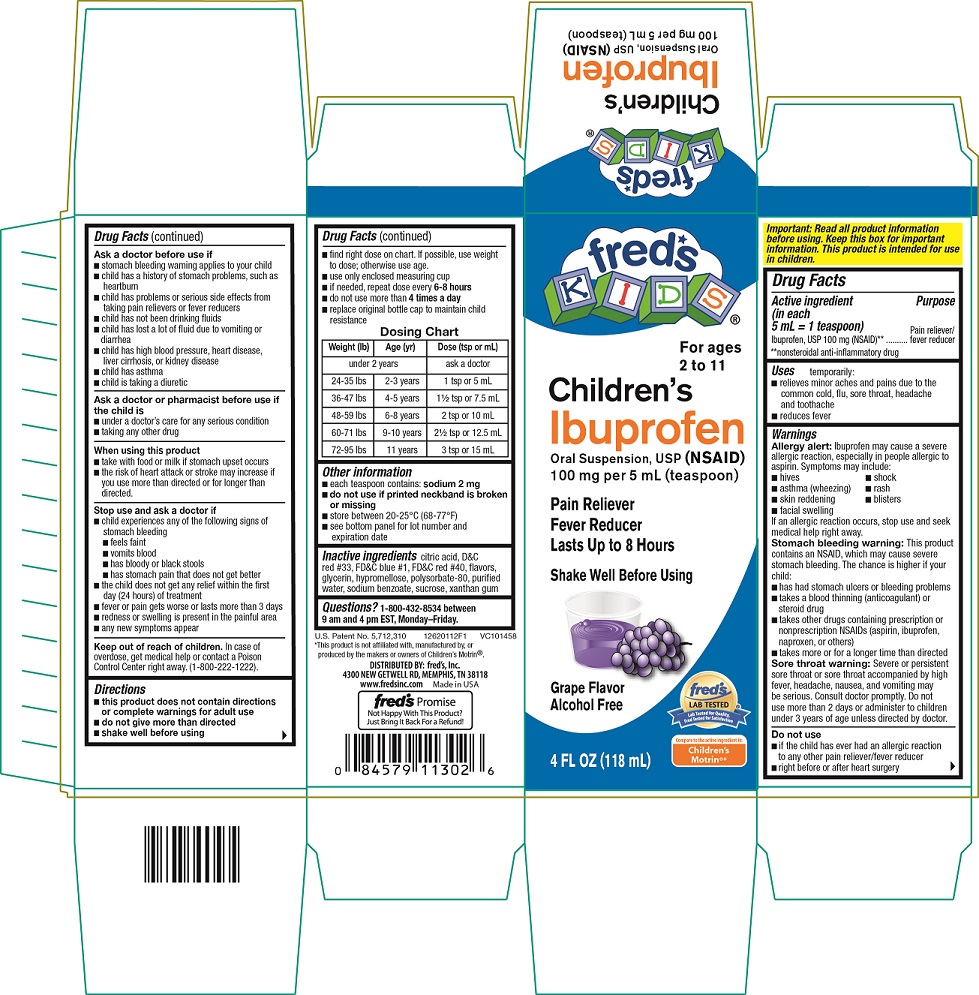
Ibuprofen Oral Suspension, USP
Active ingredient (in each 5 mL = 1 teaspoon)
Ibuprofen, USP 100 mg (NSAID)**
**nonsteroidal anti-inflammatory drug
Uses
temporarily:
- relieves minor aches and pains due to the common cold, flu, sore throat, headache and toothache
- reduces fever
Warnings
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin.
Symptoms may include:
- hives
- shock
- asthma (wheezing)
- rash
- skin reddening
- blisters
- facial swelling
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding.
The chance is higher if your child:
- has had stomach ulcers or bleeding problems
- takes a blood thinning (anticoagulant) or steroid drug
- takes other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- takes more or for a longer time than directed
Sore throat warning: Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious. Consult doctor promptly. Do not use more than 2 days or administer to children under 3 years of age unless directed by doctor.
Do not use
- if the child has ever had an allergic reaction to any other pain reliever/fever reducer
- right before or after heart surgery
Ask a doctor before use if
- stomach bleeding warning applies to your child
- child has a history of stomach problems, such as heartburn
- child has not been drinking fluids
- child has lost a lot of fluid due to vomiting or diarrhea
- child has high blood pressure, heart disease, liver cirrhosis, or kidney disease
- child is taking a diuretic
Ask a doctor or pharmacist before use if the child is
- under a doctor’s care for any serious condition
- taking any other drug
When using this product
- take with food or milk if stomach upset occurs
- the risk of heart attack or stroke may increase if you use more than directed or for longer than directed
Stop use and ask a doctor if
- child experiences any of the following signs of stomach bleeding
- feels faint
- vomits blood
- has bloody or black stools
- has stomach pain that does not get better
- the child does not get any relief within the first day (24 hours) of treatment
- fever or pain gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222).
Directions
- this product does not contain directions or complete warnings for adult use
- do not give more than directed
- shake well before using
- find right dose on chart. If possible, use weight to dose; otherwise use age.
- use only enclosed measuring cup
- if needed, repeat dose every 6-8 hours
- do not use more than 4 times a day
- replace original bottle cap to maintain child resistance
Dosing Chart
|
Weight (lb) |
Age (yr) |
Dose (tsp or mL) |
|
under 2 years |
ask a doctor |
|
|
24-35 lbs |
2-3 years |
1 tsp or 5 mL |
|
36-47 lbs |
4-5 years |
1 ½ tsp or 7.5 mL |
|
48-59 lbs |
6-8 years |
2 tsp or 10 mL |
|
60-71 lbs |
9-10 years |
2 ½ tsp or 12.5 mL |
|
72-95 lbs |
11 years |
3 tsp or 15 mL |
Other information
- each teaspoon contains: sodium 2 mg
- do not use if printed neckband is broken or missing
- store between 20 - 25°C (68 - 77°F)
- see bottom panel for lot number and expiration date
Inactive ingredients
Original Berry flavor: citric acid, D&C yellow #10, FD&C red #40, flavors, glycerin, hypromellose, polysorbate-80, purified water, sodium benzoate, sucrose, xanthan gum
Grape flavor: citric acid, D&C red #33, FD&C blue #1, FD&C red #40, flavors, glycerin, hypromellose, polysorbate -80, purified water, sodium benzoate, sucrose, xanthan gum
PRINCIPAL DISPLAY PANEL – ORIGINAL BERRY FLAVOR
fred’s KIDS®
For ages 2 to 11
Children’s
Ibuprofen
Oral Suspension, USP (NSAID)
100 mg per 5 mL (teaspoon)
Pain Reliever
Fever Reducer
Lasts Up To 8 Hours
Shake Well Before Using
Original Berry Flavor
Alcohol Free
4 fl oz (118 mL)
Compare to the active ingredient in:
Children’s Motrin®

PRINCIPAL DISPLAY PANEL – GRAPE FLAVOR
fred’s KIDS®
For ages 2 to 11
Children’s
Ibuprofen
Oral Suspension, USP (NSAID)
100 mg per 5 mL (teaspoon)
Pain Reliever
Fever Reducer
Lasts Up to 8 Hours
Shake Well Before Using
Grape Flavor
Alcohol Free
4 fl oz (118 mL)
Compare to the active ingredient in:
Children’s Motrin®
| CHILDRENS IBUPROFEN
ibuprofen suspension |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| CHILDRENS IBUPROFEN
ibuprofen suspension |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Fred's Inc. (005866116) |
| Registrant - Teva Pharmaceuticals USA, Inc. (001627975) |