Label: LUCENTIS- ranibizumab injection, solution
- NDC Code(s): 50242-080-03, 50242-080-88, 50242-082-03, 50242-082-88
- Packager: Genentech, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated February 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LUCENTIS safely and effectively. See full prescribing information for LUCENTIS.
LUCENTIS® (ranibizumab injection) for intravitreal injection
Initial U.S. Approval: 2006RECENT MAJOR CHANGES
INDICATIONS AND USAGE
LUCENTIS, a vascular endothelial growth factor (VEGF) inhibitor, is indicated for the treatment of patients with:
DOSAGE AND ADMINISTRATION
For ophthalmic intravitreal injection only (2.1)
-
Neovascular (Wet) Age-Related Macular Degeneration (AMD) (2.2):
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days).- -
- Although not as effective, patients may be treated with 3 monthly doses followed by less frequent dosing with regular assessment.
- -
- Although not as effective, patients may also be treated with one dose every 3 months after 4 monthly doses. Patients should be assessed regularly.
-
Macular Edema Following Retinal Vein Occlusion (RVO) (2.3):
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days). -
Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR) (2.4):
LUCENTIS 0.3 mg (0.05 mL of 6 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days). -
Myopic Choroidal Neovascularization (mCNV) (2.5):
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL solution) is recommended to be initially administered by intravitreal injection once a month (approximately 28 days) for up to three months. Patients may be retreated if needed.
DOSAGE FORMS AND STRENGTHS
WARNINGS AND PRECAUTIONS
- Endophthalmitis and retinal detachments may occur following intravitreal injections. Patients should be monitored following the injection (5.1).
- Increases in intraocular pressure (IOP) have been noted both pre- and post-intravitreal injection (5.2).
- There is a potential risk of arterial thromboembolic events following intravitreal use of VEGF inhibitors (5.3).
- Fatal events occurred more frequently in patients with DME and DR at baseline, who were treated monthly with LUCENTIS compared with control (5.4).
ADVERSE REACTIONS
- The most common adverse reactions (reported more frequently in LUCENTIS-treated subjects than control subjects) are conjunctival hemorrhage, eye pain, vitreous floaters, and increased IOP (6.2).
To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2024
-
Neovascular (Wet) Age-Related Macular Degeneration (AMD) (2.2):
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Neovascular (Wet) Age-Related Macular Degeneration (AMD)
1.2 Macular Edema Following Retinal Vein Occlusion (RVO)
1.3 Diabetic Macular Edema (DME)
1.4 Diabetic Retinopathy (DR)
1.5 Myopic Choroidal Neovascularization (mCNV)
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
2.2 Neovascular (Wet) Age-Related Macular Degeneration (AMD)
2.3 Macular Edema Following Retinal Vein Occlusion (RVO)
2.4 Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR)
2.5 Myopic Choroidal Neovascularization (mCNV)
2.6 Preparation for Administration
2.7 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Ocular or Periocular Infections
4.2 Hypersensitivity
5 WARNINGS AND PRECAUTIONS
5.1 Endophthalmitis and Retinal Detachments
5.2 Increases in Intraocular Pressure
5.3 Thromboembolic Events
5.4 Fatal Events in Patients with Diabetic Macular Edema and Diabetic Retinopathy at Baseline
5.5 Retinal Vasculitis with or without Occlusion
6 ADVERSE REACTIONS
6.1 Injection Procedure
6.2 Clinical Trials Experience
6.3 Immunogenicity
6.4 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Neovascular (Wet) Age-Related Macular Degeneration (AMD)
14.2 Macular Edema Following Retinal Vein Occlusion (RVO)
14.3 Diabetic Macular Edema (DME)
14.4 Diabetic Retinopathy (DR)
14.5 Myopic Choroidal Neovascularization (mCNV)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.2 Neovascular (Wet) Age-Related Macular Degeneration (AMD)
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days).
Although not as effective, patients may be treated with 3 monthly doses followed by less frequent dosing with regular assessment. In the 9 months after three initial monthly doses, less frequent dosing with 4-5 doses on average is expected to maintain visual acuity while monthly dosing may be expected to result in an additional average 1-2 letter gain. Patients should be assessed regularly [see Clinical Studies (14.1)].
Although not as effective, patients may also be treated with one dose every 3 months after 4 monthly doses. Compared with continued monthly dosing, dosing every 3 months over the next 9 months will lead to an approximate 5-letter (1-line) loss of visual acuity benefit, on average. Patients should be assessed regularly [see Clinical Studies (14.1)].
2.3 Macular Edema Following Retinal Vein Occlusion (RVO)
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days).
In Studies RVO-1 and RVO-2, patients received monthly injections of LUCENTIS for 6 months. In spite of being guided by optical coherence tomography and visual acuity re-treatment criteria, patients who were then not treated at Month 6 experienced on average, a loss of visual acuity at Month 7, whereas patients who were treated at Month 6 did not. Patients should be treated monthly [see Clinical Studies (14.2)].
2.4 Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR)
LUCENTIS 0.3 mg (0.05 mL of 6 mg/mL solution) is recommended to be administered by intravitreal injection once a month (approximately 28 days).
2.5 Myopic Choroidal Neovascularization (mCNV)
LUCENTIS 0.5 mg (0.05 mL of 10 mg/mL LUCENTIS solution) is recommended to be initially administered by intravitreal injection once a month (approximately 28 days) for up to 3 months. Patients may be retreated if needed [see Clinical Studies (14.5)].
2.6 Preparation for Administration
Prefilled Syringe:
The prefilled syringe is sterile and is for single-dose only. Do not use the product if the packaging is damaged or has been tampered with.
To prepare LUCENTIS for intravitreal administration, please adhere to these instructions for use. Read all the instructions carefully before using the prefilled syringe.
The opening of the sealed tray and all subsequent steps should be done under aseptic conditions.
For the intravitreal injection, a 30-gauge × ½ inch sterile injection needle should be used (not provided).
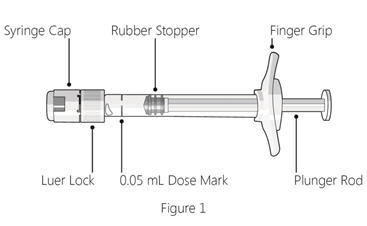
Note: the dose must be set to 0.05 mL.
Device description LUCENTIS prefilled syringes are available in 2 dose strengths: - LUCENTIS 0.5 mg prefilled syringe with a CLEAR finger grip.

- LUCENTIS 0.3 mg prefilled syringe with an ORANGE finger grip.


Check the labels on the LUCENTIS carton, syringe tray and prefilled syringe to make sure you have the correct dose strength.
Step 1: Prepare - Make sure that your pack contains a sterile prefilled syringe in a sealed tray.
- Peel the lid off the syringe tray and, using aseptic technique, remove the syringe.
Step 2: Inspect syringe - LUCENTIS should be colorless to pale yellow.
-
Do not use the prefilled syringe if:
- -
- the syringe cap is detached from the Luer lock.
- -
- the syringe is damaged.
- -
- particulates, cloudiness, or discoloration are visible.
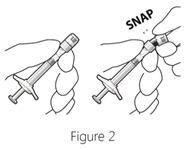
Step 3: Remove syringe cap - Snap off (do not turn or twist) the syringe cap (see Figure 2).
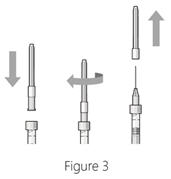
Step 4: Attach needle - Attach a 30G × ½ inch sterile injection needle firmly onto the syringe by screwing it tightly onto the Luer lock (see Figure 3).
- Carefully remove the needle cap by pulling it straight off.
Note: Do not wipe the needle at any time.
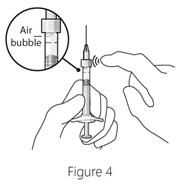
Step 5: Dislodge air bubbles - Hold the syringe with the needle pointing up.
- If there are any air bubbles, gently tap the syringe with your finger until the bubbles rise to the top (see Figure 4).
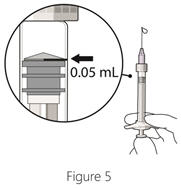
Step 6: Expel air and adjust drug dose - Hold the syringe at eye level, and carefully push the plunger rod until the edge below the dome of the rubber stopper is aligned with the 0.05 mL dose mark (see Figure 5).
Step 7: Inject - The injection procedure should be carried out under aseptic conditions.
- Insert the needle into the injection site.
- Inject slowly until rubber stopper reaches the bottom of the syringe to deliver the volume of 0.05 mL.
- After injection, do not recap the needle or detach it from the syringe. Dispose of the used syringe together with the needle in a sharps disposal container or in accordance with local requirements.
2.7 Administration
The intravitreal injection procedure should be carried out under controlled aseptic conditions, which include the use of sterile gloves, a sterile drape, and a sterile eyelid speculum (or equivalent). Adequate anesthesia and a broad-spectrum microbicide should be given prior to the injection.
Prior to and 30 minutes following the intravitreal injection, patients should be monitored for elevation in intraocular pressure using tonometry. Monitoring may also consist of a check for perfusion of the optic nerve head immediately after the injection [see Warnings and Precautions (5.2)]. Patients should also be monitored for and instructed to report any symptoms suggestive of endophthalmitis without delay following the injection [see Warnings and Precautions (5.1)].
Each prefilled syringe should only be used for the treatment of a single eye. If the contralateral eye requires treatment, a new prefilled syringe should be used and the sterile field, syringe, gloves, drapes, eyelid speculum, and injection needle should be changed before LUCENTIS is administered to the other eye.
No special dosage modification is required for any of the populations that have been studied (e.g., gender, elderly).
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Endophthalmitis and Retinal Detachments
Intravitreal injections, including those with LUCENTIS, have been associated with endophthalmitis and retinal detachments. Proper aseptic injection technique should always be used when administering LUCENTIS. In addition, patients should be monitored following the injection to permit early treatment should an infection occur [see Dosage and Administration (2.6, 2.7) and Patient Counseling Information (17)].
5.2 Increases in Intraocular Pressure
Increases in intraocular pressure have been noted both pre-injection and post-injection (at 60 minutes) while being treated with LUCENTIS. Monitor intraocular pressure prior to and following intravitreal injection with LUCENTIS and manage appropriately [see Dosage and Administration (2.7)].
5.3 Thromboembolic Events
Although there was a low rate of arterial thromboembolic events (ATEs) observed in the LUCENTIS clinical trials, there is a potential risk of ATEs following intravitreal use of VEGF inhibitors. Arterial thromboembolic events are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause).
Neovascular (Wet) Age-Related Macular Degeneration
The ATE rate in the three controlled neovascular AMD studies (AMD-1, AMD-2, AMD-3) during the first year was 1.9% (17 of 874) in the combined group of patients treated with 0.3 mg or 0.5 mg LUCENTIS compared with 1.1% (5 of 441) in patients from the control arms [see Clinical Studies (14.1)]. In the second year of Studies AMD-1 and AMD-2, the ATE rate was 2.6% (19 of 721) in the combined group of LUCENTIS-treated patients compared with 2.9% (10 of 344) in patients from the control arms. In Study AMD-4, the ATE rates observed in the 0.5 mg arms during the first and second year were similar to rates observed in Studies AMD-1, AMD-2, and AMD-3.
In a pooled analysis of 2-year controlled studies [AMD-1, AMD-2, and a study of LUCENTIS used adjunctively with verteporfin photodynamic therapy (PDT)], the stroke rate (including both ischemic and hemorrhagic stroke) was 2.7% (13 of 484) in patients treated with 0.5 mg LUCENTIS compared to 1.1% (5 of 435) in patients in the control arms [odds ratio 2.2 (95% confidence interval (0.8-7.1)].
Macular Edema Following Retinal Vein Occlusion
The ATE rate in the two controlled RVO studies during the first 6 months was 0.8% in both the LUCENTIS and control arms of the studies (4 of 525 in the combined group of patients treated with 0.3 mg or 0.5 mg LUCENTIS and 2 of 260 in the control arms) [see Clinical Studies (14.2)]. The stroke rate was 0.2% (1 of 525) in the combined group of LUCENTIS-treated patients compared to 0.4% (1 of 260) in the control arms.
Diabetic Macular Edema and Diabetic Retinopathy
Safety data are derived from studies D-1 and D-2. All enrolled patients had DME and DR at baseline [see Clinical Studies (14.3, 14.4)].
In a pooled analysis of Studies D-1 and D-2 [see Clinical Studies (14.3)], the ATE rate at 2 years was 7.2% (18 of 250) with 0.5 mg LUCENTIS, 5.6% (14 of 250) with 0.3 mg LUCENTIS, and 5.2% (13 of 250) with control. The stroke rate at 2 years was 3.2% (8 of 250) with 0.5 mg LUCENTIS, 1.2% (3 of 250) with 0.3 mg LUCENTIS, and 1.6% (4 of 250) with control. At 3 years, the ATE rate was 10.4% (26 of 249) with 0.5 mg LUCENTIS and 10.8% (27 of 250) with 0.3 mg LUCENTIS; the stroke rate was 4.8% (12 of 249) with 0.5 mg LUCENTIS and 2.0% (5 of 250) with 0.3 mg LUCENTIS.
5.4 Fatal Events in Patients with Diabetic Macular Edema and Diabetic Retinopathy at Baseline
Diabetic Macular Edema and Diabetic Retinopathy
Safety data are derived from studies D-1 and D-2. All enrolled patients had DME and DR at baseline [see Clinical Studies (14.3, 14.4)].
A pooled analysis of Studies D-1 and D-2 [see Clinical Studies (14.3)], showed that fatalities in the first 2 years occurred in 4.4% (11 of 250) of patients treated with 0.5 mg LUCENTIS, in 2.8% (7 of 250) of patients treated with 0.3 mg LUCENTIS, and in 1.2% (3 of 250) of control patients. Over 3 years, fatalities occurred in 6.4% (16 of 249) of patients treated with 0.5 mg LUCENTIS and in 4.4% (11 of 250) of patients treated with 0.3 mg LUCENTIS. Although the rate of fatal events was low and included causes of death typical of patients with advanced diabetic complications, a potential relationship between these events and intravitreal use of VEGF inhibitors cannot be excluded.
5.5 Retinal Vasculitis with or without Occlusion
Retinal vasculitis with or without occlusion, typically in the presence of preexisting intraocular inflammation or post-treatment with other intravitreal agents, have been reported with the use of LUCENTIS. Discontinue treatment with LUCENTIS in patients who develop these events. Patients should be instructed to report any change in vision without delay [see Patient Counseling Information (17)].
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the label:
- Endophthalmitis and Retinal Detachments [see Warnings and Precautions (5.1)]
- Increases in Intraocular Pressure [see Warnings and Precautions (5.2)]
- Thromboembolic Events [see Warnings and Precautions (5.3)]
- Fatal Events in patients with DME and DR at baseline [see Warnings and Precautions (5.4)]
6.1 Injection Procedure
Serious adverse reactions related to the injection procedure have occurred in < 0.1% of intravitreal injections, including endophthalmitis [see Warnings and Precautions (5.1)], rhegmatogenous retinal detachment, and iatrogenic traumatic cataract.
6.2 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data below reflect exposure to 0.5 mg LUCENTIS in 440 patients with neovascular AMD in Studies AMD-1, AMD-2, and AMD-3; in 259 patients with macular edema following RVO. The data also reflect exposure to 0.3 mg LUCENTIS in 250 patients with DME and DR at baseline [see Clinical Studies (14)].
Safety data observed in 224 patients with mCNV, as well as Studies AMD-4 and D-3, were consistent with these results. On average, the rates and types of adverse reactions in patients were not significantly affected by dosing regimen.
Ocular Reactions
Table 1 shows frequently reported ocular adverse reactions in LUCENTIS-treated patients compared with the control group.
Table 1 Ocular Reactions in the DME and DR, AMD, and RVO Studies DME and DR
2-yearAMD
2-yearAMD
1-yearRVO
6-monthAdverse Reaction LUCENTIS
0.3 mgControl LUCENTIS
0.5 mgControl LUCENTIS
0.5 mgControl LUCENTIS
0.5 mgControl n=250 n=250 n=379 n=379 n=440 n=441 n=259 n=260 Conjunctival hemorrhage 47% 32% 74% 60% 64% 50% 48% 37% Eye pain 17% 13% 35% 30% 26% 20% 17% 12% Vitreous floaters 10% 4% 27% 8% 19% 5% 7% 2% Intraocular pressure increased 18% 7% 24% 7% 17% 5% 7% 2% Vitreous detachment 11% 15% 21% 19% 15% 15% 4% 2% Intraocular inflammation 4% 3% 18% 8% 13% 7% 1% 3% Cataract 28% 32% 17% 14% 11% 9% 2% 2% Foreign body sensation in eyes 10% 5% 16% 14% 13% 10% 7% 5% Eye irritation 8% 5% 15% 15% 13% 12% 7% 6% Lacrimation increased 5% 4% 14% 12% 8% 8% 2% 3% Blepharitis 3% 2% 12% 8% 8% 5% 0% 1% Dry eye 5% 3% 12% 7% 7% 7% 3% 3% Visual disturbance or vision blurred 8% 4% 18% 15% 13% 10% 5% 3% Eye pruritis 4% 4% 12% 11% 9% 7% 1% 2% Ocular hyperemia 9% 9% 11% 8% 7% 4% 5% 3% Retinal disorder 2% 2% 10% 7% 8% 4% 2% 1% Maculopathy 5% 7% 9% 9% 6% 6% 11% 7% Retinal degeneration 1% 0% 8% 6% 5% 3% 1% 0% Ocular discomfort 2% 1% 7% 4% 5% 2% 2% 2% Conjunctival hyperemia 1% 2% 7% 6% 5% 4% 0% 0% Posterior capsule opacification 4% 3% 7% 4% 2% 2% 0% 1% Injection site hemorrhage 1% 0% 5% 2% 3% 1% 0% 0% Non-Ocular Reactions
Non-ocular adverse reactions with an incidence of ≥ 5% in patients receiving LUCENTIS for DR, DME, AMD, and/or RVO and which occurred at a ≥ 1% higher frequency in patients treated with LUCENTIS compared to control are shown in Table 2. Though less common, wound healing complications were also observed in some studies.
Table 2 Non-Ocular Reactions in the DME and DR, AMD, and RVO Studies DME and DR
2-yearAMD
2-yearAMD
1-yearRVO
6-monthAdverse Reaction LUCENTIS
0.3 mgControl LUCENTIS
0.5 mgControl LUCENTIS
0.5 mgControl LUCENTIS
0.5 mgControl n=250 n=250 n=379 n=379 n=440 n=441 n=259 n=260 Nasopharyngitis 12% 6% 16% 13% 8% 9% 5% 4% Anemia 11% 10% 8% 7% 4% 3% 1% 1% Nausea 10% 9% 9% 6% 5% 5% 1% 2% Cough 9% 4% 9% 8% 5% 4% 1% 2% Constipation 8% 4% 5% 7% 3% 4% 0% 1% Seasonal allergy 8% 4% 4% 4% 2% 2% 0% 2% Hypercholesterolemia 7% 5% 5% 5% 3% 2% 1% 1% Influenza 7% 3% 7% 5% 3% 2% 3% 2% Renal failure 7% 6% 1% 1% 0% 0% 0% 0% Upper respiratory tract infection 7% 7% 9% 8% 5% 5% 2% 2% Gastroesophageal reflux disease 6% 4% 4% 6% 3% 4% 1% 0% Headache 6% 8% 12% 9% 6% 5% 3% 3% Edema peripheral 6% 4% 3% 5% 2% 3% 0% 1% Renal failure chronic 6% 2% 0% 1% 0% 0% 0% 0% Neuropathy peripheral 5% 3% 1% 1% 1% 0% 0% 0% Sinusitis 5% 8% 8% 7% 5% 5% 3% 2% Bronchitis 4% 4% 11% 9% 6% 5% 0% 2% Atrial fibrillation 3% 3% 5% 4% 2% 2% 1% 0% Arthralgia 3% 3% 11% 9% 5% 5% 2% 1% Chronic obstructive pulmonary disease 1% 1% 6% 3% 3% 1% 0% 0% Wound healing complications 1% 0% 1% 1% 1% 0% 0% 0% 6.3 Immunogenicity
As with all therapeutic proteins, there is the potential for an immune response in patients treated with LUCENTIS. The immunogenicity data reflect the percentage of patients whose test results were considered positive for antibodies to LUCENTIS in immunoassays and are highly dependent on the sensitivity and specificity of the assays.
The pre-treatment incidence of immunoreactivity to LUCENTIS was 0%-5% across treatment groups. After monthly dosing with LUCENTIS for 6 to 24 months, antibodies to LUCENTIS were detected in approximately 1%-9% of patients.
The clinical significance of immunoreactivity to LUCENTIS is unclear at this time. Among neovascular AMD patients with the highest levels of immunoreactivity, some were noted to have iritis or vitritis. Intraocular inflammation was not observed in patients with DME and DR at baseline, or RVO patients with the highest levels of immunoreactivity.
6.4 Postmarketing Experience
The following adverse reaction has been identified during post-approval use of LUCENTIS. Because this reaction was reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
- Ocular: Tear of retinal pigment epithelium among patients with neovascular AMD
-
7 DRUG INTERACTIONS
Drug interaction studies have not been conducted with LUCENTIS.
LUCENTIS intravitreal injection has been used adjunctively with PDT. Twelve of 105 (11%) patients with neovascular AMD developed serious intraocular inflammation; in 10 of the 12 patients, this occurred when LUCENTIS was administered 7 days (± 2 days) after PDT.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of LUCENTIS administration in pregnant women.
Administration of ranibizumab to pregnant monkeys throughout the period of organogenesis resulted in a low incidence of skeletal abnormalities at intravitreal doses 13-times the predicted human exposure (based on maximal serum trough levels [Cmax]) after a single eye treatment at the recommended clinical dose. No skeletal abnormalities were observed at serum trough levels equivalent to the predicted human exposure after a single eye treatment at the recommended clinical dose [see Animal Data].
Animal reproduction studies are not always predictive of human response, and it is not known whether ranibizumab can cause fetal harm when administered to a pregnant woman. Based on the anti-VEGF mechanism of action for ranibizumab [see Clinical Pharmacology (12.1)], treatment with LUCENTIS may pose a risk to human embryofetal development.
LUCENTIS should be given to a pregnant woman only if clearly needed.
Data
Animal Data
An embryo-fetal developmental toxicity study was performed on pregnant cynomolgus monkeys. Pregnant animals received intravitreal injections of ranibizumab every 14 days starting on Day 20 of gestation, until Day 62 at doses of 0, 0.125, and 1 mg/eye. Skeletal abnormalities including incomplete and/or irregular ossification of bones in the skull, vertebral column, and hindlimbs and shortened supernumerary ribs were seen at a low incidence in fetuses from animals treated with 1 mg/eye of ranibizumab. The 1 mg/eye dose resulted in trough serum ranibizumab levels up to 13 times higher than predicted Cmax levels with single eye treatment in humans. No skeletal abnormalities were seen at the lower dose of 0.125 mg/eye, a dose which resulted in trough exposures equivalent to single eye treatment in humans. No effect on the weight or structure of the placenta, maternal toxicity, or embryotoxicity was observed.
8.2 Lactation
Risk Summary
There are no data available on the presence of ranibizumab in human milk, the effects of ranibizumab on the breastfed infant or the effects of ranibizumab on milk production/excretion.
Because many drugs are excreted in human milk, and because the potential for absorption and harm to infant growth and development exists, caution should be exercised when LUCENTIS is administered to a nursing woman.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for LUCENTIS and any potential adverse effects on the breastfed child from ranibizumab.
8.4 Pediatric Use
The safety and effectiveness of LUCENTIS in pediatric patients have not been established.
8.5 Geriatric Use
In the clinical studies, approximately 76% (2449 of 3227) of patients randomized to treatment with LUCENTIS were ≥ 65 years of age and approximately 51% (1644 of 3227) were ≥ 75 years of age [see Clinical Studies (14)]. No notable differences in efficacy or safety were seen with increasing age in these studies. Age did not have a significant effect on systemic exposure.
- 10 OVERDOSAGE
-
11 DESCRIPTION
LUCENTIS® (ranibizumab injection) is a recombinant humanized IgG1 kappa isotype monoclonal antibody fragment designed for intraocular use. Ranibizumab binds to and inhibits the biologic activity of human vascular endothelial growth factor A (VEGF-A). Ranibizumab, which lacks an Fc region, has a molecular weight of approximately 48 kilodaltons and is produced by an E. coli expression system in a nutrient medium containing the antibiotic tetracycline. Tetracycline is not detectable in the final product.
LUCENTIS is a sterile, colorless to pale yellow solution in a single-dose prefilled syringe. LUCENTIS is supplied as a preservative-free, sterile solution in a single-dose container designed to deliver 0.05 mL of 10 mg/mL LUCENTIS (0.5 mg dose prefilled syringe) or 6 mg/mL LUCENTIS (0.3 mg dose prefilled syringe) aqueous solution with 10 mM histidine HCl, 10% α,α-trehalose dihydrate, 0.01% polysorbate 20, pH 5.5.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ranibizumab binds to the receptor binding site of active forms of VEGF-A, including the biologically active, cleaved form of this molecule, VEGF110. VEGF-A has been shown to cause neovascularization and leakage in models of ocular angiogenesis and vascular occlusion and is thought to contribute to pathophysiology of neovascular AMD, mCNV, DR, DME and macular edema following RVO. The binding of ranibizumab to VEGF-A prevents the interaction of VEGF-A with its receptors (VEGFR1 and VEGFR2) on the surface of endothelial cells, reducing endothelial cell proliferation, vascular leakage, and new blood vessel formation.
12.2 Pharmacodynamics
Increased retinal thickness (i.e., center point thickness (CPT) or central foveal thickness (CFT)), as assessed by optical coherence tomography (OCT) is associated with neovascular AMD, mCNV, macular edema following RVO, and DME. Leakage from choroidal neovascularization (CNV) as assessed by fluorescein angiography (FA) is associated with neovascular AMD and mCNV. Microvascular retinal changes and neovascularization, as assessed by color fundus photography, are associated with diabetic retinopathy.
Neovascular (Wet) Age-Related Macular Degeneration
In Study AMD-3, CPT was assessed by time domain (TD)-OCT in 118 of 184 patients. TD-OCT measurements were collected at baseline, Months 1, 2, 3, 5, 8, and 12. In patients treated with LUCENTIS, CPT decreased, on average, more than in the sham group from baseline through Month 12. CPT decreased by Month 1 and decreased further at Month 3, on average. In this study, CPT data did not provide information useful in influencing treatment decisions [see Clinical Studies (14.1)].
In Study AMD-4, CFT was assessed by spectral domain (SD)-OCT in all patients; on average, CFT reductions were observed beginning at Day 7 following the first LUCENTIS injection through Month 24. CFT data did not provide information capable of predicting final visual acuity results [see Clinical Studies (14.1)].
In patients treated with LUCENTIS, the area of CNV leakage, on average, decreased by Month 3 as assessed by FA. The area of CNV leakage for an individual patient was not correlated with visual acuity.
Macular Edema Following Retinal Vein Occlusion
On average, CPT reductions were observed in Studies RVO-1 and RVO-2 beginning at Day 7 following the first LUCENTIS injection through Month 6. CPT was not evaluated as a means to guide treatment decisions [see Clinical Studies (14.2)].
Diabetic Macular Edema
On average, CPT reductions were observed in Studies D-1 and D-2 beginning at Day 7 following the first LUCENTIS injection through Month 36. CPT data did not provide information useful in influencing treatment decisions [see Clinical Studies (14.3)].
Diabetic Retinopathy
Improvements from baseline in DR severity as assessed on fundus photography were observed in Studies D-1 and D-2 at Month 3 (first scheduled DR photographic assessment after randomization) through Month 36 [see Clinical Studies (14.4)].
Myopic Choroidal Neovascularization
On average CFT reductions were observed as early as Month 1, and were greater in the LUCENTIS groups compared to PDT [see Clinical Studies (14.5)].
12.3 Pharmacokinetics
In patients with neovascular AMD, following monthly intravitreal administration of 0.5 mg LUCENTIS, mean (±SD) maximum ranibizumab serum concentrations were 1.7 (± 1.1) ng/mL. These concentrations were below the concentration range of ranibizumab (11 to 27 ng/mL) that was necessary to inhibit the biological activity of VEGF-A by 50%, as measured in an in vitro cellular proliferation assay (based on human umbilical vein endothelial cells (HUVEC)). No significant change from baseline was observed in the mean plasma VEGF concentrations following three monthly 0.5 mg intravitreal injections. The maximum observed serum concentration was dose proportional over the dose range of 0.05 to 2 mg/eye. Serum ranibizumab concentrations in RVO and DME and DR patients were similar to those observed in neovascular AMD patients.
Based on a population pharmacokinetic analysis of patients with neovascular AMD, maximum serum concentrations are predicted to be reached at approximately 1 day after monthly intravitreal administration of LUCENTIS 0.5 mg/eye. Based on the disappearance of ranibizumab from serum, the estimated average vitreous elimination half-life was approximately 9 days. Steady-state minimum concentration is predicted to be 0.22 ng/mL with a monthly dosing regimen. In humans, serum ranibizumab concentrations are predicted to be approximately 90,000-fold lower than vitreal concentrations.
In pharmacokinetic covariate analyses, 48% (520/1091) of patients had renal impairment (35% mild, 11% moderate, and 2% severe). Because the increases in plasma ranibizumab exposures in these patients are not considered clinically significant, no dosage adjustment is needed based on renal impairment status.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been conducted to determine the carcinogenic potential of ranibizumab. Based on the anti-VEGF mechanism of action of ranibizumab, treatment with LUCENTIS may pose a risk to reproductive capacity [see Females and Males of Reproductive Potential (8.3)].
-
14 CLINICAL STUDIES
Unless otherwise noted, visual acuity was measured at a distance of 4 meters.
14.1 Neovascular (Wet) Age-Related Macular Degeneration (AMD)
The safety and efficacy of LUCENTIS were assessed in three randomized, double-masked, sham- or active-controlled studies in patients with neovascular AMD. A total of 1323 patients (LUCENTIS 879, control 444) were enrolled in the three studies.
Studies AMD-1 and AMD-2
In Study AMD-1, patients with minimally classic or occult (without classic) CNV lesions received monthly LUCENTIS 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections. Data are available through Month 24. Patients treated with LUCENTIS in Study AMD-1 received a mean of 22 total treatments out of a possible 24 from Day 0 to Month 24.
In Study AMD-2, patients with predominantly classic CNV lesions received one of the following: 1) monthly LUCENTIS 0.3 mg intravitreal injections and sham PDT; 2) monthly LUCENTIS 0.5 mg intravitreal injections and sham PDT; or 3) sham intravitreal injections and active PDT. Sham PDT (or active PDT) was given with the initial LUCENTIS (or sham) intravitreal injection and every 3 months thereafter if FA showed persistence or recurrence of leakage. Data are available through Month 24. Patients treated with LUCENTIS in Study AMD-2 received a mean of 21 total treatments out of a possible 24 from Day 0 through Month 24.
In both studies, the primary efficacy endpoint was the proportion of patients who maintained vision, defined as losing fewer than 15 letters of visual acuity at 12 months compared with baseline. Almost all LUCENTIS-treated patients (approximately 95%) maintained their visual acuity. Among LUCENTIS-treated patients, 31% to 37% experienced a clinically significant improvement in vision, defined as gaining 15 or more letters at 12 months. The size of the lesion did not significantly affect the results. Detailed results are shown in Table 3, Table 4, and Figure 1 below.
Table 3 Visual Acuity Outcomes at Month 12 and Month 24 in Study AMD-1 Outcome Measure Month Sham
n=229LUCENTIS
0.5 mg
n=230Estimated Difference
(95% CI)*- *
- Adjusted estimate based on the stratified model; p < 0.01
Loss of <15 letters in visual acuity (%) 12 60% 91% 30%
(23%, 37%)24 56% 89% 33%
(26%, 41%)Gain of ≥15 letters in visual acuity (%) 12 6% 31% 25%
(18%, 31%)24 4% 30% 25%
(18%, 31%)Mean change in visual acuity (letters) (SD) 12 -11.0 (17.9) +6.3 ( 14.1) 17.1
(14.2, 20.0)24 -15.0 (19.7) +5.5 ( 15.9) 20.1
(16.9, 23.4)Table 4 Visual Acuity Outcomes at Month 12 and Month 24 in Study AMD-2 Outcome Measure Month PDT
n=141LUCENTIS
0.5 mg
n=139Estimated Difference
(95% CI)*- *
- Adjusted estimate based on the stratified model; p < 0.01
Loss of <15 letters in visual acuity (%) 12 66% 98% 32%
(24%, 40%)24 65% 93% 28%
(19%, 37%)Gain of ≥15 letters in visual acuity (%) 12 11% 37% 26%
(17%, 36%)24 9% 37% 29%
(20%, 39%)Mean change in visual acuity (letters) (SD) 12 -8.5 (17.8) +11.0 (15.8) 19.8
(15.9, 23.7)24 -9.1 (18.7) +10.9 (17.3) 20
(16.0, 24.4)- *
- Visual acuity was measured at a distance of 2 meters
Figure 1
Mean Change in Visual Acuity* from Baseline to Month 24 in Study AMD-1 and Study AMD-2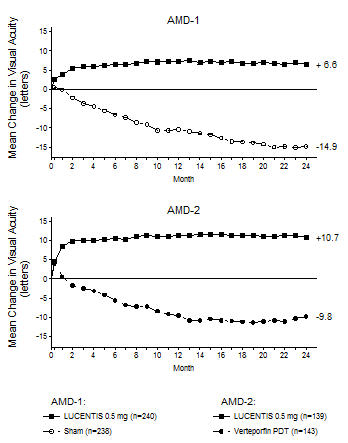
Patients in the group treated with LUCENTIS had minimal observable CNV lesion growth, on average. At Month 12, the mean change in the total area of the CNV lesion was 0.1-0.3 disc areas (DA) for LUCENTIS versus 2.3-2.6 DA for the control arms. At Month 24, the mean change in the total area of the CNV lesion was 0.3-0.4 DA for LUCENTIS versus 2.9-3.1 DA for the control arms.
Study AMD-3
Study AMD-3 was a randomized, double-masked, sham-controlled, 2-year study designed to assess the safety and efficacy of LUCENTIS in patients with neovascular AMD (with or without a classic CNV component). Data are available through Month 12. Patients received LUCENTIS 0.3 mg or 0.5 mg intravitreal injections or sham injections once a month for three consecutive doses, followed by a dose administered once every 3 months for 9 months. A total of 184 patients were enrolled in this study (LUCENTIS 0.3 mg, 60; LUCENTIS 0.5 mg, 61; sham, 63); 171 (93%) completed 12 months of this study. Patients treated with LUCENTIS in Study AMD-3 received a mean of six total treatments out of a possible 6 from Day 0 through Month 12.
In Study AMD-3, the primary efficacy endpoint was the mean change in visual acuity at 12 months compared with baseline (see Figure 2). After an initial increase in visual acuity (following monthly dosing), on average, patients dosed once every 3 months with LUCENTIS lost visual acuity, returning to baseline at Month 12. In Study AMD-3, almost all LUCENTIS-treated patients (90%) lost fewer than 15 letters of visual acuity at Month 12.
Figure 2
Mean Change in Visual Acuity from Baseline to Month 12 in Study AMD-3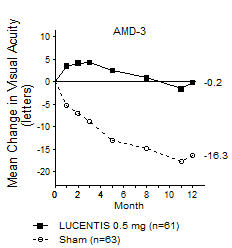
Study AMD-4
Study AMD-4 was a randomized, double-masked, active treatment-controlled, two-year study designed to assess the safety and efficacy of LUCENTIS 0.5 mg administered monthly or less frequently than monthly in patients with neovascular AMD. Patients randomized to the LUCENTIS 0.5 mg less frequent dosing arm received three monthly doses followed by monthly assessments where patients were eligible to receive LUCENTIS injections guided by pre-specified re-treatment criteria. A total of 550 patients were enrolled in the two 0.5 mg treatment groups with 467 (85%) completing through Month 24. Data are available through Month 24.
Clinical results at Month 24 remain similar to that observed at Month 12.
From Month 3 through Month 24, visual acuity decreased by 0.3 letters in the 0.5 mg less frequent dosing arm and increased by 0.7 letters in the 0.5 mg monthly arm (see Figure 3). Over this 21-month period, patients in the 0.5 mg less frequent dosing and the 0.5 mg monthly arms averaged 10.3 and 18.5 injections, respectively. The distribution of injections received in the less frequent dosing arm is shown in Figure 4.
Figure 3
Mean Change in Visual Acuity from Baseline to Month 24 in Study AMD-4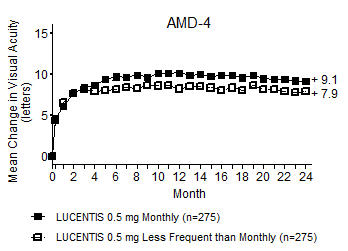
Figure 4
Distribution of Injections from Month 3 to Month 24 in the Less Frequent Dosing Arm in Study AMD-4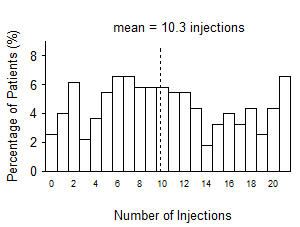
14.2 Macular Edema Following Retinal Vein Occlusion (RVO)
The safety and efficacy of LUCENTIS were assessed in two randomized, double-masked, 1-year studies in patients with macular edema following RVO. Sham controlled data are available through Month 6. Patient age ranged from 20 to 91 years, with a mean age of 67 years. A total of 789 patients (LUCENTIS 0.3 mg, 266 patients; LUCENTIS 0.5 mg, 261 patients; sham, 262 patients) were enrolled, with 739 (94%) patients completing through Month 6. All patients completing Month 6 were eligible to receive LUCENTIS injections guided by pre-specified re-treatment criteria until the end of the studies at Month 12.
In Study RVO-1, patients with macular edema following branch or hemi-RVO, received monthly LUCENTIS 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections for 6 months. All patients were eligible for macular focal/grid laser treatment beginning at Month 3 of the 6-month treatment period. Macular focal/grid laser treatment was given to 26 of 131 (20%) patients treated with 0.5 mg LUCENTIS and 71 of 132 (54%) patients treated with sham.
In Study RVO-2, patients with macular edema following central RVO received monthly LUCENTIS 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections for 6 months.
At Month 6, after monthly treatment with 0.5 mg LUCENTIS, the following clinical results were observed:
Table 5 Visual Acuity Outcomes at Month 6 in Study RVO-1 and Study RVO-2 Outcome Measure Study * Sham LUCENTIS
0.5 mgEstimated Difference
(95% CI) †Gain of ≥15 letters in visual acuity (%) RVO-1 29% 61% 31%
(20%, 43%)Gain of ≥15 letters in visual acuity (%) RVO-2 17% 48% 30%
(20%, 41%)p < 0.01 for all time points Figure 5
Mean Change in Visual Acuity from Baseline to Month 6 in Study RVO-1 and Study RVO-2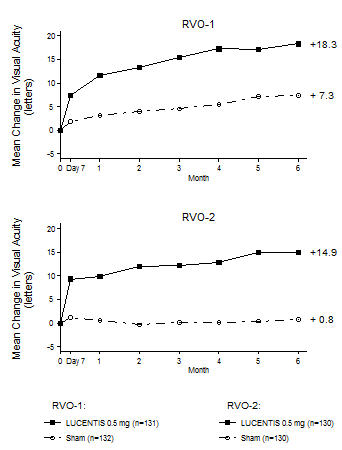
14.3 Diabetic Macular Edema (DME)
Efficacy and safety data of LUCENTIS are derived from studies D-1 and D-2 (See Section 14.4 Diabetic Retinopathy below). All enrolled patients had DR and DME at baseline.
The safety and efficacy of LUCENTIS were assessed in two randomized, double-masked, 3-year studies. The studies were sham-controlled through Month 24. Patient age ranged from 21 to 91 years, with a mean age of 62 years. A total of 759 patients (LUCENTIS 0.3 mg, 250 patients; LUCENTIS 0.5 mg, 252 patients; sham, 257 patients) were enrolled, with 582 (77%) completing through Month 36.
In Studies D-1 and D-2, patients received monthly LUCENTIS 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections during the 24-month controlled treatment period. From Months 25 through 36, patients who previously received sham were eligible to receive monthly LUCENTIS 0.5 mg and patients originally randomized to monthly LUCENTIS 0.3 mg or 0.5 mg continued to receive their assigned dose. All patients were eligible for macular focal/grid laser treatment beginning at Month 3 of the 24-month treatment period or panretinal photocoagulation (PRP) as needed. Through Month 24, macular focal/grid laser treatment was administered in 94 of 250 (38%) patients treated with LUCENTIS 0.3 mg and 185 of 257 (72%) patients treated with sham; PRP was administered in 2 of 250 (1%) patients treated with LUCENTIS 0.3 mg and 30 of 257 (12%) patients treated with sham.
Compared to monthly LUCENTIS 0.3 mg, no additional benefit was observed with monthly treatment with LUCENTIS 0.5 mg. At Month 24, after monthly treatment with LUCENTIS 0.3 mg, the following clinical results were observed:
Table 6 Visual Acuity Outcomes at Month 24 in Study D-1 and D-2 Outcome Measure Study* Sham LUCENTIS
0.3 mgEstimated Difference
(95% CI)†Gain of ≥15 letters in visual acuity (%) D-1 12% 34% 21%
(11%, 30%)D-2 18% 45% 24%
(14%, 35%)Loss of <15 letters in visual acuity (%) D-1 92% 98% 7%
(2%, 13%)D-2 90% 98% 8%
(2%, 14%)Mean change in visual acuity (letters) D-1 2.3 10.9 8.5
(5.4, 11.5)D-2 2.6 12.5 9.6
(6.1, 13.0)p < 0.01 for all time points comparing LUCENTIS 0.3 mg to sham through Month 24 Figure 6
Mean Change in Visual Acuity from Baseline to Month 36 in Study D-1 and Study D-2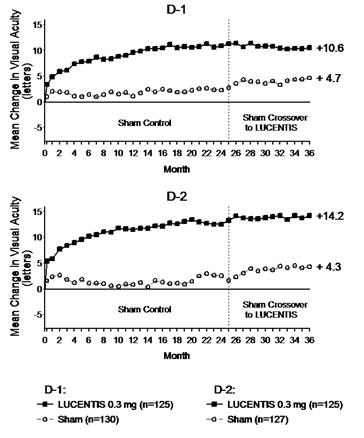
Visual acuity outcomes observed at Month 24 in patients treated with LUCENTIS 0.3 mg were maintained with continued treatment through Month 36 in both DME studies. Patients in the sham arms who received LUCENTIS 0.5 mg beginning at Month 25 achieved lesser VA gains compared to patients who began treatment with LUCENTIS at the beginning of the studies.
In Studies D-1 and D-2, patients received monthly injections of LUCENTIS for 12 or 36 months, after which 500 patients opted to continue in the long-term follow-up study. Of 298 patients who had at least 12 months of follow-up from Month 36, 58 (19.5%) patients maintained vision with no further therapy. The remaining 202 patients were followed for less than 12 months.
14.4 Diabetic Retinopathy (DR)
Efficacy and safety data of LUCENTIS are derived from Studies D-1 and D-2 [see Clinical Studies (14.3)] and D-3. All enrolled patients in Studies D-1 and D-2 had DR and DME at baseline. Study D-3 enrolled DR patients both with and without DME at baseline.
Of the 759 patients enrolled in Studies D-1 and D-2, 746 patients had a baseline assessment of fundus photography. Patients had baseline Early Treatment Diabetic Retinopathy Study Diabetic Retinopathy Severity Scores (ETDRS-DRSS) ranging from 10 to 75. At baseline, 62% of patients had non-proliferative diabetic retinopathy (NPDR) (ETDRS-DRSS less than 60) and 31% had proliferative diabetic retinopathy (PDR) (ETDRS-DRSS greater than or equal to 60). The ETDRS-DRSS could not be graded in 5% of patients at baseline, and 2% of patients had absent or questionable DR at baseline. Approximately 20% of the overall population had prior PRP.
After monthly treatment with LUCENTIS 0.3 mg, the following clinical results were observed (Table 7; Figure 7):
Table 7 ≥3-Step and ≥2-Step Improvement at Month 24 in Study D-1 and Study D-2 Outcome Measure Study* Sham LUCENTIS
0.3 mgEstimated Difference
(95% CI)†- *
- D-1: Sham, n=124; LUCENTIS 0.3 mg, n=117
D-2: Sham, n=115; LUCENTIS 0.3 mg, n=117 - †
- Adjusted estimate based on stratified model
- ‡
- p < 0.05 for all time points comparing LUCENTIS 0.3 mg to sham from Month 12 through Month 24
- §
- p < 0.05 for all time points comparing LUCENTIS 0.3 mg to sham from Month 3 through Month 24
≥3-step improvement from baseline in ETDRS-DRSS ‡ D-1 2% 17% 15%
(7%, 22%)D-2 0% 9% 9%
(4%, 14%)≥2-step improvement from baseline in ETDRS-DRSS § D-1 4% 39% 35%
(26%, 44%)D-2 7% 37% 31%
(21%, 40%)At Month 24, DR improvement by ≥3-steps in ETDRS-DRSS from baseline in subgroups examined (e.g., age, gender, race, baseline visual acuity, baseline HbA1c, prior DME therapy at baseline, baseline DR severity (NPDR, PDR)) were generally consistent with the results in the overall population.
The difference in the proportion of patients treated with LUCENTIS 0.3 mg compared to sham who achieved DR improvement based on the ETDRS-DRSS was observed as early as Month 3 for ≥2-step improvement or at Month 12 for ≥3-step improvement.
Figure 7
Proportion of Patients with ≥ 3-Step and ≥ 2-Step Improvement from Baseline in ETDRS Diabetic Retinopathy Severity Level over Time in Study D-1 and Study D-2≥3-Step Improvement from Baseline Over Time ≥2-Step Improvement from Baseline Over Time 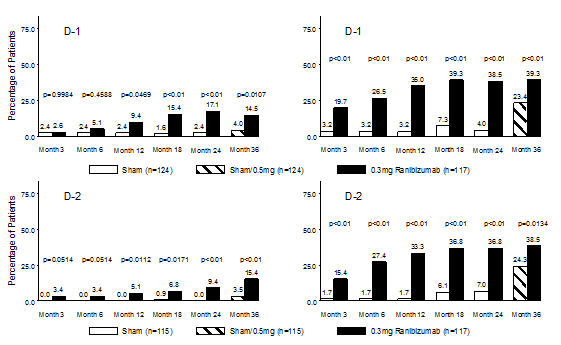
Study D-3 enrolled DR patients with and without DME; 88 (22%) eyes with baseline DME and 306 (78%) eyes without baseline DME and balanced across treatment groups. Study D-3 was a randomized, active-controlled study where patient age ranged from 20 to 83 with a mean age of 51 years. A total of 394 study eyes from 305 patients, including 89 who had both eyes randomized, were enrolled (LUCENTIS, 191 study eyes; pan-retinal photocoagulation; 203 study eyes). All eyes in the LUCENTIS group received a baseline 0.5 mg intravitreal injection followed by 3 monthly intravitreal injections, after which treatment was guided by pre-specified re-treatment criteria. Patients had baseline ETDRS-DRSS ranging from 20 to 85. At baseline, 11% of eyes had NPDR (ETDRS-DRSS less than 60), 50% had mild-to-moderate PDR (ETDRS-DRSS equal to 60, 61, or 65), and 37% had high-risk PDR (ETDRS-DRSS greater than or equal to 71).
An analysis of data from Study D-3 demonstrated that at Year 2 in the LUCENTIS group, 31.7% and 28.4% of eyes in the subgroups with baseline DME and without baseline DME, respectively, had ≥ 3-step improvement from baseline in ETDRS-DRSS.
Table 8 Proportion of Eyes with ≥ 3-Step and ≥ 2-Step Improvement from Baseline in ETDRS-DRSS at Year 2 in Study D-3 LUCENTIS group Outcome Measure
(in ETDRS-DRSS)Eyes with Baseline DME
n = 41Eyes without Baseline DME
n = 148≥ 3-step improvement from baseline
95% CI for percentage13 (31.7%)
(17.5%, 46.0%)42 (28.4%)
(21.1%, 35.6%)≥ 2-step improvement from baseline
95% CI for percentage24 (58.5%)
(43.5%, 73.6%)56 (37.8%)
(30.0%, 45.7%)Figure 8
Proportion of Eyes in the LUCENTIS group with ≥ 3-Step and ≥ 2-Step Improvement from Baseline in ETDRS-DRSS at Year 1 and Year 2 in Study D-3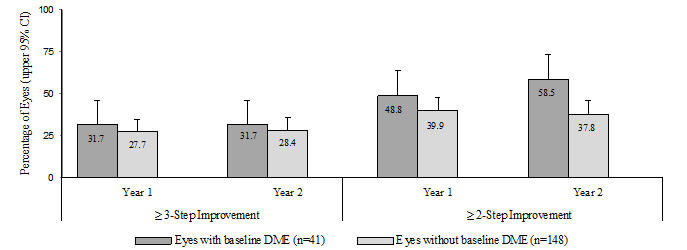
14.5 Myopic Choroidal Neovascularization (mCNV)
The efficacy and safety data of LUCENTIS were assessed in a randomized, double-masked, active-controlled 3-month study in patients with mCNV. Patients age ranged from 18 to 87 years, with a mean age of 55 years. A total of 276 patients (222 patients in the LUCENTIS treated Groups I and II; 55 patients in the active control PDT group) were enrolled. Patients randomized to the LUCENTIS groups received injections guided by pre-specified re-treatment criteria. The retreatment criteria in Group I were vision stability guided, with the Best Corrected Visual Acuity (BCVA) at the current visit being assessed for changes compared with the two preceding monthly BCVA values. The retreatment criteria in Group II were disease activity guided, based on BCVA decrease from the previous visit that was attributable to intra- or sub-retinal fluid or active leakage secondary to mCNV as assessed by OCT and/or FA compared to the previous monthly visit.
Visual gains for the two LUCENTIS 0.5 mg treatment arms were superior to the active control arm. The mean change in BCVA from baseline at Month 3 was: +12.1 letters for Group I, +12.5 letters for Group II and +1.4 letters for the PDT group. (Figure 9; Table 9). Efficacy was comparable between Group I and Group II.
Table 9 Mean Change in Visual Acuity and Proportion of Patients who Gained ≥15 letters from Baseline at Month 3 Study Arms Mean change in BCVA from baseline (Letters) Proportion of patients who gained ≥15 letters from baseline Mean (SD) Estimated Difference
(95% CI)*Percent Estimated Difference
(95% CI)*- *
- Adjusted estimates based on stratified models; p < 0.01
Group I 12.1 (10.2) 10.9
(7.6, 14.3)37.1 22.6
(9.5, 35.7)Group II 12.5 (8.8) 11.4
(8.3, 14.5)40.5 26.0
(13.1, 38.9)Control (PDT) 1.4 (12.2) 14.5 Figure 9
Mean Change in Visual Acuity from Baseline to Month 3 in mCNV Study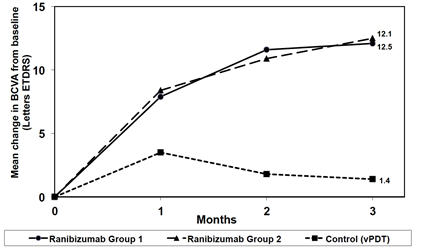
The proportion of patients who gained ≥15 letters (ETDRS) by Month 3 was 37.1% and 40.5% for LUCENTIS Groups I and II, respectively and 14.5% for the PDT group. The mean number of injections between baseline and Month 3 was 2.5 and 1.8 for Groups I and II, respectively. 41% of patients received 1, 2 or 3 injections between baseline and Month 3 with no injections afterwards.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
- Each LUCENTIS 0.5 mg carton (NDC 50242-080-03) contains a single-dose, prefilled syringe designed to deliver 0.05 mL of 10 mg/mL ranibizumab solution. The prefilled syringe has a non-retractable plunger stopper and a syringe cap consisting of a tamper-evident rigid seal with a rubber tip cap including a Luer lock adapter. The prefilled syringe has a plunger rod and a CLEAR finger grip. Each prefilled syringe is sterile and is packed in a sealed tray.
- Each LUCENTIS 0.3 mg carton (NDC 50242-082-03) contains a single-dose, prefilled syringe designed to deliver 0.05 mL of 6 mg/mL ranibizumab solution. The prefilled syringe has a non-retractable plunger stopper and a syringe cap consisting of a tamper-evident rigid seal with a rubber tip cap including a Luer lock adapter. The prefilled syringe has a plunger rod and an ORANGE finger grip. Each prefilled syringe is sterile and is packed in a sealed tray.
EACH CARTON IS FOR SINGLE-EYE USE ONLY.
-
17 PATIENT COUNSELING INFORMATION
Advise patients that in the days following LUCENTIS administration, patients are at risk of developing endophthalmitis and retinal vasculitis with or without occlusion. If the eye becomes red, sensitive to light, painful, or develops a change in vision, advise the patient to seek immediate care from an ophthalmologist [see Warnings and Precautions (5.1, 5.5)].
-
SPL UNCLASSIFIED SECTION
LUCENTIS® (ranibizumab injection) Manufactured by:
Genentech, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990LUCENTIS® is a registered trademark of Genentech, Inc.
©2024 Genentech, Inc.Representative sample of labeling (see the HOW SUPPLIED section for complete listing):
-
PRINCIPAL DISPLAY PANEL - 0.5 mg Syringe Carton
NDC 50242-080-03
LUCENTIS®
RANIBIZUMAB INJECTION0.5 mg DOSE PREFILLED SYRINGE
STERILE. SINGLE-DOSE.
INDICATED FOR
Neovascular (wet) age-related macular degeneration (wAMD)
Macular edema following retinal vein occlusion (RVO)
Myopic choroidal neovascularization (mCNV)0.5 mg
wAMD | RVO | mCNVFor intravitreal injection only.
Rx only
KEEP REFRIGERATED. DO NOT FREEZE.
PROTECT FROM LIGHT.Genentech
11013083

- PRINCIPAL DISPLAY PANEL - 0.3 mg Syringe Carton
-
INGREDIENTS AND APPEARANCE
LUCENTIS
ranibizumab injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:50242-080 Route of Administration INTRAVITREAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RANIBIZUMAB (UNII: ZL1R02VT79) (RANIBIZUMAB - UNII:ZL1R02VT79) RANIBIZUMAB 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength HISTIDINE MONOHYDROCHLORIDE (UNII: 1D5Q932XM6) 1.66 mg in 1 mL HISTIDINE (UNII: 4QD397987E) 0.32 mg in 1 mL TREHALOSE DIHYDRATE (UNII: 7YIN7J07X4) 100 mg in 1 mL POLYSORBATE 20 (UNII: 7T1F30V5YH) 0.1 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50242-080-03 1 in 1 CARTON 10/13/2016 1 0.05 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) 2 NDC:50242-080-88 1 in 1 CARTON 10/13/2016 2 0.05 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125156 06/30/2006 LUCENTIS
ranibizumab injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:50242-082 Route of Administration INTRAVITREAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RANIBIZUMAB (UNII: ZL1R02VT79) (RANIBIZUMAB - UNII:ZL1R02VT79) RANIBIZUMAB 6 mg in 1 mL Inactive Ingredients Ingredient Name Strength HISTIDINE MONOHYDROCHLORIDE (UNII: 1D5Q932XM6) 1.66 mg in 1 mL HISTIDINE (UNII: 4QD397987E) 0.32 mg in 1 mL TREHALOSE DIHYDRATE (UNII: 7YIN7J07X4) 100 mg in 1 mL POLYSORBATE 20 (UNII: 7T1F30V5YH) 0.1 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50242-082-88 1 in 1 CARTON 03/20/2018 1 0.05 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) 2 NDC:50242-082-03 1 in 1 CARTON 03/20/2018 2 0.05 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125156 08/10/2012 Labeler - Genentech, Inc. (080129000) Establishment Name Address ID/FEI Business Operations Roche Singapore Technical Operations Pte. Ltd. 937189173 ANALYSIS(50242-080, 50242-082) , API MANUFACTURE(50242-080, 50242-082) Establishment Name Address ID/FEI Business Operations Genentech, Inc. 833220176 LABEL(50242-080, 50242-082) , PACK(50242-080, 50242-082)