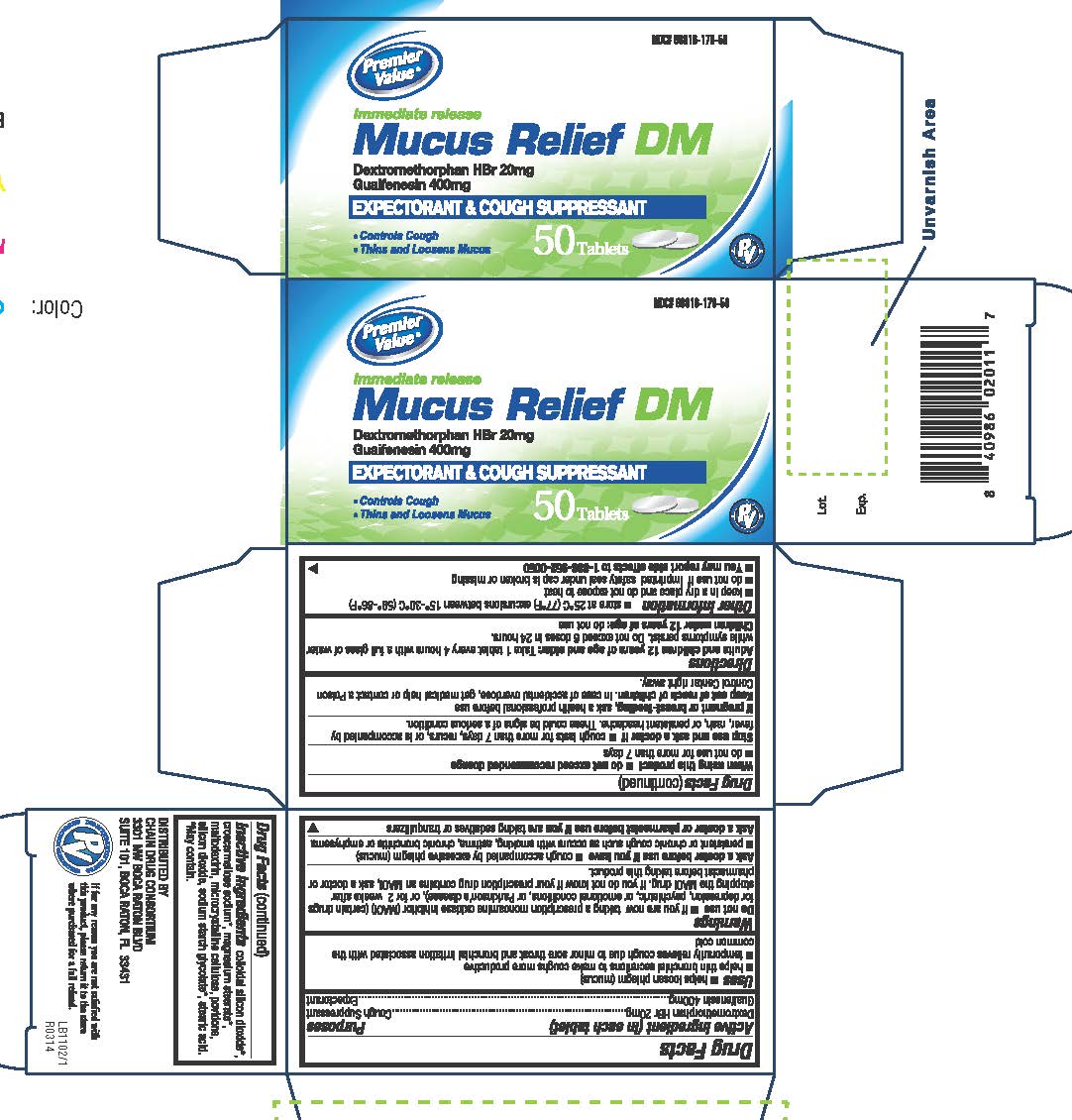
MUCUS RELIEF DM- dextromehtorphan hbr / guaifenesin tablet
Chain Drug Consortium
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Uses
helps loosen phlegm (mucus)
helps thin bronchial secretions to make coughs more productive
temporarily relieves cough due to minor sore throat and bronchial irritation associated with the common cold
Warnings
Do Not Use
■ if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Keep Out of Reach of Children
In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 12 years of age and older: Take 1 tablet every 4 hours with a full glass of water while symptoms persist. Do not exceed 6 doses in 24 hours.
Children under 12 years of age: do not use
| MUCUS RELIEF
DM
dextromehtorphan hbr / guaifenesin tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Chain Drug Consortium (101668460) |
| Registrant - Chain Drug Consortium (101668460) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| A&Z Pharmaceutical, Inc. | 926820705 | manufacture(68016-179) , pack(68016-179) , label(68016-179) , relabel(68016-179) , repack(68016-179) | |