CLEARASIL DAILY CLEAR VANISHING ACNE TREATMENT- benzoyl peroxide cream
RB Health (US) LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Clearasil
®
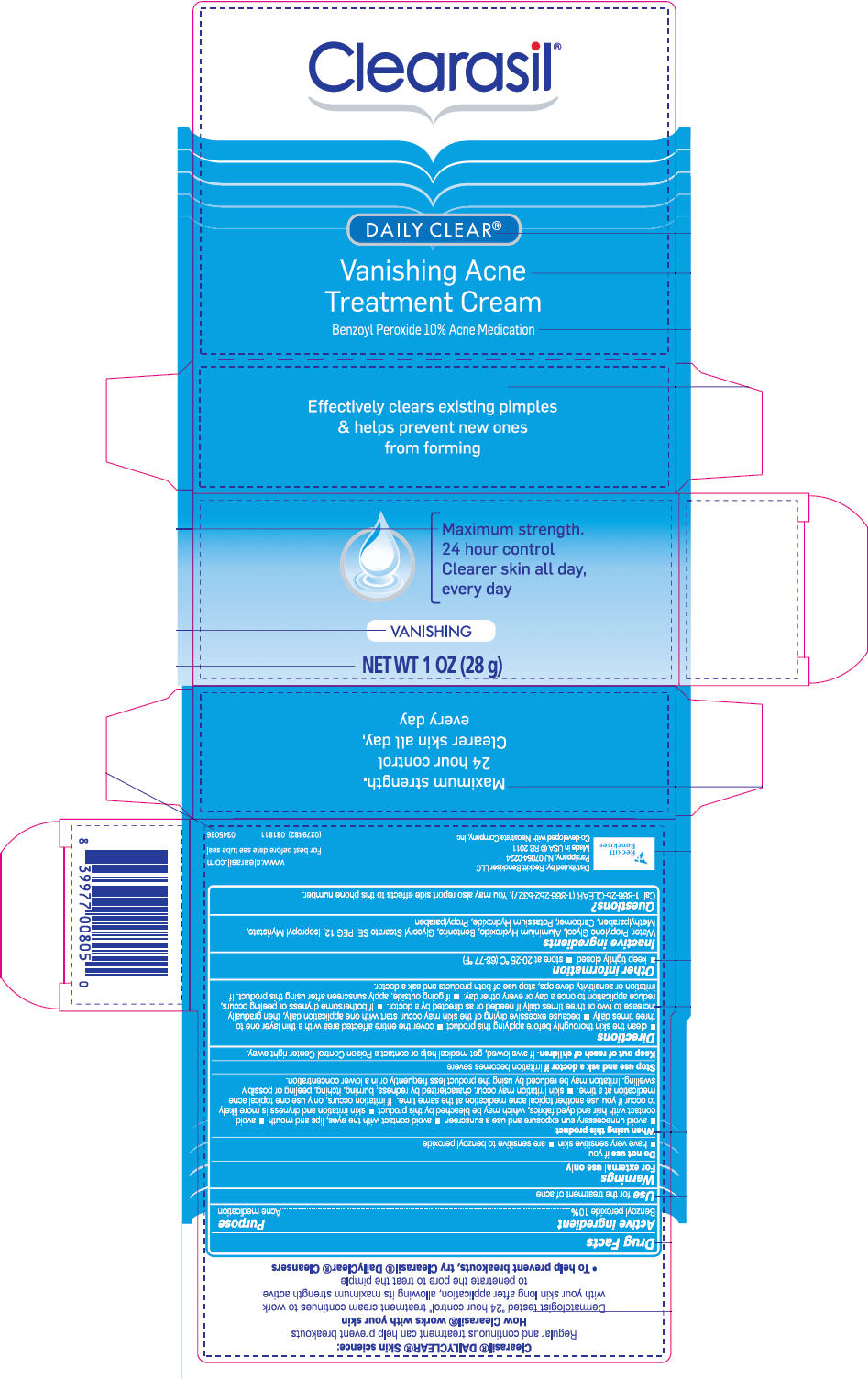
Daily Clear
® Vanishing Acne Treatment Cream
Warnings
For external use only
When using this product
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with the eyes, lips and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
Directions
- clean the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor.
- If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
- if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor.
Inactive ingredients
Water, Propylene Glycol, Aluminium Hydroxide, Bentonite, Glyceryl Stearate SE, PEG-12, Isopropyl Myristate, Methylparaben, Carbomer, Potassium Hydroxide, Propylparaben
| CLEARASIL
DAILY CLEAR VANISHING ACNE TREATMENT
benzoyl peroxide cream |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - RB Health (US) LLC (081049410) |
Revised: 5/2023
Document Id: fbe9caee-e1d1-2f32-e053-6294a90a45b4
Set id: dd074b97-e09b-48a5-bd86-f11934abd897
Version: 3
Effective Time: 20230517
RB Health (US) LLC