HECTOROL- doxercalciferol capsule, liquid filled
Genzyme Corporation
----------
DESCRIPTION
Doxercalciferol, the active ingredient in Hectorol®, is a synthetic vitamin D2 analog that undergoes metabolic activation in vivo to form 1α,25-dihydroxyvitamin D2 (1α,25-(OH)2D2), a naturally occurring, biologically active form of vitamin D2. Hectorol is available as soft gelatin capsules containing 0.5 mcg, 1 mcg or 2.5 mcg doxercalciferol. Each capsule also contains fractionated triglyceride of coconut oil, ethanol, and butylated hydroxyanisole (BHA). The capsule shells contain gelatin, glycerin and titanium dioxide. In addition, the 0.5 mcg capsule shells contain yellow iron oxide and FD&C Red No. 40, the 1 mcg capsule shells contain FD&C Yellow No. 6, and the 2.5 mcg capsule shells contain yellow iron oxide.
Doxercalciferol is a colorless crystalline compound with a calculated molecular weight of 412.66 and a molecular formula of C28H44O2. It is soluble in oils and organic solvents, but is relatively insoluble in water. Chemically, doxercalciferol is (1α,3β,5Z,7E,22E)-9,10-secoergosta-5,7,10(19),22-tetraene-1,3-diol. The structural formula is presented in Figure 1 below:
Figure 1: Chemical Structure of Doxercalciferol
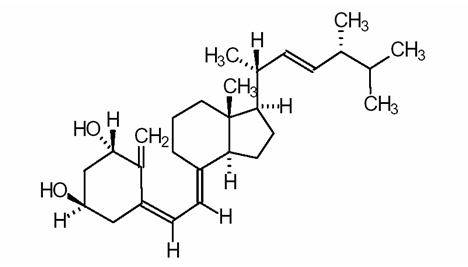
Other names frequently used for doxercalciferol are 1α-hydroxyvitamin D2, 1α-OH-D2, and 1α-hydroxyergocalciferol.
CLINICAL PHARMACOLOGY
Vitamin D levels in humans depend on two sources: (1) exposure to the ultraviolet rays of the sun for conversion of 7-dehydrocholesterol in the skin to vitamin D3 (cholecalciferol) and (2) dietary intake of either vitamin D2 (ergocalciferol) or vitamin D3. Vitamin D2 and vitamin D3 must be metabolically activated in the liver and the kidney before becoming fully active on target tissues. The initial step in the activation process is the introduction of a hydroxyl group in the side chain at C-25 by the hepatic enzyme, CYP 27 (a vitamin D-25-hydroxylase). The products of this reaction are 25-(OH)D2 and 25-(OH)D3, respectively. Further hydroxylation of these metabolites occurs in the mitochondria of kidney tissue, catalyzed by renal 25-hydroxyvitamin D-1-α-hydroxylase to produce 1α,25-(OH)2D2, the primary biologically active form of vitamin D2, and 1α,25-(OH)2D3 (calcitriol), the biologically active form of vitamin D3.
Mechanism of Action
Calcitriol (1α,25-(OH)2D3) and 1α,25-(OH)2D2 regulate blood calcium at levels required for essential body functions. Specifically, the biologically active vitamin D metabolites control the intestinal absorption of dietary calcium, the tubular reabsorption of calcium by the kidney and, in conjunction with parathyroid hormone (PTH), the mobilization of calcium from the skeleton. They act directly on bone cells (osteoblasts) to stimulate skeletal growth, and on the parathyroid glands to suppress PTH synthesis and secretion. These functions are mediated by the interaction of these biologically active metabolites with specific receptor proteins in the various target tissues. In patients with chronic kidney disease (CKD), deficient production of biologically active vitamin D metabolites (due to lack of or insufficient 25-hydroxyvitamin D-1-alpha-hydroxylase activity) leads to secondary hyperparathyroidism, which contributes to the development of metabolic bone disease.
Pharmacokinetics and Metabolism
Doxercalciferol is absorbed from the gastrointestinal tract and activated by CYP 27 in the liver to form 1α,25-(OH)2D2 (major metabolite) and 1α,24-dihydroxyvitamin D2 (minor metabolite). Activation of doxercalciferol does not require the involvement of the kidneys.
In healthy volunteers, peak blood levels of 1α,25-(OH)2D2, the major metabolite of doxercalciferol, are attained at 11-12 hours after repeated oral doses of 5 to 15 mcg of Hectorol and the mean elimination half-life of 1α,25-(OH)2D2 is approximately 32 to 37 hours with a range of up to 96 hours. The mean elimination half-life in patients with end-stage renal disease (ESRD) on dialysis appears to be similar. Hemodialysis causes a temporary increase in 1α,25-(OH)2D2 mean concentrations, presumably due to volume contraction. 1α,25-(OH)2D2 is not removed from blood during hemodialysis.
Clinical Studies
Dialysis:
The safety and effectiveness of Hectorol were evaluated in two double-blind, placebo-controlled, multicentered clinical studies (Study A and Study B) in a total of 138 patients with chronic kidney disease on hemodialysis (Stage 5 CKD). Patients in Study A were an average age of 52 years (range: 22-75), were 55% male, and were 58% African-American, 31% Caucasian, and 11% Hispanic, and had been on hemodialysis for an average of 53 months. Patients in Study B were an average of 52 years (range: 27-75), were 45% male, and 99% African-American, and 1% Caucasian, and had been on hemodialysis for an average of 56 months. After randomization to two groups, eligible patients underwent an 8-week washout period during which no vitamin D derivatives were administered to either group. Subsequently, all patients received Hectorol in an open-label fashion for 16 weeks followed by a double-blind period of 8 weeks during which patients received either Hectorol or placebo. The initial dose of Hectorol during the open-label phase was 10 micrograms after each dialysis session (3 times weekly) for a total of 30 mcg per week. The dosage of Hectorol was adjusted as necessary by the investigator in an attempt to achieve intact parathyroid hormone (iPTH) levels within a targeted range of 150 to 300 pg/mL. The maximum dosage was limited to 20 mcg after each dialysis session (60 mcg/week). If at any time during the trial iPTH fell below 150 pg/mL, Hectorol was immediately suspended and restarted at a lower dosage the following week.
Results:
One hundred and six of the 138 patients who were treated with Hectorol during the 16-week open-label phase achieved iPTH levels ≤ 300 pg/mL. Ninety-four of these patients exhibited plasma iPTH levels ≤ 300 pg/mL on at least 3 occasions. Eighty-seven patients had plasma iPTH levels < 150 pg/mL on at least one occasion during the open-label phase of study participation.
Mean weekly doses during the 16-week open-label period in Study A ranged from 14.8 mcg to 28.7 mcg. In Study B, the mean weekly doses during the 16-week open-label period ranged from 19.2 mcg to 28 mcg.
Decreases in plasma iPTH from baseline values were calculated using as baseline the average of the last 3 values obtained during the 8-week washout phase and are displayed in Table 1below.
| iPTH (pg/mL)
means ± s.d.(n*) p Value v. Baseline p Value v. Placebo |
|||
|---|---|---|---|
| Hectorol® | Placebo | ||
|
|||
| Study A |
Baseline |
797.2 ± 443.8 (30) |
847.1 ± 765.5 (32) |
|
Week 16 |
384.3 ± 397.8 (24) |
526.5 ± 872.2 (29) |
|
|
Week 24 |
404.4 ± 262.9 (21) |
672.6 ± 356.9 (24) |
|
| Study B | Baseline |
973.9 ± 567.0 (41) |
990.4 ± 488.3 (35) |
|
Week 16 |
476.1 ± 444.5 (37) |
485.9 ± 443.4 (32) |
|
|
Week 24 |
459.8 ± 443.0 (35) |
871.9 ± 623.6 (30) |
|
In both studies, iPTH levels increased progressively and significantly in 65.9% of the patients during the 8-week washout (control) period during which no vitamin D derivatives were administered. In contrast, Hectorol treatment resulted in a statistically significant reduction from baseline in mean iPTH levels during the 16-week open-label treatment period in more than 93.5% of the 138 treated patients. During the double-blind period (weeks 17 to 24), the reduction in mean iPTH levels was maintained in the Hectorol treatment group compared to a return to near baseline in the placebo group.
In the clinical trials, the values for iPTH varied widely from patient to patient and from week to week for individual patients. Table 2shows the numbers of patients within each group who achieved and maintained iPTH levels below 300 pg/mL during the open-label and double-blind phases. Seventy-four of 138 patients (53.6%) had plasma iPTH levels within the target range (150-300 pg/mL) during Weeks 14-16.
| 1 | 2 | ≥3 | |||||
|---|---|---|---|---|---|---|---|
| Hectorol® | Placebo | Hectorol® | Placebo | Hectorol® | Placebo | ||
|
Study A |
Weeks 1-16 |
2/30 |
2/32 |
0/30 |
0/32 |
22/30 |
23/32 |
|
Weeks 17-24 |
0/24 |
9/29 |
3/24 |
1/29 |
17/24 |
5/29 |
|
|
Study B |
Weeks 1-16 |
2/41 |
4/35 |
1/41 |
0/35 |
29/41 |
21/35 |
|
Weeks 17-24 |
2/37 |
6/32 |
1/37 |
4/32 |
26/37 |
4/32 |
|
During the 8-week double-blind phase, more patients achieved and maintained the target range of values for iPTH with Hectorol than with placebo.
Pre-dialysis:
The safety and effectiveness of Hectorol were evaluated in two clinical studies in 55 patients with Stage 3 or Stage 4 chronic kidney disease. Eighty-two percent of the patients were male, the average age was 64.6 years, 51% were Caucasian, 40% African-American, and the average serum iPTH level at baseline was 194.6 pg/mL. While levels of 25-(OH) vitamin D were not evaluated at baseline, retrospective assessments of stored serum revealed that the mean ± SD serum 25-(OH) vitamin D was 18.5 ± 8.1 ng/mL (range: < 5 to 54 ng/mL) in the study population.
After randomization to two groups, eligible patients underwent an 8-week washout period during which no vitamin D derivatives were administered to either group. Subsequently, one group received Hectorol and the other placebo during a double-blind period of 24 weeks. The initial dose of Hectorol was 1 mcg per day. The dosage of Hectorol was adjusted as necessary by the investigator in order to reduce intact parathyroid hormone (iPTH) levels to a target of ≥ 30% below post-washout baseline. The maximum dosage was limited to 3.5 mcg per day. If at any time during the trial iPTH fell below 15 pg/mL, Hectorol was immediately suspended and restarted at a lower dosage the following week.
Results:
Decreases in the mean plasma iPTH from baseline values were calculated using as baseline the average of the last 2 values obtained during the 8-week washout phase. In analyses of pooled data from the two studies, iPTH levels decreased from baseline by an average of 101.4 pg/mL in the Hectorol group and by 4.4 pg/mL in the placebo group (p<0.001). Greater reductions of iPTH with Hectorol compared to placebo were observed in each study. Twenty (74%) of 27 subjects in the Hectorol group achieved mean plasma iPTH suppression of ≥ 30% from baseline for the last four weeks of treatment, whereas two (7%) of the 28 subjects treated with placebo achieved this level of iPTH suppression. In the Hectorol-treated patients, the reductions in plasma iPTH were associated with a reduction in serum bone-specific alkaline phosphatase.
INDICATIONS AND USAGE
CONTRAINDICATIONS
Hectorol should not be given to patients with a tendency towards hypercalcemia or current evidence of vitamin D toxicity.
WARNINGS
Overdosage of any form of vitamin D, including Hectorol, is dangerous (see OVERDOSAGE). Progressive hypercalcemia due to overdosage of vitamin D and its metabolites may be so severe as to require emergency attention. Acute hypercalcemia may exacerbate tendencies for cardiac arrhythmias and seizures and may potentiate the action of digitalis drugs. Chronic hypercalcemia can lead to generalized vascular calcification and other soft-tissue calcification. The serum calcium times serum phosphorus (Ca X P) product should be maintained at < 55 mg2/dL2 in patients with chronic kidney disease. Radiographic evaluation of suspect anatomical regions may be useful in the early detection of this condition.
Since doxercalciferol is a precursor for 1α,25-(OH)2D2, a potent metabolite of vitamin D2, pharmacologic doses of vitamin D and its derivatives should be withheld during Hectorol treatment to avoid possible additive effects and hypercalcemia.
Oral calcium-based or other non-aluminum-containing phosphate binders and a low phosphate diet should be used to control serum phosphorus levels in patients with chronic kidney disease. Uncontrolled serum phosphorus exacerbates secondary hyperparathyroidism and can lessen the effectiveness of Hectorol in reducing blood PTH levels. If hypercalcemia occurs after initiating Hectorol therapy, the dose of Hectorol and/or calcium-containing phosphate binders should be decreased. If hyperphosphatemia occurs after initiating Hectorol, the dose of Hectorol should be decreased and/or the dose of phosphate binders increased. (See dosing recommendations for Hectorol under DOSAGE AND ADMINISTRATION section.)
Magnesium-containing antacids and Hectorol should not be used concomitantly in patients on chronic renal dialysis because such use may lead to the development of hypermagnesemia.
PRECAUTIONS
General
Active vitamin D sterols should not be used as initial treatment of nutritional vitamin D deficiency (as defined by low 25-hydroxy vitamin D). Patients should be checked and treated for nutritional vitamin D deficiency prior to initiating treatment with Hectorol.
The principal adverse effects of treatment with Hectorol are hypercalcemia, hyperphosphatemia, hypercalciuria, and oversuppression of PTH (iPTH less than 150 pg/mL). Prolonged hypercalcemia can lead to calcification of soft tissues, including the heart and arteries, and hyperphosphatemia can exacerbate hyperparathyroidism. Hypercalciuria can accelerate the onset of renal failure through nephrocalcinosis. Oversuppression of PTH may lead to adynamic bone syndrome. All of these potential adverse effects should be managed by regular patient monitoring and appropriate dosage adjustments. During treatment with Hectorol, patients usually require dose titration, as well as adjustment in co-therapy (i.e., dietary phosphate binders) in order to maximize PTH suppression while maintaining serum calcium and phosphorus within prescribed ranges.
Dialysis:
In four adequate and well-controlled studies, the incidence of hypercalcemia and hyperphosphatemia increased during therapy with Hectorol. The observed increases during Hectorol treatment, although occurring at a low rate, underscore the importance of regular safety monitoring of serum calcium and phosphorus levels throughout treatment. Patients with higher pre-treatment serum levels of calcium (> 10.5 mg/dL) or phosphorus (> 6.9 mg/dL) were more likely to experience hypercalcemia or hyperphosphatemia. Therefore, Hectorol should not be given to patients with a recent history of hypercalcemia or hyperphosphatemia, or evidence of vitamin D toxicity.
Pre-dialysis:
In two clinical studies, the incidences of hypercalcemia and hyperphosphatemia during therapy with Hectorol were similar to placebo therapy, and no episodes of hypercalciuria were observed. The baseline median 25-(OH) vitamin D levels of patients enrolled in these studies was 17.2 ng/mL. Ninety-three percent of patients had 25-(OH) vitamin D levels less than 30 ng/mL; 26% had 25-(OH) vitamin D levels ≥ 20 to < 30 ng/mL; 58% had levels > 10 to < 20 ng/mL; 7% had levels > 5 to < 10 ng/mL; and 2% had levels < 5 ng/mL. The incidences of hypercalcemia, hyperphosphatemia, and hypercalciuria in patients treated with Hectorol for hyperparathyroidism related to pre-dialysis renal insufficiency has not been fully studied when 25-(OH) vitamin D levels are greater than or equal to 30 ng/mL.
Information for the Patient
The patient, spouse, or guardian should be informed about compliance with dosage instructions, adherence to instructions about diet, calcium supplementation, and avoidance of the use of nonprescription drugs without prior approval from their physician. Patients should also be carefully informed about the symptoms of hypercalcemia (see ADVERSE REACTIONS section).
Patients' total combined elemental calcium intake (dietary and phosphate binder) should not exceed 2 g daily.
Laboratory Tests
Serum or plasma iPTH and serum calcium, phosphorus, and alkaline phosphatase should be determined periodically. In the early phase of treatment for dialysis patients, iPTH, serum calcium, and serum phosphorus should be determined prior to initiation of Hectorol treatment and weekly thereafter. For pre-dialysis patients, serum levels of calcium and phosphorus and plasma levels of iPTH should be monitored at least every two weeks for 3 months after initiation of Hectorol therapy or following dose-adjustments in Hectorol therapy, then monthly for 3 months, and every 3 months thereafter.
Drug Interactions
Specific drug interaction studies have not been conducted. Cholestyramine has been reported to reduce intestinal absorption of fat-soluble vitamins; therefore, it may impair intestinal absorption of doxercalciferol. Magnesium-containing antacids and Hectorol should not be used concomitantly because such use may lead to the development of hypermagnesemia (see WARNINGS). The use of mineral oil or other substances that may affect absorption of fat may influence the absorption and availability of Hectorol. Although not examined specifically, enzyme inducers (such as glutethimide and phenobarbital) may affect the 25-hydroxylation of Hectorol and may necessitate dosage adjustments. Cytochrome P450 inhibitors (such as ketoconazole and erythromycin) may inhibit the 25-hydroxylation of Hectorol. Hence, formation of the active Hectorol moiety may be hindered.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week carcinogenicity study in rats, there was an increased incidence of benign and malignant adrenal pheochromocytomas in both males and females at oral doses of 0.04, 0.13 and 0.39 mcg/kg/day (≤ 1 times the human exposure in pre-dialysis patients with a maximum recommended dose of 3.5 mcg/day or 24.5 mcg/week). This increased incidence of pheochromocytomas in rats may be due to altered calcium homeostasis by doxercalciferol. No evidence of genetic toxicity was observed in anin vitro bacterial mutagenicity assay (Ames test) or a mouse lymphoma gene mutation assay. Doxercalciferol caused structural chromatid and chromosome aberrations in an in vitro human lymphocyte clastogenicity assay with metabolic activation. However, doxercalciferol was negative in an in vivo mouse micronucleus clastogenicity assay. Doxercalciferol had no effect on male or female fertility in rats at oral doses up to 2.5 mcg/kg/day (approximately 3 times the maximum recommended human dose of 60 mcg/week based on mcg/m2 body surface area).
Use in Pregnancy
Pregnancy Category B
Reproduction studies in rats and rabbits, at doses up to 20 mcg/kg/day and 0.1 mcg/kg/day (approximately 25 times and less than the maximum recommended human dose of 60 mcg/week based on mcg/m2 body surface area, respectively) have revealed no teratogenic or fetotoxic effects due to doxercalciferol. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
It is not known whether doxercalciferol is excreted in human milk. Because other vitamin D derivatives are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from doxercalciferol, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
ADVERSE REACTIONS
Dialysis:
Hectorol has been evaluated for safety in clinical studies in 165 patients with chronic kidney disease on hemodialysis. In two placebo-controlled, double-blind, multicenter studies, discontinuation of therapy due to any adverse event occurred in 2.9% of 138 patients treated with Hectorol for four to six months (dosage titrated to achieve target iPTH levels, see CLINICAL PHARMACOLOGY/Clinical Studies) and in 3.3% of 61 patients treated with placebo for two months. Adverse events occurring in the Hectorol group at a frequency of 2% or greater and more frequently than in the placebo group are presented in Table 3 below:
| Adverse Event | Hectorol® (n=61) % | Placebo (n=61) % |
|---|---|---|
| Body as a Whole | ||
| Abscess | 3.3 | 0.0 |
| Headache | 27.9 | 18.0 |
| Malaise | 27.9 | 19.7 |
| Cardiovascular System | ||
| Bradycardia | 6.6 | 4.9 |
| Digestive System | ||
| Anorexia | 4.9 | 3.3 |
| Constipation | 3.3 | 3.3 |
| Dyspepsia | 4.9 | 1.6 |
| Nausea/Vomiting | 21.3 | 19.7 |
| Musculoskeletal System | ||
| Arthralgia | 4.9 | 0.0 |
| Metabolic and Nutritional | ||
| Edema | 34.4 | 21.3 |
| Weight increase | 4.9 | 0.0 |
| Nervous System | ||
| Dizziness | 11.5 | 9.8 |
| Sleep disorder | 3.3 | 0.0 |
| Respiratory System | ||
| Dyspnea | 11.5 | 6.6 |
| Skin | ||
| Pruritus | 8.2 | 6.6 |
| A patient who reported the same medical term more than once was counted only once for that medical term. | ||
Pre-dialysis:
Hectorol has been evaluated for safety in clinical studies in 55 patients (27 active and 28 placebo) with chronic kidney disease, Stages 3 or 4. In two placebo-controlled, double-blind, multicenter studies, discontinuation of therapy due to any adverse event occurred in one (3.7%) of 27 patients treated with Hectorol for 24 weeks (dosage titrated to achieve target iPTH levels, see CLINICAL PHARMACOLOGY/Clinical Studies) and in three (10.7%) of 28 patients treated with placebo for 24 weeks. Adverse events occurring in the Hectorol group at a frequency of 5% or greater and more frequently than in the placebo group are as follows: Body as a Whole – Infection, Chest Pain; Digestive System – Constipation, Dyspepsia; Hematologic and Lymphatic– Anemia; Metabolic and Nutritional– Dehydration; Nervous System – Depression, Hypertonia, Insomnia, Paresthesia; Respiratory System – Cough increased, Dyspnea, Rhinitis.
Potential adverse effects of Hectorol are, in general, similar to those encountered with excessive vitamin D intake. The early and late signs and symptoms of vitamin D intoxication associated with hypercalcemia include:
Early
Weakness, headache, somnolence, nausea, vomiting, dry mouth, constipation, muscle pain, bone pain, metallic taste, and anorexia.
Late
Polyuria, polydipsia, anorexia, weight loss, nocturia, conjunctivitis (calcific), pancreatitis, photophobia, rhinorrhea, pruritus, hyperthermia, decreased libido, elevated blood urea nitrogen (BUN), albuminuria, hypercholesterolemia, elevated serum aspartate transaminase (AST) and alanine transaminase (ALT), ectopic calcification, hypertension, cardiac arrhythmias, sensory disturbances, dehydration, apathy, arrested growth, urinary tract infections, and, rarely, overt psychosis.
OVERDOSAGE
Administration of Hectorol to patients in excess doses can cause hypercalcemia, hypercalciuria, hyperphosphatemia, and oversuppression of PTH secretion leading in certain cases to adynamic bone disease. High intake of calcium and phosphate concomitant with Hectorol may lead to similar abnormalities. High levels of calcium in the dialysate bath may contribute to hypercalcemia.
Treatment of Hypercalcemia and Overdosage
General treatment of hypercalcemia (greater than 1 mg/dL above the upper limit of the normal range in dialysis patients; > 10.7 mg/dL in pre-dialysis patients) consists of immediate suspension of Hectorol therapy, institution of a low calcium diet, and withdrawal of calcium supplements. Serum calcium levels should be determined at least weekly until normocalcemia ensues. Hypercalcemia usually resolves in 2 to 7 days. When serum calcium levels have returned to within normal limits, Hectorol therapy may be reinstituted at a dose that is lower (at least 2.5 mcg in dialysis patients and 0.5 mcg in pre-dialysis patients) than prior therapy. In dialysis patients, serum calcium levels should be obtained weekly after all dosage changes and during subsequent dosage titration. Persistent or markedly elevated serum calcium levels may be corrected by dialysis against a reduced calcium or calcium-free dialysate.
Treatment of Accidental Overdosage of Doxercalciferol
The treatment of acute accidental overdosage of Hectorol should consist of general supportive measures. If drug ingestion is discovered within a relatively short time (10 minutes), induction of emesis or gastric lavage may be of benefit in preventing further absorption. If drug ingestion is discovered later than 10 minutes post-ingestion, the administration of mineral oil may promote its fecal elimination. Serial serum electrolyte determinations (especially calcium), rate of urinary calcium excretion, and assessment of electrocardiographic abnormalities due to hypercalcemia should be obtained. Such monitoring is critical in patients receiving digitalis. Discontinuation of supplemental calcium and institution of a low calcium diet are also indicated in accidental overdosage. If persistent and markedly elevated serum calcium levels occur, treatment with standard medical care should be followed, as needed. Based on similarities between Hectorol and its active metabolite, 1α,25-(OH)2D2, it is expected that Hectorol is not removed from the blood by dialysis.
DOSAGE AND ADMINISTRATION
Adult Administration:
The optimal dose of Hectorol must be carefully determined for each patient. Table 4provides the current recommended therapeutic target levels for iPTH in patients with chronic kidney disease:
| CKD Stage | GFR (mL/min/1.73m2) | Target "intact" PTH (pg/mL) |
|---|---|---|
|
From Table 15 of National Kidney Foundation, K/DOQI Clinical Practice Guidelines for Bone Metabolism and Disease in Chronic Kidney Disease. Am J Kidney Dis 42:S1-S202, 2003 (suppl 3) |
||
| 3 | 30-59 | 35-70 |
| 4 | 15-29 | 70-110 |
| 5 | < 15 (or dialysis) | 150-300 |
Dialysis:
The recommended initial dose of Hectorol is 10 mcg administered three times weekly at dialysis (approximately every other day). The initial dose should be adjusted, as needed, in order to lower blood iPTH into the range of 150 to 300 pg/mL. The dose may be increased at 8-week intervals by 2.5 mcg if iPTH is not lowered by 50% and fails to reach the target range. The maximum recommended dose of Hectorol is 20 mcg administered three times a week at dialysis for a total of 60 mcg per week. Drug administration should be suspended if iPTH falls below 100 pg/mL and restarted one week later at a dose that is at least 2.5 mcg lower than the last administered dose. During titration, iPTH, serum calcium, and serum phosphorus levels should be obtained weekly. If hypercalcemia, hyperphosphatemia, or a serum calcium times serum phosphorus product greater than 55 mg2/dL2 is noted, the dose of Hectorol should be decreased or suspended and/or the dose of phosphate binders should be appropriately adjusted. If suspended, the drug should be restarted at a dose that is at least 2.5 mcg lower.
Dosing must be individualized and based on iPTH levels with monitoring of serum calcium and serum phosphorus levels. The following is a suggested approach in dose titration:
| Initial Dosing | ||
|---|---|---|
|
iPTH Level |
Hectorol® Dose |
|
|
> 400 pg/mL |
10 mcg three times per week at dialysis |
|
| Dose Titration | ||
|---|---|---|
|
iPTH Level |
Hectorol® Dose |
|
|
Above 300 pg/mL |
Increase by 2.5 mcg at eight-week intervals as necessary |
|
|
150 - 300 pg/mL |
Maintain |
|
|
< 100 pg/mL |
Suspend for one week, then resume at a dose that is at least 2.5 mcg lower |
|
Pre-dialysis:
The recommended initial dose of Hectorol is 1 mcg administered once daily. The initial dose should be adjusted, as needed, in order to lower blood iPTH to within target ranges (see table below). The dose may be increased at 2-week intervals by 0.5 mcg to achieve the target range of iPTH. The maximum recommended dose of Hectorol is 3.5 mcg administered once per day.
Serum levels of calcium and phosphorus and plasma levels of iPTH should be monitored at least every two weeks for 3 months after initiation of Hectorol therapy or following dose adjustments in Hectorol therapy, then monthly for 3 months, and every 3 months thereafter. If hypercalcemia, hyperphosphatemia, or a serum calcium times phosphorus product greater than 55 mg2/dL2 is noted, the dose of Hectorol should be decreased or suspended and/or the dose of phosphate binders should be appropriately adjusted. If suspended, the drug should be restarted at a dose that is at least 0.5 mcg lower.
Dosing must be individualized and based on iPTH levels with monitoring of serum calcium and serum phosphorus levels. Table 6 presents a suggested approach in dose titration:
| Initial Dosing | ||
|---|---|---|
|
iPTH Level |
Hectorol® Dose |
|
|
> 70 pg/mL (Stage 3) |
1 mcg once per day |
|
|
> 110 pg/mL (Stage 4) |
||
| Dose Titration | ||
|---|---|---|
|
iPTH Level |
Hectorol® Dose |
|
|
Above 70 pg/mL (Stage 3) |
Increase by 0.5 mcg at two-week intervals as necessary |
|
|
110 pg/mL (Stage 4) |
||
|
35 - 70 pg/mL (Stage 3) |
Maintain |
|
|
70 - 110 pg/mL (Stage 4) |
||
|
< 35 pg/mL (Stage 3) |
Suspend for one week, then resume at a dose that is at least 0.5 mcg lower |
|
|
< 70 pg/mL (Stage 4) |
||
HOW SUPPLIED
NDC 58468-0120-1
0.5 mcg doxercalciferol in soft gelatin, salmon, oval capsules, imprinted g; foil induction sealed bottles of 50.
NDC 58468-0124-1
1 mcg doxercalciferol in soft gelatin, peach, oval capsules, imprinted g; foil induction sealed bottles of 50.
NDC 58468-0121-1
2.5 mcg doxercalciferol in soft gelatin, butter yellow, oval capsules, imprinted g; foil induction sealed bottles of 50.
Store at 25°C (77°F): excursions permitted to 15-30°C (59-86°F)
[see USP controlled room temperature]
Manufactured by Catalent Pharma Solutions for
Genzyme Corporation, 500 Kendall Street, Cambridge, MA 02142 800-847-0069
HECTOROL and GENZYME are registered trademarks of Genzyme Corporation.
Package Carton - Principal Display Panel - 0.5 mcg Capsules
NDC 58468-0120-1
Hectorol®
(doxercalciferol capsules)
0.5 mcg
50 Capsules
Rx only
| HECTOROL
doxercalciferol capsule, liquid filled |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| HECTOROL
doxercalciferol capsule, liquid filled |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| HECTOROL
doxercalciferol capsule, liquid filled |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Genzyme Corporation (025322157) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Catalent Pharma Solutions LLC | 051762268 | MANUFACTURE(58468-0120, 58468-0121, 58468-0124) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Anderson Packaging, Inc. | 053217022 | LABEL(58468-0120, 58468-0121, 58468-0124) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Assia Chemical Industries, Ltd. | 514678507 | API MANUFACTURE(58468-0120, 58468-0121, 58468-0124) | |