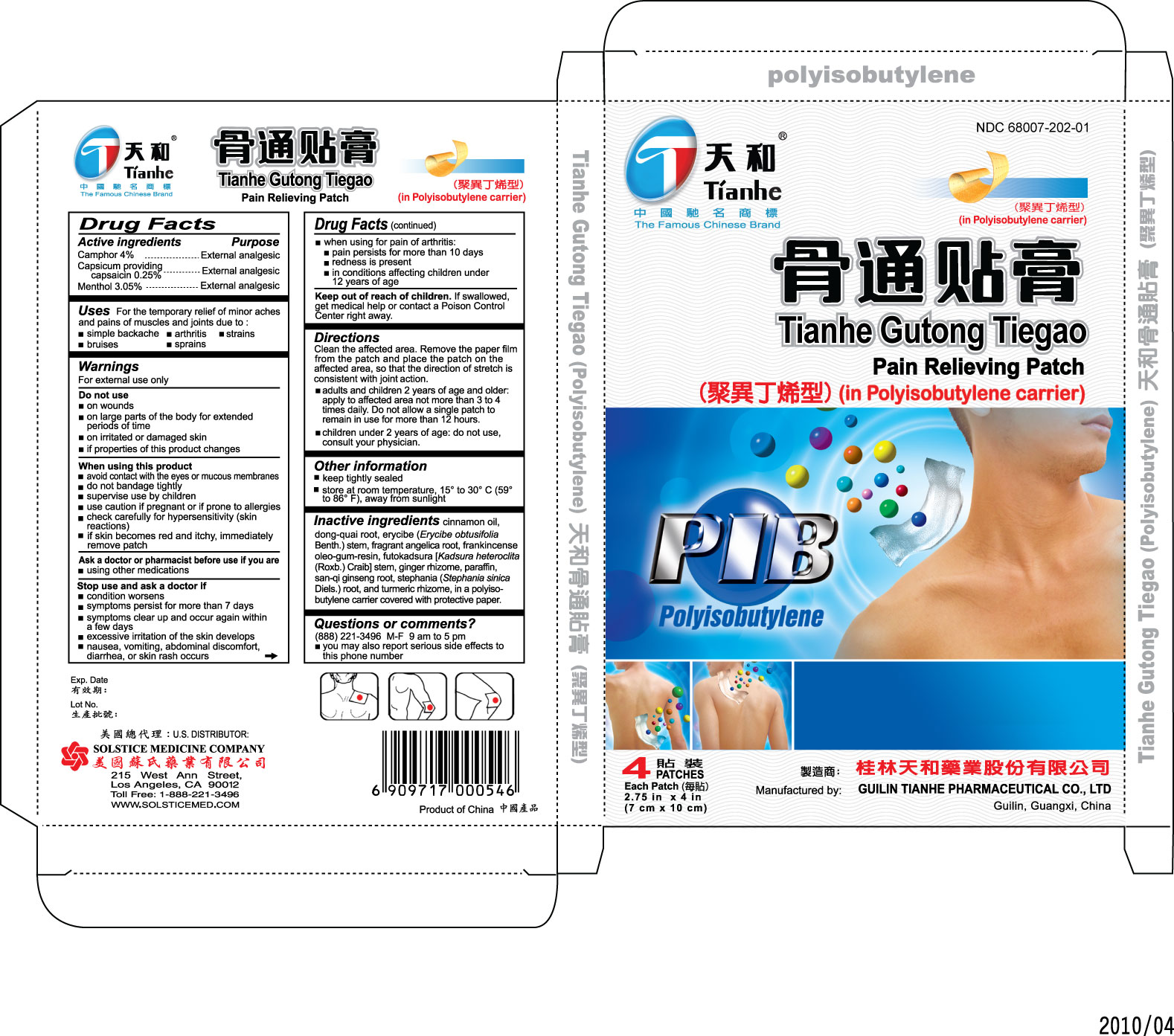
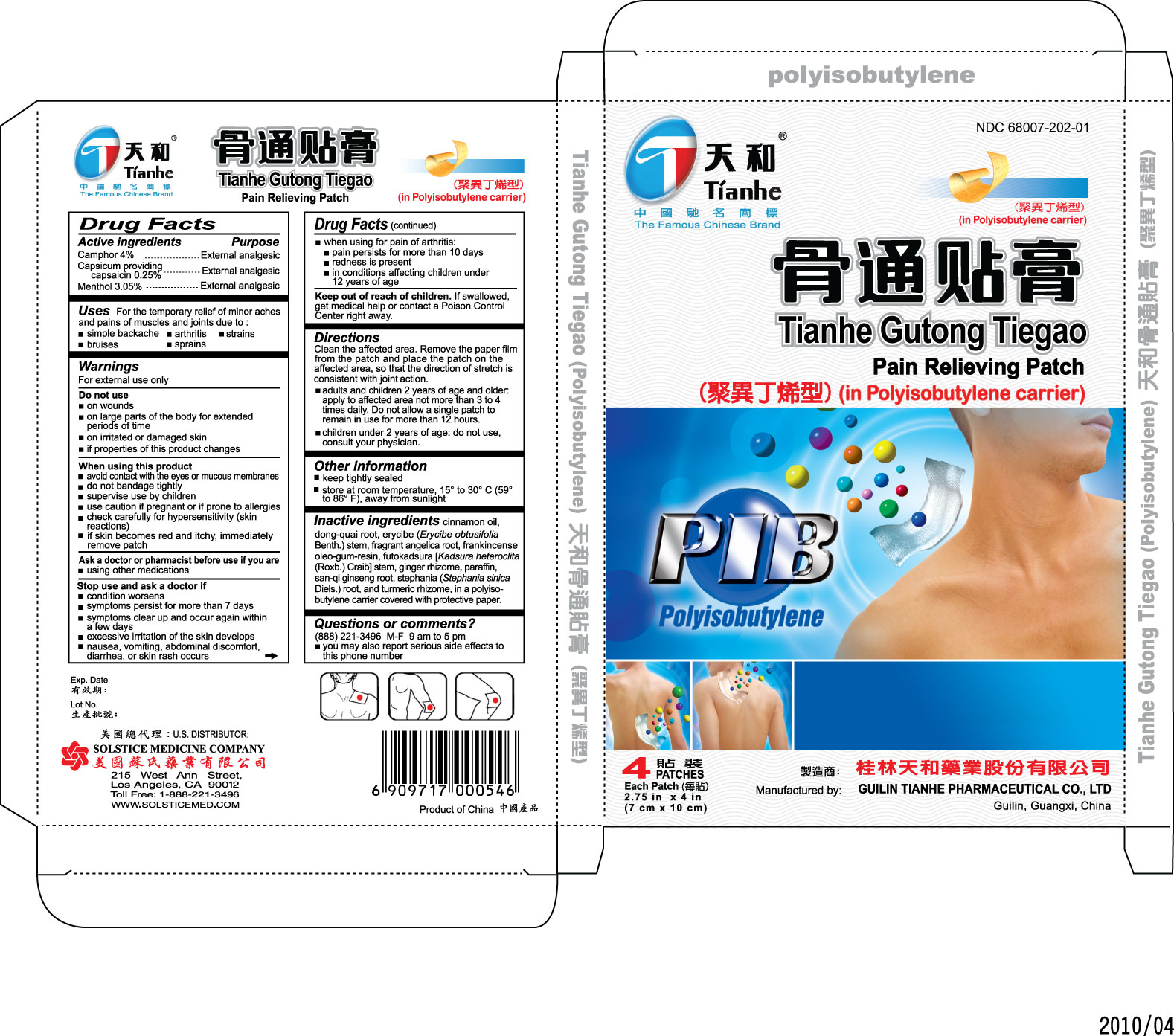
Label: TIANHE GUTONG TIEGAO (IN POLYISOBUTYLENE CARRIER)- external analgesic patch patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 68007-202-01 - Packager: GUILIN TIANHE PHARMACEUTICAL CO LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 24, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- DO NOT USE
- WHEN USING
- ASK DOCTOR/PHARMACIST
-
STOP USE
Stop use and ask a doctor if
- condition worsens
- symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- excessive irritation of the skin develops
- nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
- when using for pain of arthritis:
-
- pain persists for more than 10 days
- redness is present in conditions affecting children under 12 years of age
- pain persists for more than 10 days
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions Clean the affected area. Remove the paper film from the patch and place the patch on the affected area, so that the direction of stretch is consistent with joint action.
- adults and children 2 years of age and older: apply to affected area no more than 3 to 4 times daily. Do not allow a single patch to remain in use for more than 12 hours.
- children under 2 years of age: do not use, consult your physician.
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Inactive ingredients cinnamon oil, dong-quai root, erycibe (Erycibe
obtusifolia Benth.) stem, fragrant angelica root, frankincense oleo-gum-resin,
futokadsura [Kadsura heteroclita (Roxb.) Craib] stem, ginger rhizome, paraffin,
san-qi ginseng root, stephania (Stephania sinica Diels.) root, and turmeric
rhizome, in a polyisobutylene carrier covered with protective paper.
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TIANHE GUTONG TIEGAO (IN POLYISOBUTYLENE CARRIER)
external analgesic patch patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68007-202 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (NATURAL) (UNII: N20HL7Q941) (CAMPHOR (NATURAL) - UNII:N20HL7Q941) CAMPHOR (NATURAL) 4 g CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.25 g MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 3.05 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68007-202-01 4 in 1 BOX Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 05/24/2010 Labeler - GUILIN TIANHE PHARMACEUTICAL CO LTD (528565427) Establishment Name Address ID/FEI Business Operations GUILIN TIANHE PHARMACEUTICAL CO LTD 528565427 manufacture