Label: EQUATE ALLERGY RELIEF- cetirizine hydrochloride tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 49035-312-05, 49035-312-56, 49035-312-57, 49035-312-58 - Packager: Wal-Mart Stores, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 3, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
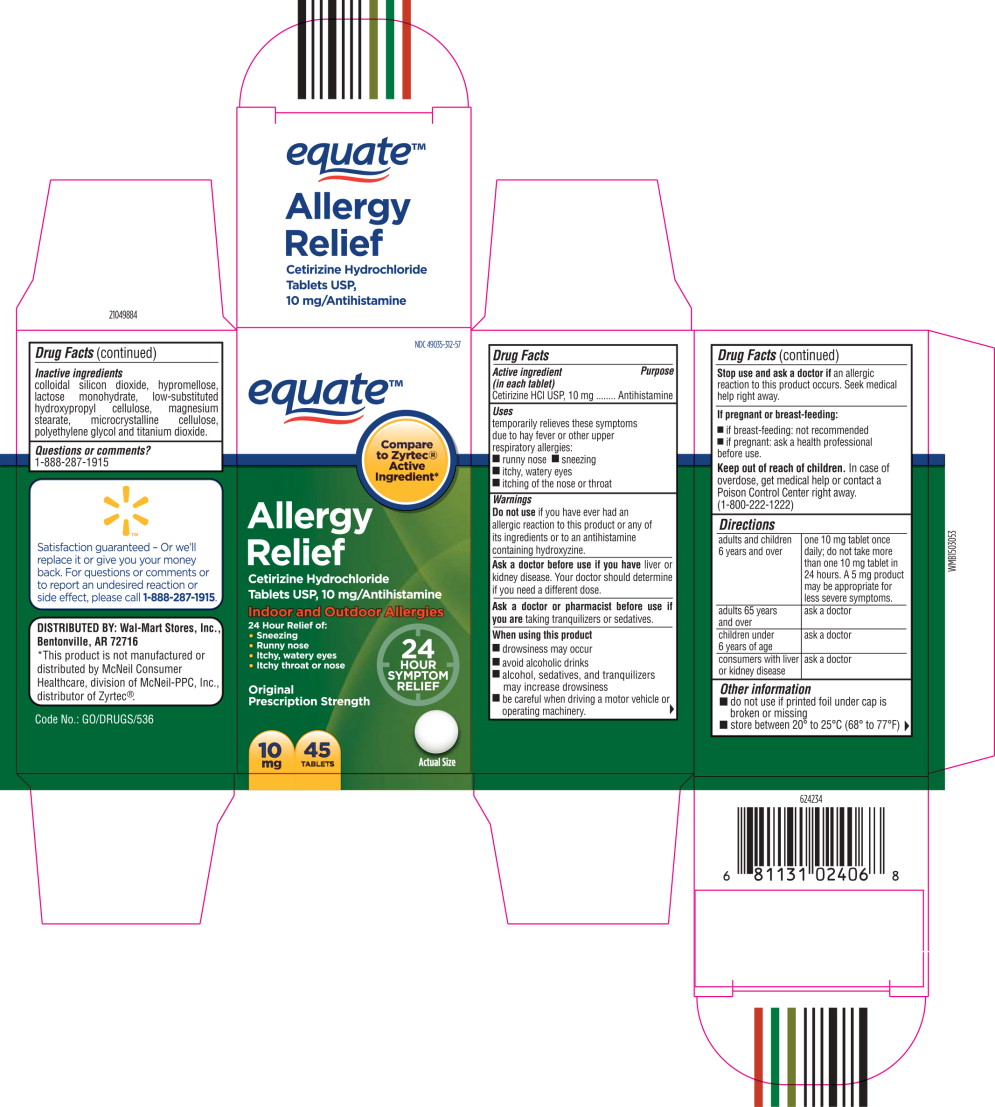
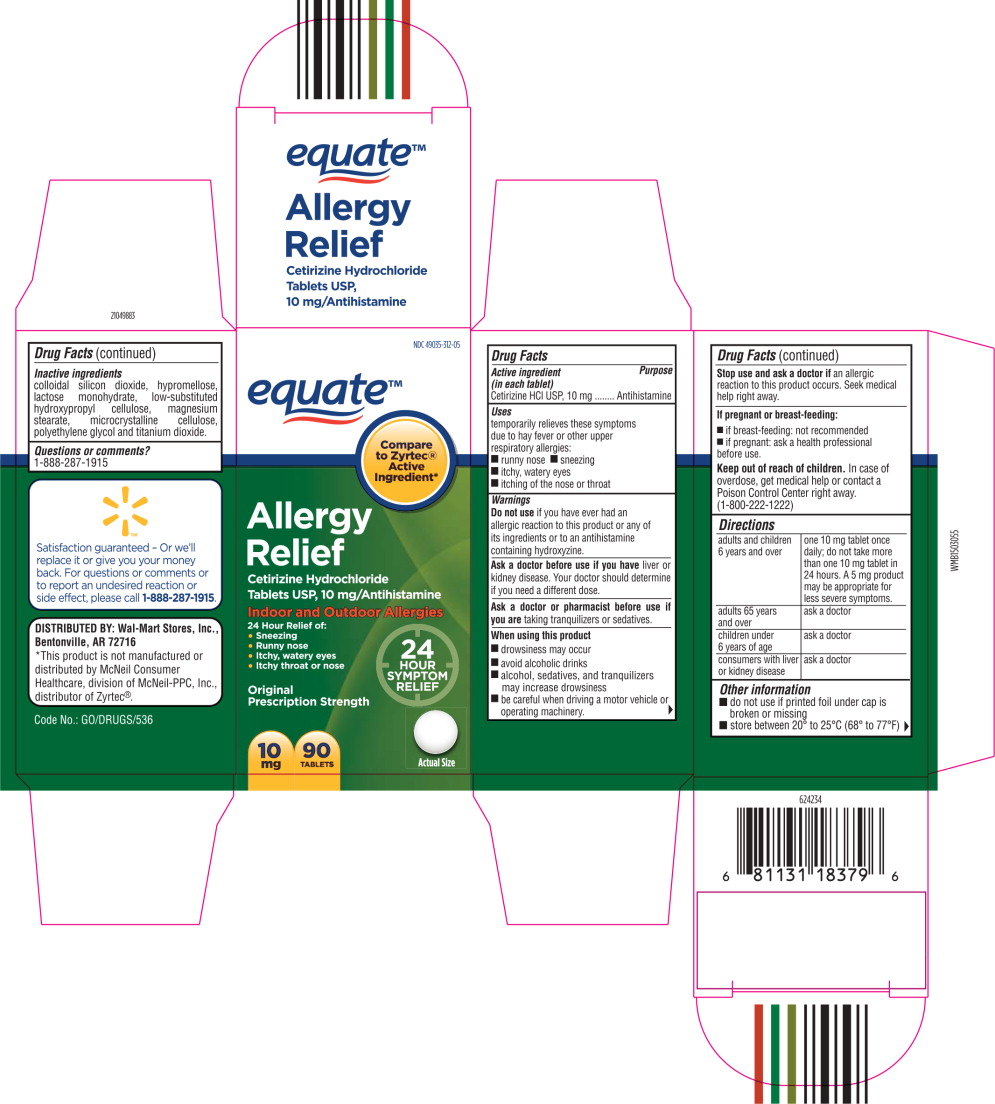
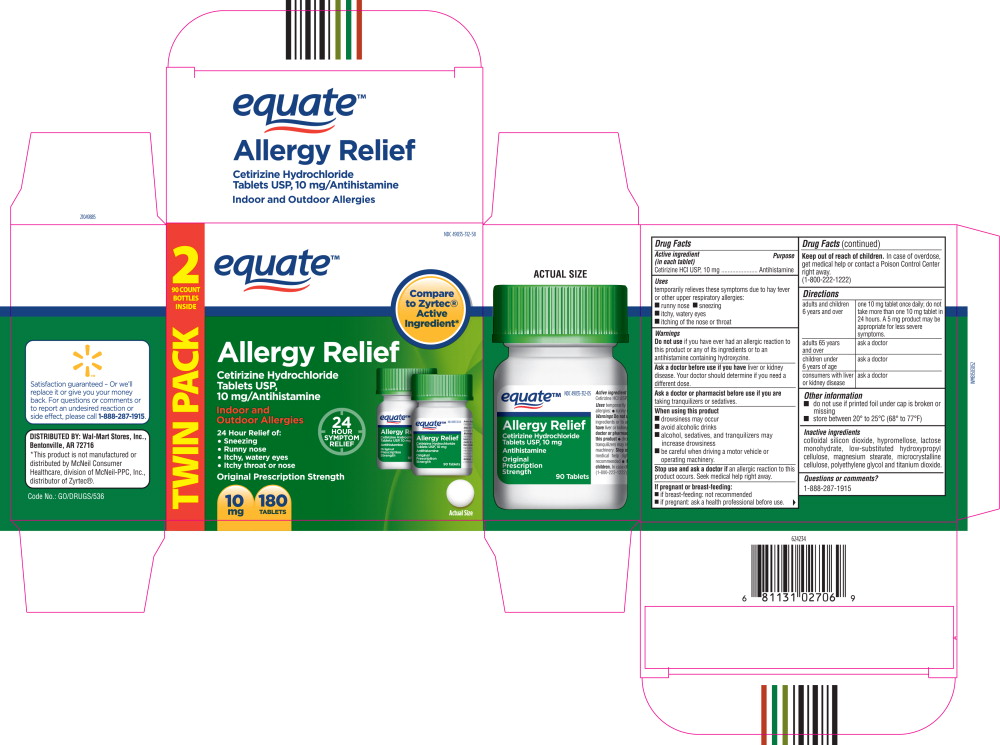
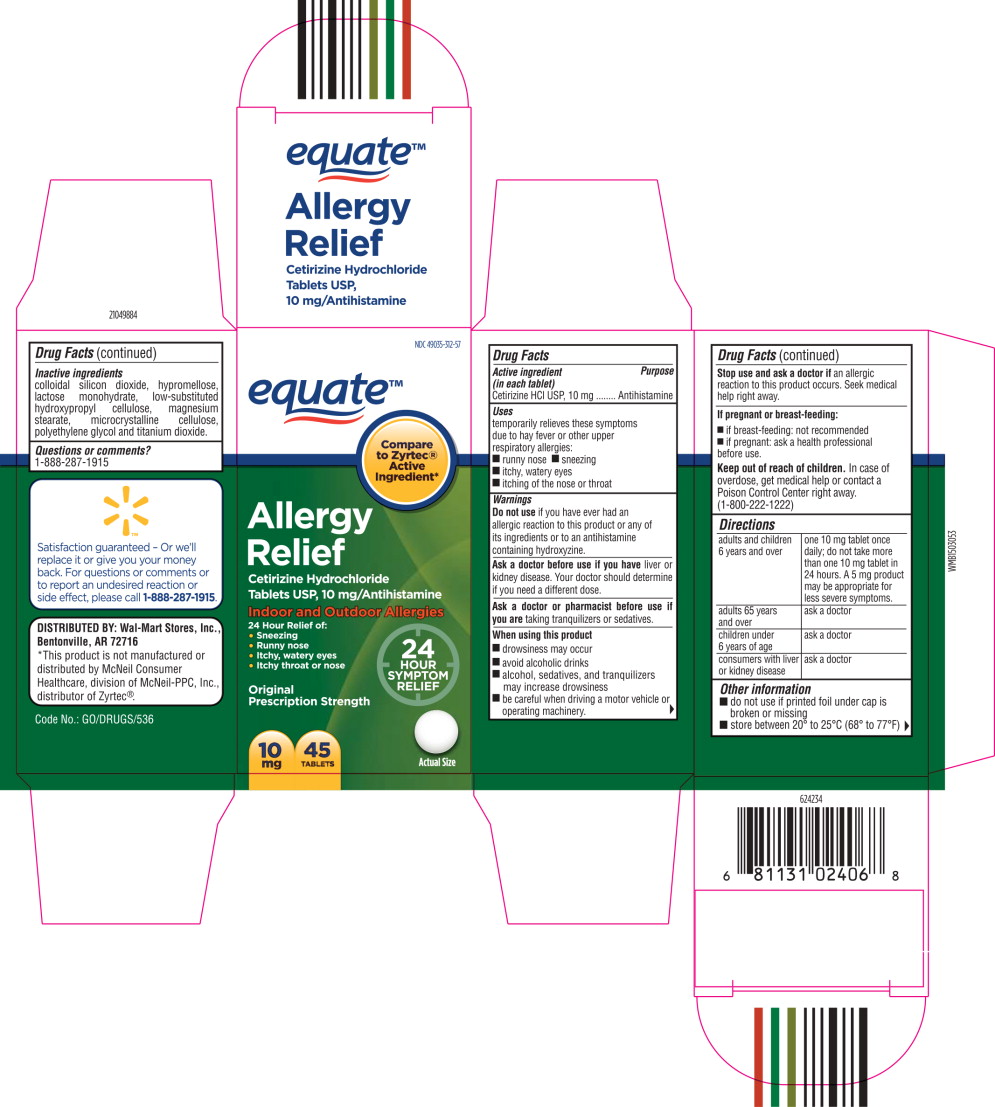
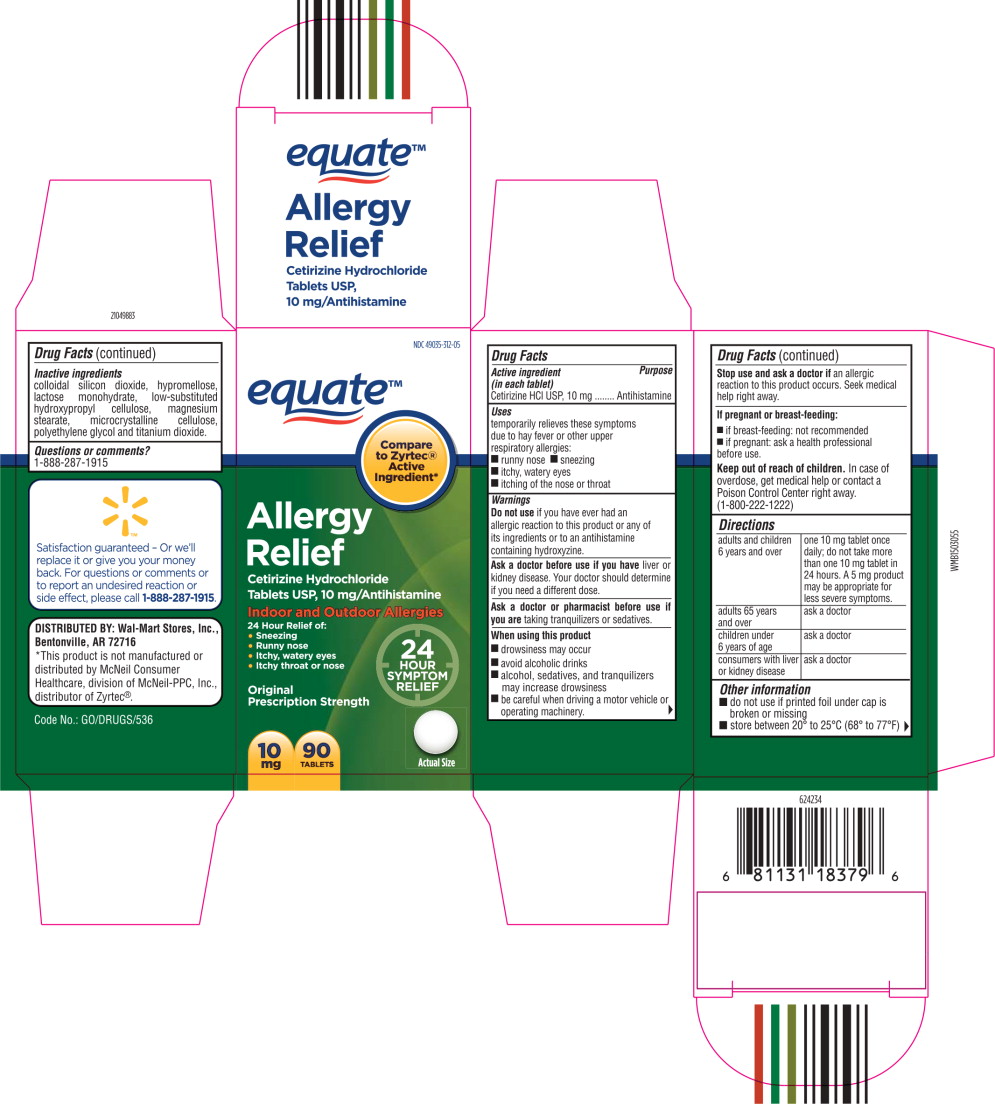
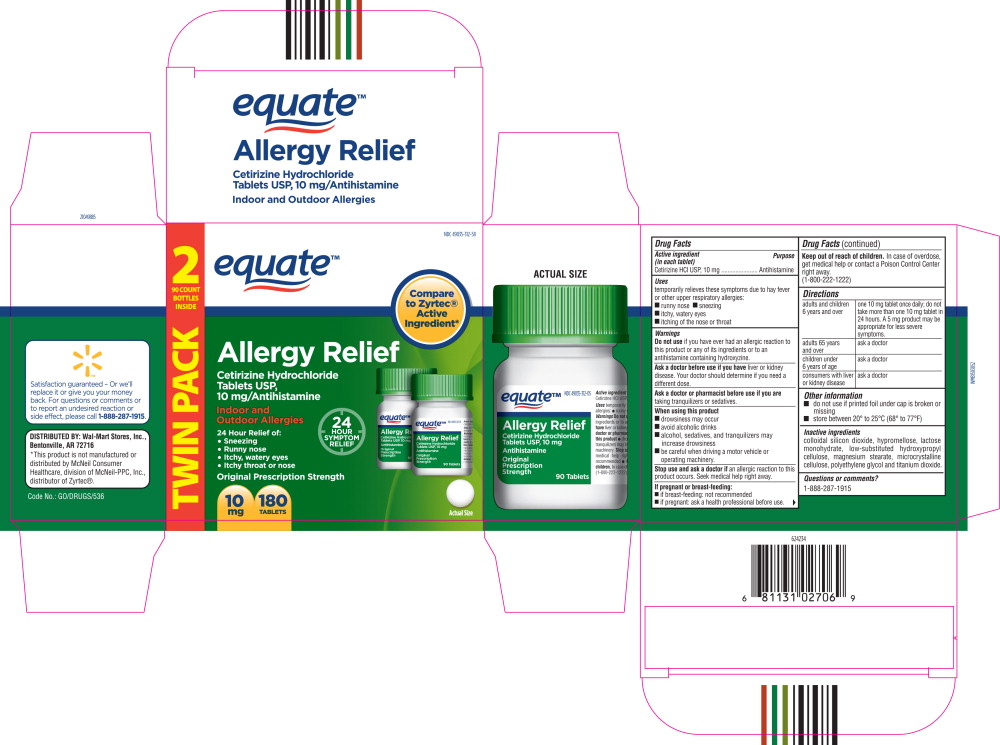
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if
an allergic reaction to this product occurs. Seek medical help right away.
-
Directions
adults and children 6 years and over one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. adults 65 years and over ask a doctor children under 6 years of age ask a doctor consumers with liver or kidney disease ask a doctor - Other information
- Inactive ingredients
- Questions or Comments?
- Principal Display Panel - Carton Label
- Principal Display Panel - Carton Label
- Principal Display Panel - Bottle Label
- Principal Display Panel - Carton Label
- Principal Display Panel - Bottle Label
- Principal Display Panel - Carton Label
-
INGREDIENTS AND APPEARANCE
EQUATE ALLERGY RELIEF
cetirizine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-312 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength cetirizine hydrochloride (UNII: 64O047KTOA) (cetirizine - UNII:YO7261ME24) cetirizine hydrochloride 10 mg Inactive Ingredients Ingredient Name Strength colloidal silicon dioxide (UNII: ETJ7Z6XBU4) hypromellose 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) lactose monohydrate (UNII: EWQ57Q8I5X) hydroxypropyl cellulose, low substituted (UNII: 2165RE0K14) magnesium stearate (UNII: 70097M6I30) cellulose, microcrystalline (UNII: OP1R32D61U) polyethylene glycols (UNII: 3WJQ0SDW1A) titanium dioxide (UNII: 15FIX9V2JP) Product Characteristics Color white (white) Score no score Shape ROUND (ROUND) Size 8mm Flavor Imprint Code C;121 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-312-56 1 in 1 CARTON 1 14 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:49035-312-57 1 in 1 CARTON 2 45 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:49035-312-05 1 in 1 CARTON 3 90 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:49035-312-58 2 in 1 CARTON 4 90 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077318 07/25/2013 Labeler - Wal-Mart Stores, Inc. (051957769) Registrant - Cipla Ltd. (650072015) Establishment Name Address ID/FEI Business Operations Cipla limited 650072015 MANUFACTURE(49035-312) Establishment Name Address ID/FEI Business Operations Cipla limited 916940208 API MANUFACTURE(49035-312)