MORPHINE SULFATE- morphine sulfate injection, solution
Hospira, Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Morphine Sulfate
Injection, USP
DESCRIPTION
Morphine Sulfate Injection, USP is a sterile, nonpyrogenic solution of morphine sulfate in water for injection.
Each mL contains morphine sulfate, pentahydrate 10 mg or 25 mg and sodium metabisulfite 0.6 mg or 1.5 mg respectively, added as an antioxidant. May contain sodium hydroxide and/or hydrochloric acid for pH adjustment. pH 4.5 (2.5 to 6.5).
The solution contains no bacteriostat or antimicrobial agent and is intended as a single-dose injection to provide analgesia via the intravenous route only after dilution in the ADD-Vantage Flexible Diluent Container. It is not for intrathecal or epidural use. Do not use the injection if its color is darker than pale yellow, if it is discolored in any other way or if it contains a precipitate.
Morphine, the most important alkaloid of opium, is classified pharmacologically as a narcotic analgesic.
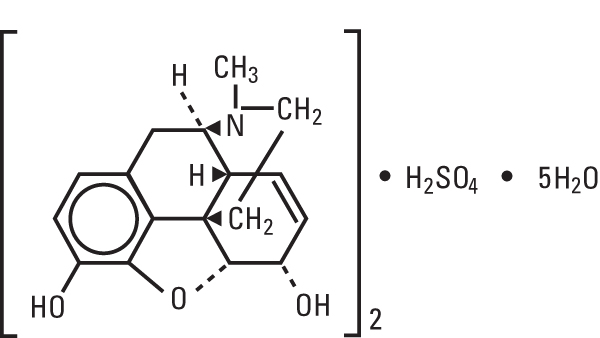
Morphine Sulfate, USP (pentahydrate) is chemically designated 7, 8-didehydro-4, 5α-epoxy-17-methylmorphinan-3, 6α-diol sulfate (2:1) (salt), pentahydrate, a white crystalline powder, soluble in water. It has the following structural formula:
CLINICAL PHARMACOLOGY
Morphine exerts its primary effects on the central nervous system and organs containing smooth muscle. Pharmacologic effects include analgesia, drowsiness, alteration in mood (euphoria), reduction in body temperature (at low doses), dose-related depression of respiration, interference with adrenocortical response to stress (at high doses), reduction in peripheral resistance with little or no effect on cardiac index and miosis.
Morphine, as other opioids, acts as an agonist interacting with stereospecific and saturable binding sites/receptors in the brain, spinal cord and other tissues. These sites have been classified as µ receptors and are widely distributed throughout the central nervous system being present in highest concentration in the limbic system (frontal and temporal cortex, amygdala and hippocampus), thalamus, striatum, hypothalamus, midbrain and laminae I, II, IV and V of the dorsal horn in the spinal cord. It has been postulated that exogenously administered morphine exerts its analgesic effect, in part, by altering the central release of neurotransmitter from afferent nerves sensitive to noxious stimuli. Peripheral threshold or responsiveness to noxious stimuli is unaffected leaving monosynaptic reflexes such as the patellar or the Achilles tendon reflex intact.
Central nervous system effects of intravenously administered morphine sulfate are influenced by ability to cross the blood-brain barrier.
Nausea and vomiting may be prominent and are thought to be the result of central stimulation of the chemoreceptor trigger zone. Histamine release is common; allergic manifestations of urticaria and, rarely, anaphylaxis may occur. Bronchoconstriction may occur either as an idiosyncratic reaction or from large dosages.
Approximately one-third of intravenous morphine is bound to plasma proteins. Free morphine is rapidly redistributed in parenchymatous tissues. The major metabolic pathway is through conjugation with glucuronic acid in the liver. Elimination half-life is approximately 1.5 to 2 hours in healthy volunteers. For intravenously administered morphine, 90% is excreted in the urine within 24 hours and traces are detectable in urine up to 48 hours. About 7-10% of administered morphine eventually appears in the feces as conjugated morphine.
INDICATIONS AND USAGE
Morphine Sulfate Injection, USP is a systemic narcotic analgesic for administration by the intravenous route. It is used for the management of pain not responsive to non-narcotic analgesics.
CONTRAINDICATIONS
Morphine Sulfate Injection, USP is contraindicated in those medical conditions which would preclude the administration of opioids by the intravenous route – allergy to morphine or other opiates, acute bronchial asthma, upper airway obstruction.
This product is contraindicated in patients known to be hypersensitive to bisulfites.
WARNINGS
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Morphine Sulfate Injection, USP administration should be limited to use by those familiar with the management of respiratory depression. Rapid intravenous administration may result in chest wall rigidity.
FACILITIES WHERE MORPHINE SULFATE INJECTION, USP IS ADMINISTERED MUST BE EQUIPPED WITH RESUSCITATIVE EQUIPMENT, OXYGEN, NALOXONE INJECTION, AND OTHER RESUSCITATIVE DRUGS.
PRECAUTIONS
General
Morphine Sulfate Injection, USP should be administered with extreme caution in aged or debilitated patients, in the presence of increased intracranial/intraocular pressure and in patients with head injury. Pupilliary changes (miosis) may obscure the course of intracranial pathology. Care is urged in patients who have a decreased respiratory reserve e.g., emphysema, severe obesity, kyphoscoliosis).
Seizures may result from high doses. Patients with known seizure disorders should be carefully observed for evidence of morphine-induced seizure activity.
Smooth muscle hypertonicity may result in biliary colic, difficulty in urination and possible urinary retention requiring catheterization.
Elimination half-life may be prolonged in patients with reduced metabolic rates and with hepatic or renal dysfunction. Hence, care should be exercised in administering morphine in these conditions, particularly with repeated dosing.
Patients with reduced circulating blood volume, impaired myocardial function or on sympatholytic drugs should be observed carefully for orthostatic hypotension, particularly in transport.
Patients with chronic obstructive pulmonary disease and patients with acute asthmatic attack may develop acute respiratory failure with administration of morphine. Use in these patients should be reserved for those whose conditions require endotracheal intubation and respiratory support or control of ventilation.
Drug Interactions
Depressant effects of morphine are potentiated by either concomitant administration or in the presence of other CNS depressants such as alcohol, sedatives, antihistaminics or psychotropic drugs (e.g., MAO inhibitors, phenothiazines, butyrophenones and tricyclic antidepressants). Premedication or intra-anesthetic use of neuroleptics with morphine may increase the risk of respiratory depression.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies of morphine sulfate in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
Pregnancy
Teratogenic effects – Pregnancy Category C. Animal reproduction studies have not been conducted with morphine sulfate. It is also not known whether morphine sulfate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Morphine sulfate should be given to a pregnant woman only if clearly needed.
Nonteratogenic effects. Infants born from mothers who have been taking morphine chronically may exhibit withdrawal symptoms.
Labor and Delivery
Intravenous morphine readily passes into the fetal circulation and may result in respiratory depression in the neonate. Naloxone and resuscitative equipment should be available for reversal of narcotic-induced respiratory depression in the neonate. In addition, intravenous morphine may reduce the strength, duration and frequency of uterine contraction resulting in prolonged labor.
ADVERSE REACTIONS
The most serious side effect is respiratory depression. Because of delay in maximum CNS effect with intravenously administered drug (30 min), rapid administration may result in overdosing. (See WARNINGS and OVERDOSAGE.)
While low doses of intravenously administered morphine have little effect on cardiovascular stability, high doses are excitatory, resulting from sympathetic hyperactivity and an increase in circulating catecholamines. Excitation of the central nervous system resulting in convulsions may accompany high doses of morphine given intravenously. Dysphoric reactions may occur and toxic psychoses have been reported.
Nausea and vomiting are frequently seen in patients following morphine administration. Pruritus and nausea/vomiting frequently can be alleviated by the intravenous administration of low doses of naloxone (0.2 mg).
Tolerance and dependence to chronically administered morphine, by whatever route, is known to occur (see DRUG ABUSE AND DEPENDENCE).
Miscellaneous side effects include constipation, headache, anxiety, depression of cough reflex, interference with thermal regulation and oliguria. Evidence of histamine release such as urticaria, wheals and/or local tissue irritation may occur.
In general, side effects are amenable to reversal by narcotic antagonists. NALOXONE HYDROCHLORIDE INJECTION AND RESUSCITATIVE EQUIPMENT SHOULD BE IMMEDIATELY AVAILABLE FOR ADMINISTRATION IN CASE OF LIFE-THREATENING OR INTOLERABLE SIDE EFFECTS.
DRUG ABUSE AND DEPENDENCE
Controlled Substance
Morphine Sulfate Injection, USP is a Schedule II controlled substance.
Abuse
Morphine has recognized abuse and dependence potential.
OVERDOSAGE
Overdosage is characterized by respiratory depression with or without concomitant CNS depression. Since respiratory arrest may result either through direct depression of the respiratory center or as the result of hypoxia, primary attention should be given to the establishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. The narcotic antagonist, naloxone, is a specific antidote. Naloxone hydrochloride (see package insert for full prescribing information) should be administered intravenously, simultaneously with respiratory resuscitation. Patients should be closely observed for evidence of renarcotization. In painful conditions, reversal of narcotic effect may result in acute onset of pain and release of catecholamines. Careful administration of naloxone may permit reversal of side effects without affecting analgesia.
DOSAGE AND ADMINISTRATION
Morphine Sulfate Injection, USP is administered by intravenous infusion only after dilution in an ADD-Vantage Flexible Diluent Container (See INSTRUCTIONS FOR USE). CAUTION: The 250 mg ADD-Vantage morphine vial must be diluted in a 250 mL ADD-Vantage Flexible Diluent Container only.
Morphine sulfate should be given in the smallest effective dose and as infrequently as possible in order to minimize the development of tolerance and physical dependence. Reduced dosage is indicated in poor-risk patients, in very young or very old patients, and in patients receiving other CNS depressants.
In patients with severe, chronic pain, dosage should be adjusted according to the severity of the pain and the response and tolerance of the patient. In patients with exceptionally severe, chronic pain or in those who have become tolerant to the analgesic effect of opiate agonists, it may be necessary to exceed the usual dosage.
Morphine sulfate should be administered by slow IV infusion after dilution. The usual dose for adults is 1 mg with a range of 0.1 mg to 5 mg per incremental dose. The physician should adjust the dosage depending on the patient's response. A controlled infusion device should be used to monitor the infusion rate.
Severe, Chronic Pain: When morphine sulfate is administered by continuous IV infusion for relief of severe, chronic pain associated with cancer, the dosage of the drug must be individualized according to the response and tolerance of the patient. If specific dose related information is required, the clinician should consult an appropriate text.
For IV administration, morphine sulfate should be injected slowly. Rapid IV injection of the drug may result in an increased frequency of opiate-induced adverse effects; severe respiratory depression, apnea, hypotension, peripheral circulatory collapse, chest wall rigidity, cardiac arrest and anaphylactoid reactions have occurred following rapid IV injection. The rate of continuous IV infusion of the drug must be individualized according to the response and tolerance of the patient.
Parenteral drug products should be inspected for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer unless solution is clear and container is undamaged. Discard unused portion.
INSTRUCTIONS FOR USE
To Use Vial in ADD-Vantage
Flexible Diluent Container
To Open:
Peel overwrap at corner and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Assemble Vial and
Flexible Diluent Container:
(Use Aseptic Technique)
1. Remove the protective covers from the top of the vial and the vial port on the diluent container as follows:
a. To remove the breakaway vial cap, swing the pull ring over the top of the vial and pull down far enough to start the opening (see FIGURE 1.), then pull straight up to remove the cap. (See FIGURE 2.) NOTE: Once the breakaway cap has been removed, do not access vial with syringe.
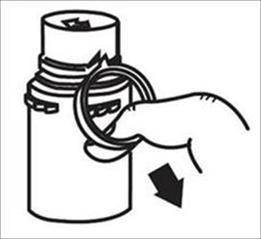 |  |
| Fig. 1 | Fig. 2 |
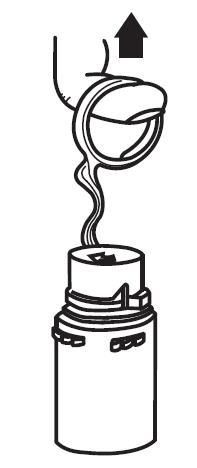
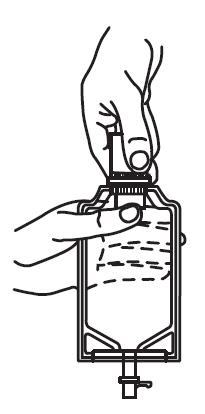
b. To remove the vial port cover, grasp the tab on the pull ring, pull up to break the three tie strings, then pull back to remove the cover. (See FIGURE 3.)
2. Screw the vial into the vial port until it will go no further. THE VIAL MUST BE SCREWED IN TIGHTLY TO ASSURE A SEAL. This occurs approximately 1/2 turn (180°) after the first audible click. (See FIGURE 4.) The clicking sound does not assure a seal; the vial must be turned as far as it will go.
NOTE: Once vial is seated, do not attempt to remove. (See FIGURE 4.)
3. Recheck the vial to assure that it is tight by trying to turn it further in the direction of assembly.
4. Label appropriately.
 |  |
| Fig. 3 | Fig. 4 |
To Prepare Admixture:
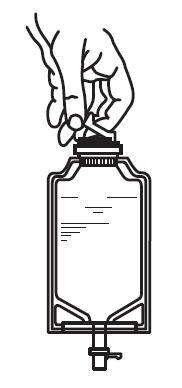
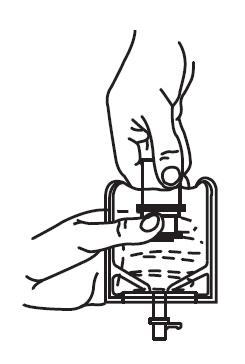
1. Squeeze the bottom of the diluent container gently to inflate the portion of the container surrounding the end of the drug vial.
2. With the other hand, push the drug vial down into the container telescoping the walls of the container. Grasp the inner cap of the vial through the walls of the container. (See FIGURE 5.)
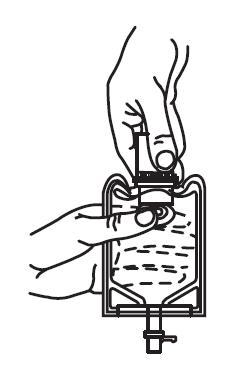
3. Pull the inner cap from the drug vial. (See FIGURE 6.) Verify that the rubber stopper has been pulled out, allowing the drug and diluent to mix.
4. Mix container contents thoroughly and use within 24 hours.
 |  |
| Fig. 5 | Fig. 6 |
Preparation for Administration
(Use Aseptic Technique)
- Confirm the activation and admixture of vial contents.
- Check for leaks by squeezing container firmly. If leaks are found, discard unit as sterility may be impaired.
- Close flow control clamp of administration set.
- Remove cover from outlet port at bottom of container.
- Insert piercing pin of administration set into port with a twisting motion until the pin is firmly seated. NOTE: See full directions on administration set carton.
- Lift the free end of the hanger loop on the bottom of the vial, breaking the two tie strings. Bend the loop outward to lock it in the upright position, then suspend container from hanger.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- Open flow control clamp and clear air from set. Close clamp.
- Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
- Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible containers in series connections.
HOW SUPPLIED
Morphine Sulfate Injection, USP is supplied in amber ADD-Vantage vials as follows:
|
NDC No. |
Concentration (mg/mL) |
Total Morphine (mg) |
|
0409-6177-14 |
25 |
100 |
|
0409-6179-14 |
25 |
250 |
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Protect from light. Store in carton until time of use.
Revised: May, 2010
Printed in USA EN-2513
Hospira, Inc., Lake Forest, IL 60045 USA
PRINCIPAL DISPLAY PANEL - 4 mL Vial Label
1 x 4 mL Single-dose ADD-Vantage® Vial
Morphine Sulfate Inj., USP
100 mg (25 mg/mL)
WARNING: Contains Sulfites
CII
For I.V. use after dilution.
Not for intrathecal or epidural use.
Hospira
Hospira, Inc., Lake Forest, IL 60045 USA

PRINCIPAL DISPLAY PANEL - 4 mL Vial Carton
One/NDC 0409-6177-14
4 mL
Single-dose
ADD-Vantage™ Vial
Rx only
CII
MORPHINE
SULFATE
Injection, USP
100 mg (25 mg/mL)
WARNING: Contains
Sulfites
For IV use after dilution.
Not for intrathecal or
epidural use.
CA-2478

PRINCIPAL DISPLAY PANEL - 4 mL Vial Tray
10 x 4 mL Single-dose ADD-Vantage™ Vials
NDC 0409-6177-14
Rx only
MORPHINE SULFATE
Injection, USP
100 mg (25 mg/mL)
CII
WARNING: Contains Sulfites
For IV use after dilution.
Not for intrathecal or epidural use.
Hospira

PRINCIPAL DISPLAY PANEL - 10 mL Vial Label
1 x 10 mL Single-dose ADD-Vantage® Vial
Morphine Sulfate Inj., USP
250 mg (25 mg/mL)
WARNING: Contains Sulfites.
CII
For I.V. use after dilution in a 250 mL
ADD-Vantage Flexible Diluent Container only.
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton
One/NDC 0409-6179-14
10 mL
Single-dose
ADD-Vantage® Vial
Rx only
CII
MORPHINE
SULFATE
Injection, USP
250 mg (25 mg/mL)
WARNING: Contains
Sulfites.
For I.V. use after dilution in a
250 mL ADD-Vantage Flexible
Diluent Container only.
Not for intrathecal or
epidural use.
CA-1852

PRINCIPAL DISPLAY PANEL - 10 mL Vial Tray
10 x 10 mL Single-dose ADD-Vantage™ Vials
NDC 0409-6179-14
Rx only
MORPHINE SULFATE
Injection, USP
250 mg (25 mg/mL)
CII
WARNING: Contains Sulfites.
For IV use after dilution in a 250 mL
ADD-Vantage Flexible Diluent Container only.
Not for intrathecal or epidural use.
Hospira

| MORPHINE SULFATE
morphine sulfate injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MORPHINE SULFATE
morphine sulfate injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 093132819 | ANALYSIS(0409-6177, 0409-6179) , LABEL(0409-6177, 0409-6179) , MANUFACTURE(0409-6177, 0409-6179) , PACK(0409-6177, 0409-6179) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 827731089 | ANALYSIS(0409-6177, 0409-6179) | |