Label: HYPERHEP B (hepatitis b immune globulin- human injection
-
NDC Code(s):
13533-636-01,
13533-636-02,
13533-636-03,
13533-636-05, view more13533-636-10, 13533-636-20, 13533-636-30, 13533-636-50
- Packager: GRIFOLS USA, LLC
- Category: PLASMA DERIVATIVE
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated April 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Hepatitis B Immune Globulin (Human) — HyperHEP B® is a clear or slightly opalescent, and colorless or pale yellow sterile solution of human hepatitis B immune globulin for intramuscular administration. HyperHEP B contains no preservative. HyperHEP B is prepared from pools of human plasma collected from healthy donors by a combination of cold ethanol fractionation, caprylate precipitation and filtration, caprylate incubation, anion exchange chromatography, nanofiltration and low pH incubation. HyperHEP B consists of a 15% to 18% protein solution at a pH of 4.1 to 4.8 in 0.16 M to 0.26 M glycine. The product contains anti-HBs antibody equivalent to or exceeding the potency of anti-HBs in a U.S. reference hepatitis B immune globulin (Center for Biologics Evaluation and Research, FDA). The U.S. reference has been tested against the World Health Organization standard Hepatitis B Immune Globulin and found to be equal to 220 international units (IU) per mL.
When medicinal biological products are administered, the risk of infectious diseases due to transmission of pathogens cannot be totally excluded. However, in the case of products prepared from human plasma, the risk of transmission of pathogens is reduced by epidemiological surveillance of the donor population and selection of individual donors by medical interview; testing of individual donations and plasma pools; and the presence in the manufacturing processes of steps with demonstrated capacity to inactivate/remove pathogen.
In the manufacturing process of HyperHEP B, there are several steps with the capacity for viral inactivation or removal.(1) The main steps of the manufacturing process that contribute to the virus clearance capacity are as follows:
- Caprylate precipitation/depth filtration
- Caprylate incubation
- Depth filtration
- Column chromatography
- Nanofiltration
- Low pH final container incubation
To provide additional assurance of the pathogen safety of the final product, the capacity of the HyperHEP B manufacturing process to remove and/or inactivate viruses has been demonstrated by laboratory spiking studies on a scaled down process model using a wide range of viruses with diverse physicochemical properties.
The combination of all of the above mentioned measures provides the final product with a high margin of safety from the potential risk of transmission of infectious viruses.
The caprylate/chromatography manufacturing process was also investigated for its capacity to decrease the infectivity of an experimental agent of transmissible spongiform encephalopathy (TSE), considered as a model for the variant Creutzfeldt-Jakob disease (vCJD), and Creutzfeldt-Jakob disease (CJD) agents.(1) These studies provide reasonable assurance that low levels of vCJD/CJD agent infectivity, if present in the starting material, would be removed by the caprylate/chromatography manufacturing process.
- Caprylate precipitation/depth filtration
-
CLINICAL PHARMACOLOGY
Hepatitis B Immune Globulin (Human) provides passive immunization for individuals exposed to the hepatitis B virus (HBV) as evidenced by a reduction in the attack rate of hepatitis B following its use.(2-7) The administration of the usual recommended dose of this immune globulin generally results in a detectable level of circulating anti-HBs which persists for approximately 2 months or longer. The highest antibody (IgG) serum levels were seen in the following distribution of subjects studied: (8)
DAY % OF SUBJECTS 3 38.9% 7 41.7% 14 11.1% 21 8.3% Mean values for half-life were between 17.5 and 25 days, with the shortest being 5.9 days and the longest 35 days.(8)
Cases of type B hepatitis are rarely seen following exposure to HBV in persons with preexisting anti-HBs. No confirmed instance of transmission of hepatitis B has been associated with this product.
In a clinical study, 12 healthy human adults receiving another hyperimmune immune globulin product, Rabies Immune Globulin (Human), HyperRAB®, prepared by the same manufacturing process, detectable passive antibody titers were observed in the serum of all subjects by 24 hours post injection and persisted through the 21 day study period.
-
INDICATIONS AND USAGE
Recommendations on post-exposure prophylaxis are based on available efficacy data and on the likelihood of future HBV exposure for the person requiring treatment. In all exposures, a regimen combining Hepatitis B Immune Globulin (Human) with hepatitis B vaccine will provide both short- and long-term protection, will be less costly than the two-dose Hepatitis B Immune Globulin (Human) treatment alone, and is the treatment of choice.(9)
HyperHEP B is indicated for post-exposure prophylaxis in the following situations:
Acute Exposure to Blood Containing HBsAg
After either parenteral exposure, e.g., by accidental “needlestick” or direct mucous membrane contact (accidental splash), or oral ingestion (pipetting accident) involving HBsAg-positive materials such as blood, plasma or serum. For inadvertent percutaneous exposure, a regimen of two doses of Hepatitis B Immune Globulin (Human), one given after exposure and one a month later, is about 75% effective in preventing hepatitis B in this setting.
Perinatal Exposure of Infants Born to HBsAg-positive Mothers
Infants born to HBsAg-positive mothers are at risk of being infected with hepatitis B virus and becoming chronic carriers.(6,9-11) This risk is especially great if the mother is HBeAg-positive.(13-15) For an infant with perinatal exposure to an HBsAg-positive and HBeAg-positive mother, a regimen combining one dose of Hepatitis B Immune Globulin (Human) at birth with the hepatitis B vaccine series started soon after birth is 85%–95% effective in preventing development of the HBV carrier state.(9,15) Regimens involving either multiple doses of Hepatitis B Immune Globulin (Human) alone or the vaccine series alone have 70%–90% efficacy, while a single dose of Hepatitis B Immune Globulin (Human) alone has only 50% efficacy.(9,16)
Sexual Exposure to an HBsAg-positive Person
Sex partners of HBsAg-positive persons are at increased risk of acquiring HBV infection. For sexual exposure to a person with acute hepatitis B, a single dose of Hepatitis B Immune Globulin (Human) is 75% effective if administered within 2 weeks of last sexual exposure.(9)
Household Exposure to Persons with Acute HBV Infection
Since infants have close contact with primary care-givers and they have a higher risk of becoming HBV carriers after acute HBV infection, prophylaxis of an infant less than 12 months of age with Hepatitis B Immune Globulin (Human) and hepatitis B vaccine is indicated if the mother or primary care-giver has acute HBV infection.(9)
Administration of Hepatitis B Immune Globulin (Human) either preceding or concomitant with the commencement of active immunization with Hepatitis B Vaccine provides for more rapid achievement of protective levels of hepatitis B antibody, than when the vaccine alone is administered.(17) Rapid achievement of protective levels of antibody to hepatitis B virus may be desirable in certain clinical situations, as in cases of accidental inoculations with contaminated medical instruments.(17) Administration of Hepatitis B Immune Globulin (Human) either 1 month preceding or at the time of commencement of a program of active vaccination with Hepatitis B Vaccine has been shown not to interfere with the active immune response to the vaccine.(17)
- CONTRAINDICATIONS
-
WARNINGS
HyperHEP B is made from human plasma. Products made from human plasma may contain infectious agents, such as viruses, and, theoretically, the Creutzfeldt-Jakob Disease (CJD) agent that can cause disease. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain current virus infections, and by inactivating and/or removing certain viruses. Despite these measures, such products can still potentially transmit disease. There is also the possibility that unknown infectious agents may be present in such products. Individuals who receive infusions of blood or plasma products may develop signs and/or symptoms of some viral infections, particularly hepatitis C. ALL infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Grifols Therapeutics LLC [1-800-520-2807].
The physician should discuss the risks and benefits of this product with the patient, before prescribing or administering it to the patient.
HyperHEP B should be given with caution to patients with a history of prior systemic allergic reactions following the administration of human immune globulin preparations. Epinephrine should be available.
In patients who have severe thrombocytopenia or any coagulation disorder that would contraindicate intramuscular injections, Hepatitis B Immune Globulin (Human) should be given only if the expected benefits outweigh the risks.
-
PRECAUTIONS
General
HyperHEP B should not be administered intravenously because of the potential for serious reactions. Injections should be made intramuscularly, and care should be taken to draw back on the plunger of the syringe before injection in order to be certain that the needle is not in a blood vessel. Intramuscular injections are preferably administered in the deltoid muscle of the upper arm or lateral thigh muscle. The gluteal region should not be used as an injection site because of the risk of injury to the sciatic nerve.(18) An individual decision as to which muscle is injected must be made for each patient based on the volume of material to be administered.
Drug Interactions
Although administration of Hepatitis B Immune Globulin (Human) did not interfere with measles vaccination,(19) it is not known whether Hepatitis B Immune Globulin (Human) may interfere with other live virus vaccines. Therefore, use of such vaccines should be deferred until approximately 3 months after Hepatitis B Immune Globulin (Human) administration. Hepatitis B Vaccine may be administered at the same time, but at a different injection site, without interfering with the immune response.(17) No interactions with other products are known.
-
ADVERSE REACTIONS
Local pain and tenderness at the injection site, urticaria and angioedema may occur; anaphylactic reactions, although rare, have been reported following the injection of human immune globulin preparations.(20)
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
Acute Exposure to Blood Containing HBsAg(16)
Table 1 summarizes prophylaxis for percutaneous (needlestick or bite), ocular, or mucous-membrane exposure to blood according to the source of exposure and vaccination status of the exposed person. For greatest effectiveness, passive prophylaxis with Hepatitis B Immune Globulin (Human) should be given as soon as possible after exposure (its value beyond 7 days of exposure is unclear). If Hepatitis B Immune Globulin (Human) is indicated (see Table 1), an injection of 0.06 mL/kg of body weight should be administered intramuscularly (see PRECAUTIONS) as soon as possible after exposure and within 24 hours, if possible. Consult Hepatitis B Vaccine package insert for dosage information regarding that product.
- *
- Hepatitis B Immune Globulin (Human), dose 0.06 mL / kg IM.
- †
- HB Vaccine dose 20 μg IM for adults; 10 μg IM for infants or children under 10 years of age. First dose within 1 week; second and third doses, 1 and 6 months later.
- ‡
- Less than 10 sample ratio units (SRU) by radioimmunoassay (RIA), negative by enzyme immunoassay (EIA).
Table 1. (adapted from (21))
Recommendations for Hepatitis B Prophylaxis Following Percutaneous or Permucosal ExposureExposed Person Source Unvaccinated Vaccinated HBsAg-Positive 1. Hepatitis B Immune Globulin (Human) x1 immediately* 1. Test exposed person for anti-HBs. 2. Initiate HB Vaccine Series† 2. If inadequate antibody,‡ Hepatitis B Immune Globulin (Human) (x1) immediately plus HB Vaccine booster dose, or 2 doses of HBIG,* one as soon as possible after exposure and the second 1 month later. Known Source
(High Risk)1. Initiate HB Vaccine Series 1. Test Source for HBsAg only if exposed is vaccine nonresponder; if source is HBsAg-positive, give Hepatitis B Immune Globulin (Human) x1 immediately plus HB Vaccine booster dose, or 2 doses of HBIG,* one as soon as possible after exposure and the second 1 month later. 2. Test source for HBsAg. If positive, Hepatitis B Immune Globulin (Human) x1 Low Risk
HBsAg-PositiveInitiate HB Vaccine series Nothing required. Unknown
SourceInitiate HB Vaccine series within 7 days of exposure Nothing required. For persons who refuse Hepatitis B Vaccine, a second dose of Hepatitis B Immune Globulin (Human) should be given 1 month after the first dose.
Prophylaxis of Infants Born to HBsAg and HBeAg Positive Mothers
Efficacy of prophylactic Hepatitis B Immune Globulin (Human) in infants at risk depends on administering Hepatitis B Immune Globulin (Human) on the day of birth. It is therefore vital that HBsAg-positive mothers be identified before delivery.
Hepatitis B Immune Globulin (Human) (0.5 mL) should be administered intramuscularly (IM) to the newborn infant after physiologic stabilization of the infant and preferably within 12 hours of birth. Hepatitis B Immune Globulin (Human) efficacy decreases markedly if treatment is delayed beyond 48 hours. Hepatitis B Vaccine should be administered IM in three doses of 0.5 mL of vaccine (10 μg) each. The first dose should be given within 7 days of birth and may be given concurrently with Hepatitis B Immune Globulin (Human) but at a separate site. The second and third doses of vaccine should be given 1 month and 6 months, respectively, after the first. If administration of the first dose of Hepatitis B Vaccine is delayed for as long as 3 months, then a 0.5 mL dose of Hepatitis B Immune Globulin (Human) should be repeated at 3 months. If Hepatitis B Vaccine is refused, the 0.5 mL dose of Hepatitis B Immune Globulin (Human) should be repeated at 3 and 6 months. Hepatitis B Immune Globulin (Human) administered at birth should not interfere with oral polio and diphtheria-tetanus-pertussis vaccines administered at 2 months of age.(16)
Sexual Exposure to an HBsAg-positive Person
All susceptible persons whose sex partners have acute hepatitis B infection should receive a single dose of HBIG (0.06 mL/kg) and should begin the hepatitis B vaccine series if prophylaxis can be started within 14 days of the last sexual contact or if sexual contact with the infected person will continue (see Table 2 below). Administering the vaccine with HBIG may improve the efficacy of postexposure treatment. The vaccine has the added advantage of conferring long-lasting protection.(9)
Household Exposure to Persons with Acute HBV Infection
Prophylactic treatment with a 0.5 mL dose of Hepatitis B Immune Globulin (Human) and hepatitis B vaccine is indicated for infants <12 months of age who have been exposed to a primary care-giver who has acute hepatitis B. Prophylaxis for other household contacts of persons with acute HBV infection is not indicated unless they have had identifiable blood exposure to the index patient, such as by sharing toothbrushes or razors. Such exposures should be treated like sexual exposures. If the index patient becomes an HBV carrier, all household contacts should receive hepatitis B vaccine. (9)
Hepatitis B Immune Globulin (Human) may be administered at the same time (but at a different site), or up to 1 month preceding Hepatitis B Vaccination without impairing the active immune response from Hepatitis B Vaccination.(17)
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Administer intramuscularly. Do not inject intravenously.
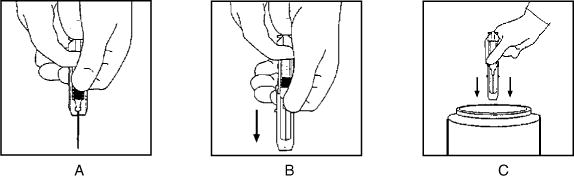
Hepatitis B Immune Globulin (Human) — HyperHEP B® is supplied in a syringe with an attached UltraSafe® Needle Guard for your protection and convenience, as well as in vials. Please follow instructions below for proper use of syringe and UltraSafe® Needle Guard.
Directions for Syringe Usage
-
Remove the prefilled syringe from the package. Lift syringe by barrel,
not by plunger. -
Twist the plunger rod clockwise until the threads are seated.
-
With the needle shield secured on the syringe tip, push the plunger rod forward a few millimeters to break any friction seal between the stopper and the glass syringe barrel.
-
Remove the needle shield and expel air bubbles. [Do not remove the needle shield to prepare the product for administration until immediately prior to the anticipated injection time.]
-
Proceed with hypodermic needle puncture.
-
Aspirate prior to injection to confirm that the needle is not in a vein or artery.
- Inject the medication.
-
Keeping your hands behind the needle, grasp the guard with free hand and slide forward toward needle until it is completely covered and guard clicks into place. If audible click is not heard, guard may not be completely activated. (See Diagrams A and B)
-
Place entire prefilled glass syringe with guard activated into an approved sharps container for proper disposal. (See Diagram C)
A number of factors could reduce the efficacy of this product or even result in an ill effect following its use. These include improper storage and handling of the product after it leaves our hands, diagnosis, dosage, method of administration and biological differences in individual patients. Because of these factors, it is important that this product be stored properly and that the directions be followed carefully during use.
-
HOW SUPPLIED
HyperHEP B is supplied in a 0.5 mL neonatal single dose syringe with attached needle, a 1 mL single dose syringe with attached needle and a 1 mL and a 5 mL single dose vial.
HyperHEP B contains no preservative and is not made with natural rubber latex.NDC Number Size 13533-636-03 0.5 mL syringe 13533-636-02 1 mL syringe 13533-636-01 1 mL vial 13533-636-05 5 mL vial - STORAGE
- CAUTION
-
REFERENCES
- Barnette D, Roth NJ, Hotta J, et al. Pathogen safety profile of a 10% IgG preparation manufactured using a depth filtration-modified process. Biologicals 2012;40:247-53.
- Grady GF, Lee VA: Hepatitis B immune globulin — prevention of hepatitis from accidental exposure among medical personnel. N Engl J Med 293(21):1067–70, 1975.
- Seeff LB, Zimmerman HJ, Wright EC, et al: Efficacy of hepatitis B immune serum globulin after accidental exposure. Lancet 2(7942):939-41, 1975.
- Krugman S, Giles JP: Viral hepatitis, type B (MS-2-strain). Further observations on natural history and prevention. N Engl J Med 288(15):755-60, 1973.
- Current trends: Health status of Indochinese refugees: malaria and hepatitis B. MMWR 28(39):463-4; 469-70, 1979.
- Jhaveri R, Rosenfeld W, Salazar JD, et al: High titer multiple dose therapy with HBIG in newborn infants of HBsAg positive mothers. J Pediatr 97(2):305–8, 1980.
- Hoofnagle JH, Seeff LB, Bales ZB, et al: Passive-active immunity from hepatitis B immune globulin. Ann Intern Med 91(6):813-8, 1979.
- Scheiermann N, Kuwert EK: Uptake and elimination of hepatitis B immunoglobulins after intramuscular application in man. Dev Biol Stand 54:347-55, 1983.
- Recommendations of the Immunization Practices Advisory Committee (ACIP): Hepatitis B Virus: A Comprehensive Strategy for Eliminating Transmission in the United States Through Universal Childhood Vaccination. Appendix A: Postexposure Prophylaxis for Hepatitis B. MMWR 40(RR-13):21-25, 1991.
- Stevens CE, Beasley RP, Tsui J, et al: Vertical transmission of hepatitis B antigen in Taiwan. N Engl J Med 292(15):771-4, 1975.
- Shiraki K, Yoshihara N, Kawana T, et al: Hepatitis B surface antigen and chronic hepatitis in infants born to asymptomatic carrier mothers. Am J Dis Child 131(6):644-7, 1977.
- Recommendation of the Immunization Practices Advisory Committee (ACIP): Immune globulins for protection against viral hepatitis. MMWR 30(34):423-8; 433-5, 1981.
- Okada K, Kamiyama I, Inomata M, et al: e antigen and anti-e in the serum of asymptomatic carrier mothers as indicators of positive and negative transmission of hepatitis B virus to their infants. N Engl J Med 294(14):746-9, 1976.
- Beasley RP, Trepo C, Stevens CE, et al: The e antigen and vertical transmission of hepatitis B surface antigen. Am J Epidemiol 105(2):94-8, 1977.
- Beasley RP, Hwang LY, Lee GCY, et al: Prevention of perinatally transmitted hepatitis B virus infections with hepatitis B immune globulin and hepatitis B vaccine. Lancet 2(8359): 1099-102, 1983.
- Recommendation of the Immunization Practices Advisory Committee (ACIP): Recommendations for protection against viral hepatitis. MMWR 34(22):313–35, 1985.
- Szmuness W, Stevens CE, Olesko WR, et al: Passive-active immunisation against hepatitis B: immunogenicity studies in adult Americans. Lancet 1:575–77, 1981.
- Recommendations of the Advisory Committee on Immunization Practices (ACIP) and the AmericanAcademy of Family Physicians (AAFP): General recommendations on immunization. MMWR 2002:51 (RR02), 1-36.
- Beasley RP, Hwang LY: Measles vaccination not interfered with by hepatitis B immune globulin. Lancet 1:161, 1982.
- Ellis EF, Henney CS: Adverse reactions following administration of human gamma globulin. J Allerg 43(1):45-54, 1969.
- Recommendations of the Immunization Practices Advisory Committee (ACIP): Update on Adult Immunization. Table 9. Recommendations for postexposure prophylaxis for percutaneous or permucosal exposure to hepatitis B, United States. MMWR 40(RR-12):70, 1991.
- Recommendations of the Immunization Practices Advisory Committee (ACIP): Update on Adult Immunization. Table 10. Recommendations for postexposure prophylaxis for perinatal and sexual exposure to hepatitis B, United States. MMWR 40(RR-12):71, 1991.
Rev. 8/2022

Grifols Therapeutics LLC
Research Triangle Park, NC 27709 USA
U.S. License No. 18713063868
-
PACKAGE LABEL
≥ 110 IU / 0.5 mL
Hepatitis B
Immune
Globulin
(Human)
HyperHEP B®
0.5 mL
Solution for Intramuscular
Injection
≥ 220 IU/mL
Contents: One 0.5 mL neonatal
single-dose disposable syringe
with attached needle.
Hepatitis B Immune Globulin
(Human) is a sterile solution
of immunoglobulin containing
15% — 18% protein stabilized
with 0.16 M to 0.26 M glycine.
FOR INTRAMUSCULAR
INJECTION ONLY.
DO NOT GIVE INTRAVENOUSLY.
NDC 13533-636-03
GRIFOLS
The patient and physician should
discuss the risks and benefits of this
product.
The potency of the solution in each
syringe is equivalent to or exceeds
that of the U.S. Food and Drug
Administration Reference Hepatitis B
Immune Globulin. The U.S. reference
has been tested against the World
Health Organization standard
Hepatitis B Immune Globulin and
found to be equal to 220 international
units (IU) per mL.
0.5 mL
For complete dosage and
administration information, read
enclosed package insert.
For directions for syringe usage, see
enclosed package insert.
Store at 2°C to 8°C (36°F to 46°F).
Do not freeze.
Do not use if the syringe is prematurely
engaged.
Not returnable for credit or
exchange.
Rx only
Not made with natural rubber latex.
No preservative
Discard unused portion.
Grifols Therapeutics LLC
Research Triangle Park,
NC 27709 USA
U.S. License No. 1871
GRIFOLS
NDC 13533-636-03 GTIN 00313533636036
LOT XXXXXXXXXXXX
EXP DDMMMYYYY
SN XXXXXXXXXXXXXXXXXX
HyperHEP B®
≥ 110 IU / 0.5 mL
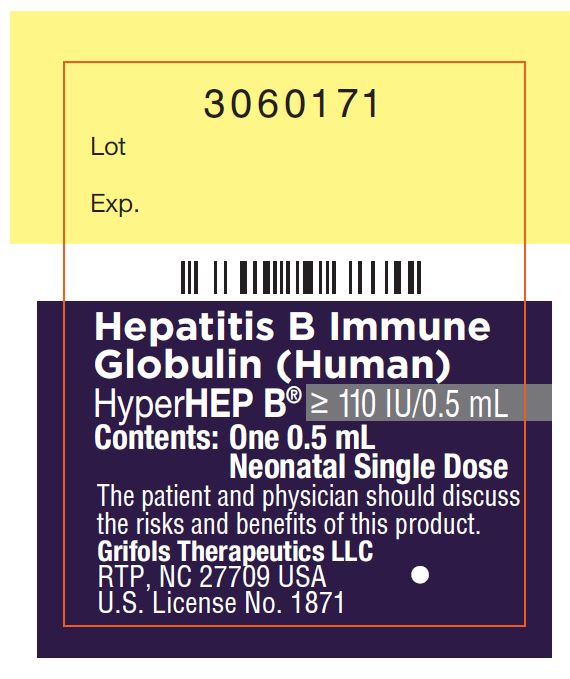
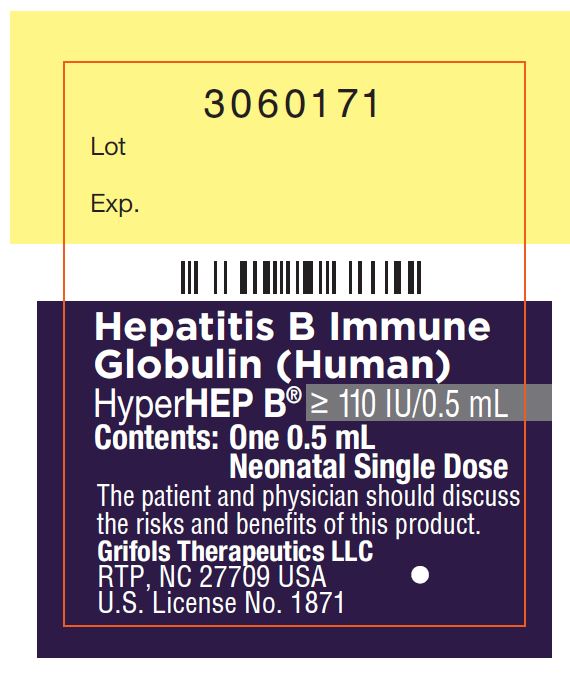
Carton: 3060176
3060171
Lot
Exp.
Hepatitis B Immune
Globulin (Human)
HyperHEP B® ≥ 110 IU/0.5 mL
Contents: One 0.5 mL
Neonatal Single Dose
The patient and physician should discuss
the risks and benefits of this product.
Grifols Therapeutics LLC
RTP, NC 27709 USA
U.S. License No. 1871
≥ 220 IU / 1 mL
Hepatitis B
Immune
Globulin
(Human)
HyperHEP B®
1 mL
Solution for Intramuscular
Injection
≥ 220 IU/mL
FOR INTRAMUSCULAR
INJECTION ONLY. DO NOT GIVE
INTRAVENOUSLY.
NDC 13533-636-01
GRIFOLS
The patient and physician should
discuss the risks and benefits of
this product.
One Single-Dose Vial
Discard unused portion.
For complete dosage and
administration information, read
enclosed package insert.
Store at 2°C to 8°C (36°F to
46°F). Do not freeze.
1 mL
If the shrink band is absent or
shows any sign of tampering, do
not use the product and notify
Grifols Therapeutics LLC
immediately.
Not returnable for credit or
exchange.
Rx only
GRIFOLS
Hepatitis B Immune Globulin
(Human) is a sterile solution of
immunoglobulin containing 15%–18% protein
stabilized with 0.16 M to 0.26 M glycine.
1 mL
Grifols Therapeutics LLC
Research Triangle Park,
NC 27709 USA
U.S. License No. 1871
GRIFOLS
The potency is equivalent to or
exceeds that of the U.S. Food and
Drug Administration Reference
Hepatitis B Immune Globulin. The
U.S. reference has been tested
against the World Health
Organization standard Hepatitis B
Immune Globulin and found to be
equal to 220 international units
(IU) per mL.
1 mL
Not made with natural rubber
latex.
No preservative
GRIFOLS
NDC 13533-636-01
GTIN 00313533636012
LOT XXXXXXXXXX
EXP DDMMMYYYY
SN XXXXXXXXXXXXXXXX
HyperHEP B®
≥ 220 IU / 1 mL
Carton: 3060178
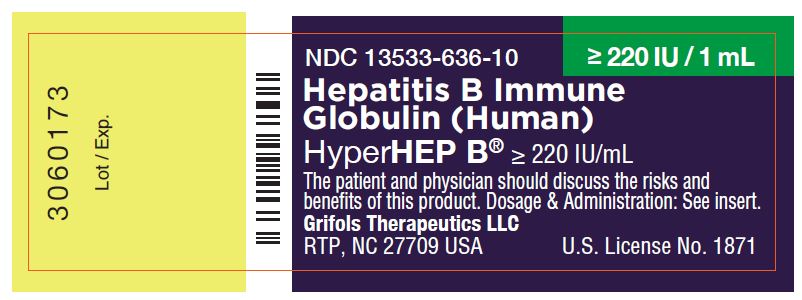
NDC 13533-636-10 ≥ 220 IU / 1 mL
Hepatitis B Immune
Globulin (Human)
HyperHEP B® ≥ 220 IU/mL
The patient and physician should discuss the risks and
benefits of this product. Dosage & Administration: See insert.
Grifols Therapeutics LLC
RTP, NC 27709 USA U.S. License No. 1871
3060173
Lot / Exp.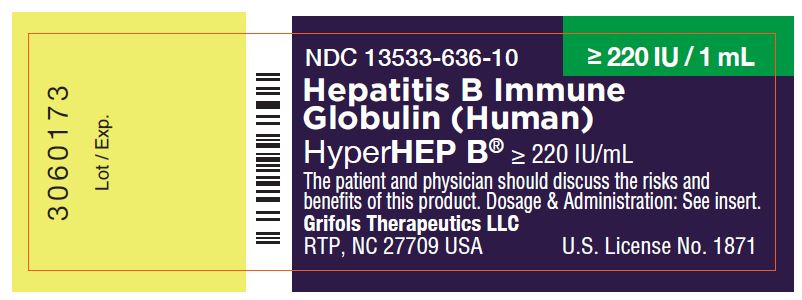
-
INGREDIENTS AND APPEARANCE
HYPERHEP B
hepatitis b immune globulin (human) injectionProduct Information Product Type PLASMA DERIVATIVE Item Code (Source) NDC:13533-636 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Human Hepatitis B Virus Immune Globulin (UNII: XII270YC6M) (Human Hepatitis B Virus Immune Globulin - UNII:XII270YC6M) Human Hepatitis B Virus Immune Globulin 220 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength Glycine (UNII: TE7660XO1C) Water (UNII: 059QF0KO0R) Product Characteristics Color YELLOW (colorless or pale yellow or light brown) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13533-636-01 1 in 1 CARTON 1 NDC:13533-636-10 1 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 2 NDC:13533-636-02 1 in 1 CARTON 2 NDC:13533-636-20 1 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) 3 NDC:13533-636-03 1 in 1 CARTON 3 NDC:13533-636-30 0.5 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) 4 NDC:13533-636-05 1 in 1 CARTON 4 NDC:13533-636-50 5 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA101146 10/09/1996 Labeler - GRIFOLS USA, LLC (048987452) Establishment Name Address ID/FEI Business Operations Grifols Therapeutics LLC 611019113 manufacture(13533-636)