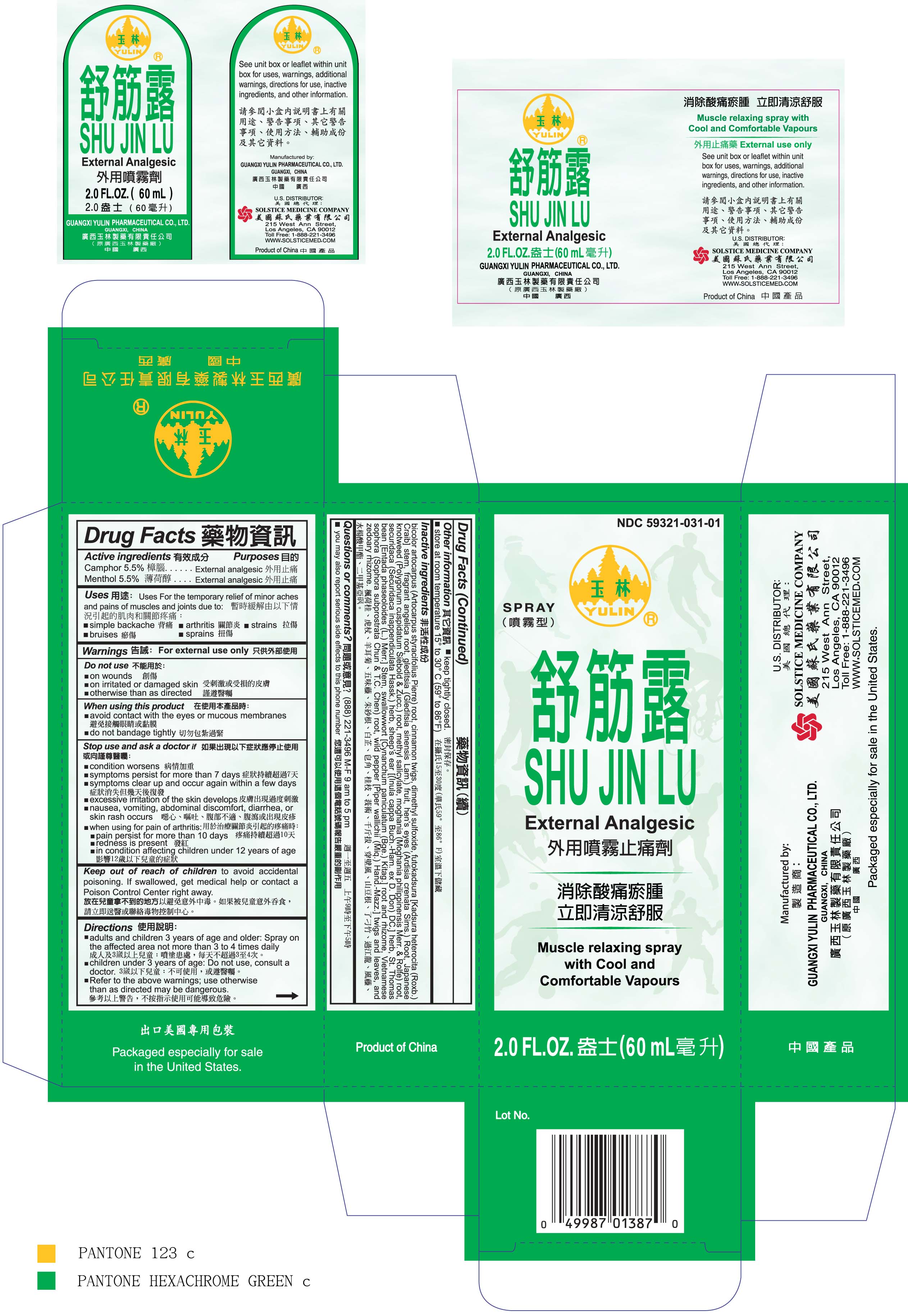
Label: SHU JIN LU- camphor and menthol spray
- NDC Code(s): 59321-031-01
- Packager: GUANGXI YULIN PHARMACEUTICAL CO LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 15, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
-
STOP USE
Stop use and ask a doctor if
condition worsens
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
excessive irritation of the skin develops
nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
when using for pain of arthritis:
pain persists for more than 10 days
redness is present
in conditions affecting children under 12 years of age
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Inactive ingredients
bicolor artocarpus (Artocarpus styracifolius Pierre) root,
cinnamon twigs,
dimethyl sulfoxide,
futokadsura Kadsura heterocita (Roxb.) Craib stem,
fragrant angelica root,
gleditsia (Gleditsia sinensis Lam.) fruit,
hen’s eyes (Ardisia crenata Sims.) Root,
Japanese knotweed (Polygonum cuspidatum Siebold Zucc.) root,
methyl salicylate,
moghania (Moghania philippinensis Merr. Rolfe) root,
securidaca (Securidaca inappendiculata Hassk.) herb,
sheep’s ear (Inula cappa Buch.-Ham. ex D. Don) DC. herb,
St. Thomas bean Entada phaseoloides (L.) Merr. Stem,
swallowwort Cynanchum paniculatum (Bge.) Kitag. root and rhizome,
Vietnamese sophora (Sophora subprostrata Chun T.C. Chen) root,
wild pepper Piper wallichii (Miq.) Hand.-Mazz. twigs and leaves,
and zedoary rhizome.
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SHU JIN LU
camphor and menthol sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59321-031 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 5.5 g in 100 mL MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 5.5 g in 100 mL Inactive Ingredients Ingredient Name Strength CINNAMON (UNII: 5S29HWU6QB) DIMETHYL SULFOXIDE (UNII: YOW8V9698H) ANGELICA DAHURICA ROOT (UNII: 1V63N2S972) GLEDITSIA SINENSIS FRUIT (UNII: A6W0J6UO7Q) ARDISIA JAPONICA STEM (UNII: Q6CE3213C7) POLYGONUM CUSPIDATUM ROOT (UNII: 7TRV45YZF7) METHYL SALICYLATE (UNII: LAV5U5022Y) ENTADA PHASEOLOIDES LEAF (UNII: W20C93AH7K) CHELIDONIUM MAJUS (UNII: 7E889U5RNN) SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) ZEDOARY (UNII: 123C43G128) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59321-031-01 60 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 04/07/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 04/07/2011 Labeler - GUANGXI YULIN PHARMACEUTICAL CO LTD (653862581) Establishment Name Address ID/FEI Business Operations GUANGXI YULIN PHARMACEUTICAL CO LTD 653862581 manufacture(59321-031)