LACTULOSE- lactulose solution
Cardinal Health
----------
LACTULOSE SOLUTION, USP
DESCRIPTION
Lactulose is a synthetic disaccharide in solution form for oral administration. Each 15 mL of lactulose solution contains: 10 g lactulose (and less than 1.6 g galactose, less than 1.2 g lactose, and 1.2 g of other sugars). Also contains FD&C Yellow No. 6, purified water, USP and wild cherry flavoring. A minimal quantity of sodium hydroxide, NF is used to adjust pH when necessary. The pH range is 2.5 to 6.5.
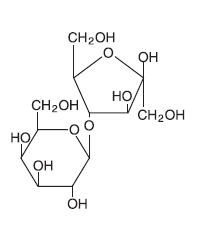
Lactulose is a colonic acidifier which promotes laxation.The chemical name for lactulose is 4-O-β-D-galactopyranosyl-D-fructofuranose. It has the following structural formula:
The molecular weight is 342.30. It is freely soluble in water.
CLINICAL PHARMACOLOGY
Lactulose is poorly absorbed from the gastrointestinal tract and no enzyme capable of hydrolysis of this disaccharide is present in human gastrointestinal tissue. As a result, oral doses of lactulose reach the colon virtually unchanged. In the colon, lactulose is broken down primarily to lactic acid, and also to small amounts of formic and acetic acids, by the action of colonic bacteria, which results in an increase in osmotic pressure and slight acidification of the colonic contents. This in turn causes an increase in stool water content and softens the stool.
Since lactulose does not exert its effect until it reaches the colon, and since transit time through the colon may be slow, 24 to 48 hours may be required to produce the desired bowel movement.
Lactulose given orally to man and experimental animals resulted in only small amounts reaching the blood. Urinary excretion has been determined to be 3% or less and is essentially complete within 24 hours.
INDICATIONS AND USAGE
For the treatment of constipation. In patients with a history of chronic constipation, lactulose solution therapy increases the number of bowel movements per day and the number of days on which bowel movements occur.
CONTRAINDICATIONS
Since lactulose solution contains galactose (less than 1.6 g/15 mL), it is contraindicated in patients who require a low galactose diet.
WARNINGS
A theoretical hazard may exist for patients being treated with lactulose solution who may be required to undergo electrocautery procedures during proctoscopy or colonoscopy. Accumulation of H2 gas in significant concentration in the presence of an electrical spark may result in an explosive reaction. Although this complication has not been reported with lactulose, patients on lactulose therapy undergoing such procedures should have a thorough bowel cleansing with a non-fermentable solution. Insufflation of CO2 as an additional safeguard may be pursued but is considered to be a redundant measure.
PRECAUTIONS
General
Since lactulose solution contains galactose (less than 1.6 g/15 mL) and lactose (less than 1.2 g/15 mL) it should be used with caution in diabetics.
Information for Patients
In the event that an unusual diarrheal condition occurs, contact your physician.
Laboratory Tests
Elderly, debilitated patients who receive lactulose for more than six months should have serum electrolytes (potassium, chloride, carbon dioxide) measured periodically.
Drug Interactions
Results of preliminary studies in humans and rats suggest that non-absorbable antacids given concurrently with lactulose may inhibit the desired lactulose-induced drop in colonic pH. Therefore, a possible lack of desired effect of treatment should be taken into consideration before such drugs are given concomitantly with lactulose solution.
Carcinogenesis, Mutagenesis, Impairment of Fertility
There are no known human data on long-term potential for carcinogenicity, mutagenicity, or impairment of fertility.
There are no known animal data on long-term potential for mutagenicity.
Administration of lactulose solution in the diet of mice for 18 months in concentrations of 3 and 10 percent (v/w) did not produce any evidence of carcinogenicity.
In studies of mice, rats, and rabbits, doses of lactulose solution up to 6 or 12 mL/kg/day produced no deleterious effects in breeding, conception, or parturition.
Pregnancy
Teratogenic Effects
Pregnancy category B
Reproduction studies have been performed in mice, rats, and rabbits at doses up to 3 or 6 times the usual human oral dose and have revealed no evidence of impaired fertility or harm to the fetus due to lactulose. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
ADVERSE REACTIONS
Precise frequency data are not available.
Initial dosing may produce flatulence and intestinal cramps, which are usually transient. Excessive dosage can lead to diarrhea with potential complications such as loss of fluids, hypokalemia, and hypernatremia.
Nausea and vomiting have been reported.
OVERDOSAGE
DOSAGE AND ADMINISTRATION
The usual dose is 1 to 2 tablespoonfuls (15 to 30 mL, containing 10 g to 20 g of lactulose) daily. The dose may be increased to 60 mL daily if necessary. Twenty-four to 48 hours may be required to produce a normal bowel movement.
Note: Some patients have found that lactulose solution may be more acceptable when mixed with fruit juice, water or milk.
HOW SUPPLIED
Lactulose Solution is available as a cherry-flavored solution, 10 g/15 mL, supplied as follows:
NDC 66689-039-01: 15 mL unit dose cup.
NDC 66689-039-50: Case contains 50 unit dose cups of 15 mL (66689-039-01), packaged in 5 trays of 10 unit dose cups each.
Lactulose Solution is available as a cherry-flavored solution, 20 g/30 mL, supplied as follows:
NDC 66689-038-01: 30 mL unit dose cup.
NDC 66689-038-50: Case contains 50 unit dose cups of 30 mL (66689-038-01), packaged in 5 trays of 10 unit dose cups each.
NDC 66689-038-99: Case contains 100 unit dose cups of 30 mL (66689-038-01), packaged in 10 trays of 10 unit dose cups each.
Lactulose Solution contains lactulose 667 mg/mL (10 g/15 mL).
When ordering this product, include the product code number or NDC in the description.
Storage
Store at 20°-25°C (68°-77°F) [See USP Controlled Room Temperature]. Avoid subfreezing temperatures.
Under recommended storage conditions, a normal darkening of color may occur. Such darkening is characteristic of sugar solutions and does not affect therapeutic action. Prolonged exposure to temperatures above 86°F (30°C) or to direct light may cause extreme darkening and turbidity which may be pharmaceutically objectionable. If this condition develops, do not use.
Prolonged exposure to freezing temperatures may cause change to a semi-solid, too viscous to pour. Viscosity will return to normal upon warming to room temperature.
Dispense in a tight, light-resistant container as defined in the USP.
| LACTULOSE
lactulose solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Cardinal Health (188557102) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Cardinal Health | 188557102 | REPACK(55154-4055) | |