STAY ALERT AID- caffeine tablet
Rite Aid
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Rite Aid 44-226-Delisted
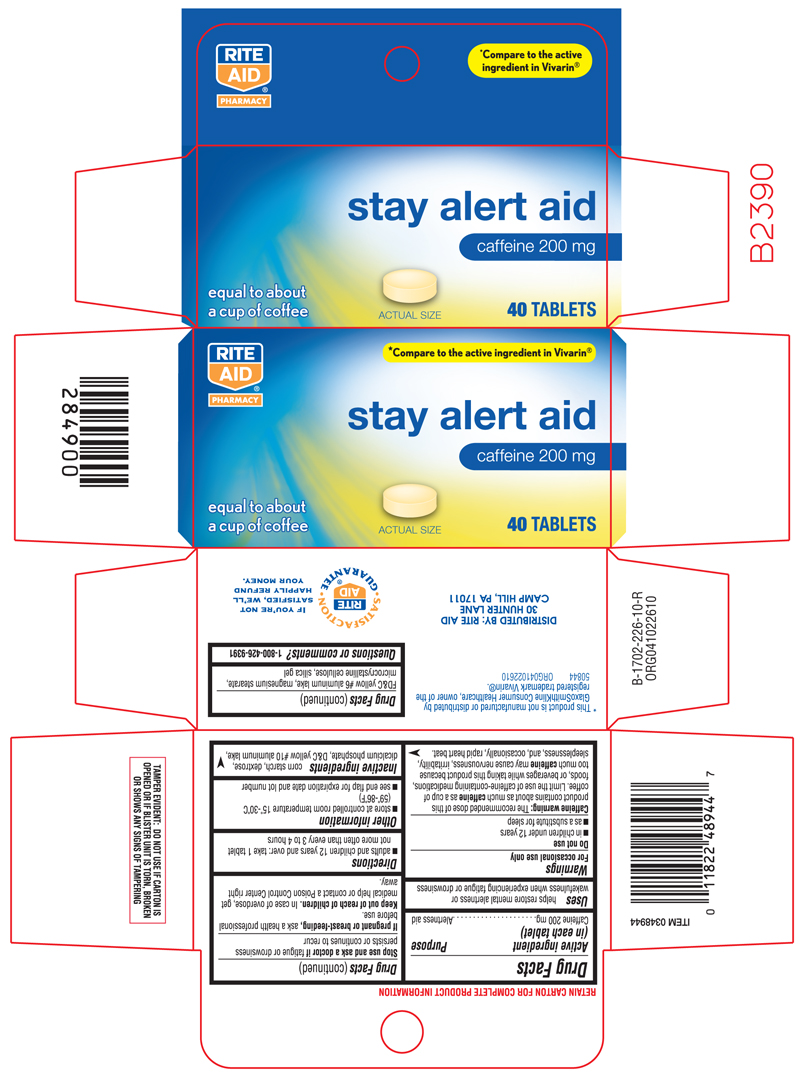
Warnings
For occasional use only
Caffeine warning:
The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heart beat.
Directions
-
adults and children 12 years and over: take 1 tablet not more often than every 3 to 4 hours
Other information
- store at controlled room temperature 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, dextrose, dicalcium phosphate, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, magnesium stearate, microcrystalline cellulose, silica gel
Principal Display Panel
RITE AID®
PHARMACY
*Compare to the active ingredient in Vivarin®
stay alert aid
caffeine 200 mg
equal to about a cup of coffee
actual size
40 TABLETS
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF A BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by GlaxoSmithKline Consumer Healthcare, owner of the registered trademark Vivarin®
50844 ORG041022610
DISTRIBUTED BY:
RITE AID
30 HUNTER LANE
CAMP HILL, PA 17011
Rite Aid 44-226
| STAY ALERT AID
caffeine tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Rite Aid (014578892) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(11822-0226) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(11822-0226) | |