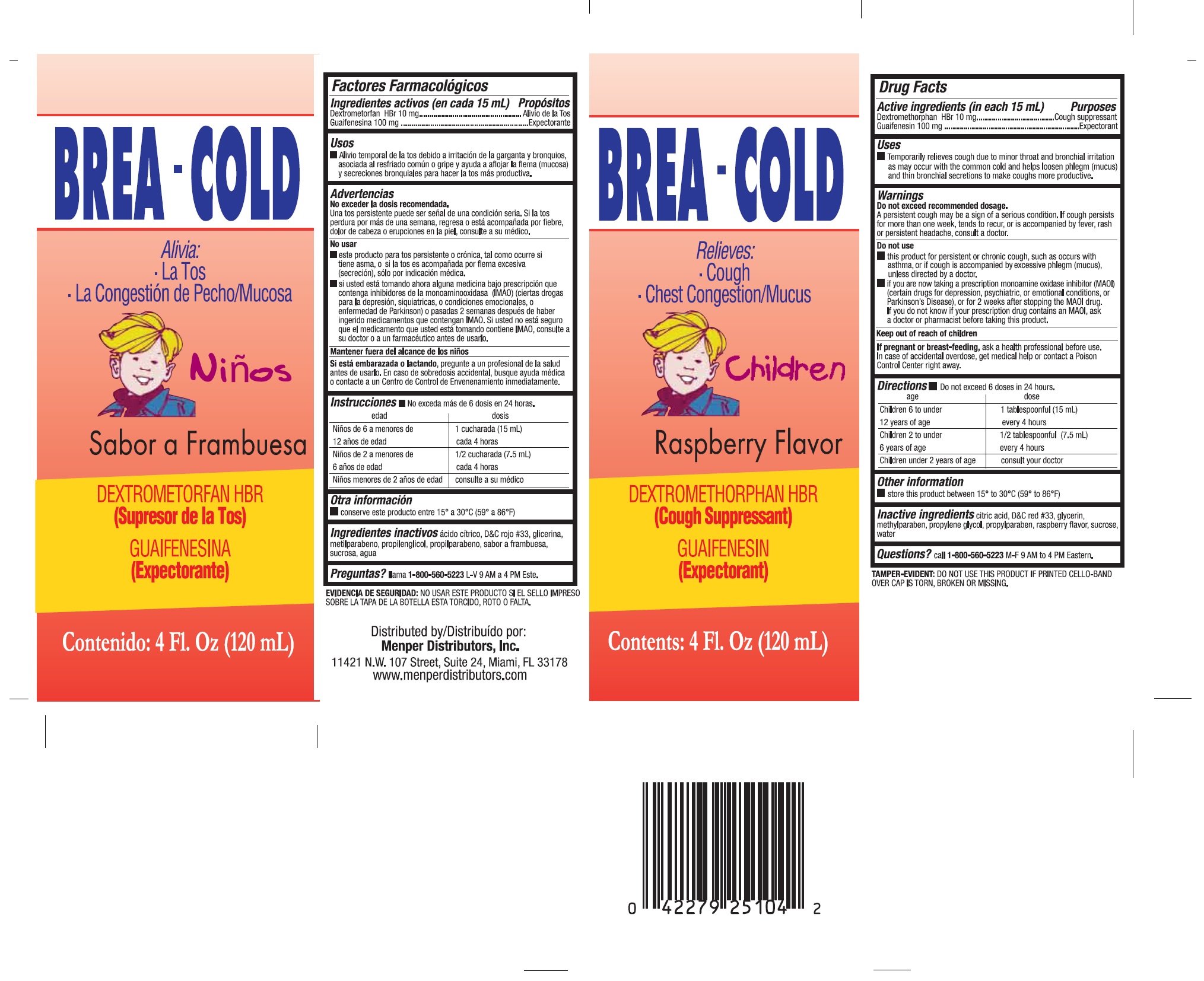
Label: BREACOLD CHILDREN- guaifenesin, dextromethorphan hbr liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 53145-007-04 - Packager: Menper Distributors, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 18, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each 15mL tsp)
- purpose
- Uses
- Warnings Do not exceed recommended dosage.
-
Do not use:
- This product for persistent or chronic cough, such as occurs with asthma, or if cough is accompanied by excessive phlegm (mucus), unless directed by a doctor.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- KEEP OUT OF REACH OF CHILDREN
- If you are pregnant or breast-feeding,
-
Directions
Do not exceed 6 doses in 24 hours.
Age Dose
Children 6 to under 1 teaspoonful (5 mL)
12 years of age..................................................... every 4 hours
Children 2 to under 1/2 teaspoonful (2.5 mL)
6 years of age....................................................... every 4 hours
Children under 2 years of age.................................. consult your doctor
- Other information
- Inactive ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BREACOLD CHILDREN
guaifenesin, dextromethorphan hbr liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53145-007 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 100 mg in 15 mL DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg in 15 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID ACETATE (UNII: DSO12WL7AU) D&C RED NO. 33 (UNII: 9DBA0SBB0L) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) RASPBERRY (UNII: 4N14V5R27W) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SUCROSE (UNII: C151H8M554) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53145-007-04 1 in 1 CARTON 02/21/2012 1 120 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 02/21/2012 Labeler - Menper Distributors, Inc. (101947166)