PAIN RELIEVER EXTRA STRENGTH- acetaminophen tablet, film coated
CARDINAL HEALTH
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Leader 44-531 Delisted
Uses
- temporarily relieves minor aches and pains due to:
- headache
- the common cold
- backache
- minor pain of arthritis
- muscular aches
- toothache
- premenstrual and menstrual cramps
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. The maximum daily dose of this product is 6 tablets (3,000 mg) in 24 hours. Severe liver damage may occur if you take:
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Stop use and ask a doctor if
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
-
do not take more than directed
(see overdose warning) - adults and children 12 years and over
- take 2 tablets every 6 hours while symptoms last
- do not take more than 6 tablets in 24 hours unless directed by a doctor
- do not take for more than 10 days unless directed by a doctor
- children under 12 years: do not use this adult extra strength product in children under 12 years of age; this will provide more than the recommended dose (overdose) of acetaminophen and may cause liver damage
Other information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, dextrose monohydrate, FD&C red #40, FD&C yellow #6, maltodextrin, povidone, sodium carboxymethylcellulose, sodium starch glycolate, stearic acid, sucralose, titanium dioxide
Principal display panel
NDC 37205-593-78
Compare to Extra Strength Tylenol® EZ Tabs
active ingredient*
LEADER®
EXTRA STRENGTH
Pain Reliever
Acetaminophen
Pain Reliever/Fever Reducer • Contains No Aspirin
Easy to Swallow
Sweet Coated
Easy Tabs
100 TABLETS - 500 mg EACH
Actual Size
*This product is not manufactured or distributed by McNeil Consumer Healthcare, owner of the registered trademark Extra Strength Tylenol® EZ Tabs.
50844 REV0913B53112
DISTRIBUTED BY CARDINAL HEALTH, DUBLIN, OHIO 43017
CIN 4527750 www.myleader.com 1-800-200-6313
All Leader® Brand products are 100% satisfaction guaranteed or return to place of purchase for a full refund.
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Leader44-531A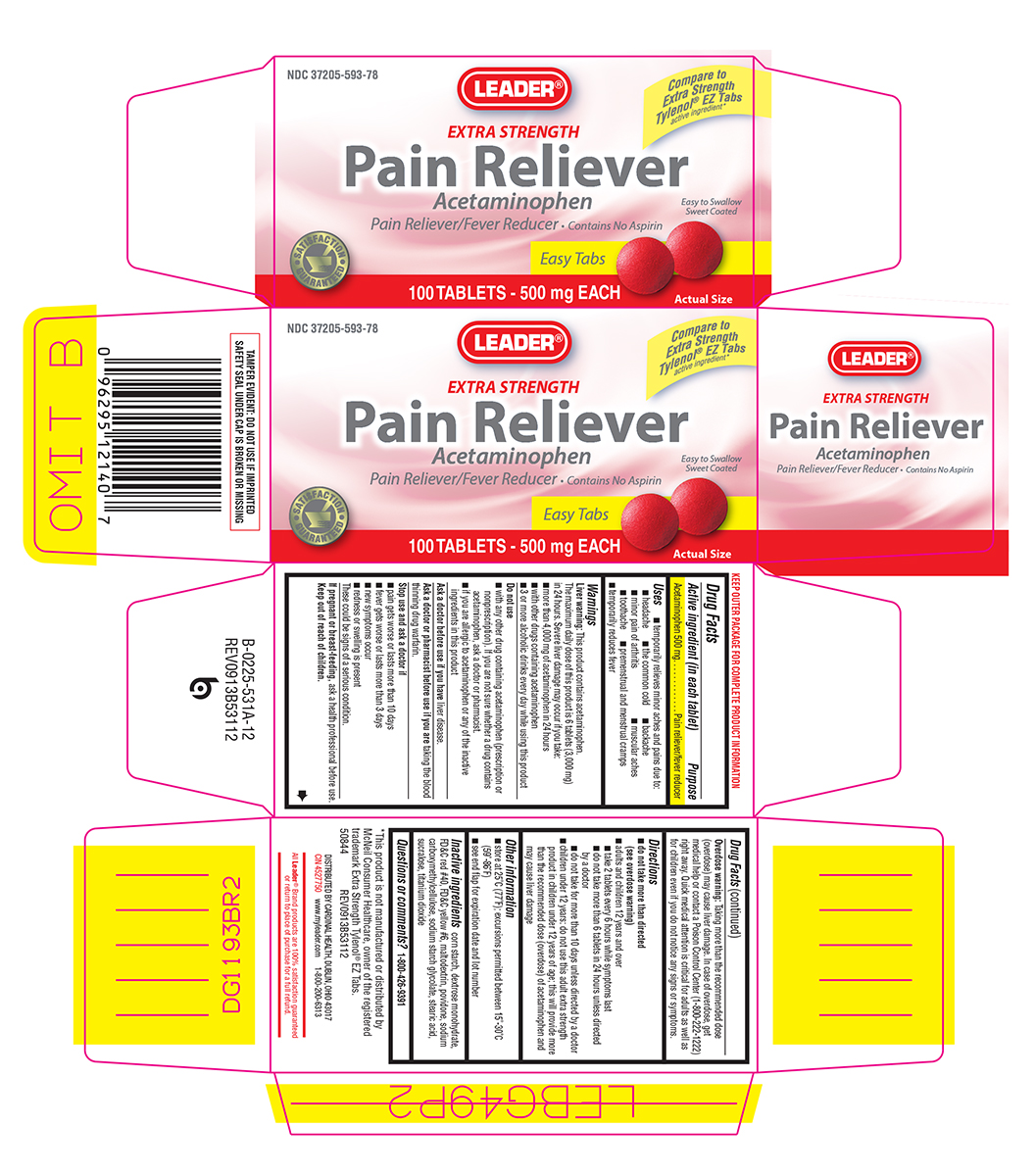
| PAIN RELIEVER
EXTRA STRENGTH
acetaminophen tablet, film coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - CARDINAL HEALTH (097537435) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(37205-593) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | MANUFACTURE(37205-593) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | PACK(37205-593) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | PACK(37205-593) | |