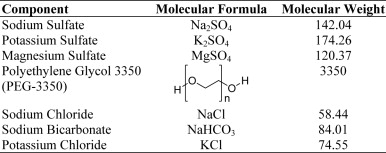
SUCLEAR- sodium sulfate, potassium sulfate, magnesium sulfate, polyethylene glycol 3350, sodium chloride, sodium bicarbonate and potassium chloride
Braintree Laboratories, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use SUCLEAR safely and effectively. See full prescribing information for SUCLEAR
SUCLEAR (sodium sulfate, potassium sulfate and magnesium sulfate oral solution; and PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution) Initial U.S. Approval: 2013 INDICATIONS AND USAGESuclear is a combination of osmotic laxatives and indicated for cleansing of the colon in preparation for colonoscopy in adults (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSCONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (≥2%) are overall discomfort, abdominal distension, abdominal pain, nausea, vomiting and headache (6). To report SUSPECTED ADVERSE REACTIONS, contact Braintree Laboratories, Inc. at 1-800-874-6756 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSSee 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 2/2013 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Suclear is indicated for cleansing of the colon as a preparation for colonoscopy in adults.
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Overview
There are two dosing regimens. Each requires two separate dosing times:
- Split-Dose (2-Day) [see Dosage and Administration (2.2)]
- Day-Before (1-Day) [see Dosage and Administration (2.3)].
Both Suclear dosing regimens require administration of Suclear using one 6-oz bottle of oral solution (containing sodium sulfate, potassium sulfate, and magnesium sulfate) diluted with water to 16-oz, followed by a 2-L jug of powder (polyethylene glycol 3350, sodium chloride, sodium bicarbonate and potassium chloride) dissolved in water. Additional fluids must be consumed in both dosing regimens [see Dosage and Administration (2.2, 2.3)]. Instruct patients to consume only clear liquids (no solid food or milk) and to avoid alcohol on the day before colonoscopy until after completion of the colonoscopy. [see Warnings and Precautions (5.1)].
2.2 Split-Dose (2-Day) Regimen (Preferred Method)
The Split-Dose regimen is the preferred dosing method. Instruct patients to take two separate doses in conjunction with fluids, as follows:
Dose 1 - Evening before the colonoscopy (10 to 12 hours prior to Dose 2)
- Dilute the 6-oz oral solution prior to use by pouring the entire contents of the bottle into the 16-oz mixing container and then filling the container with cool water to the fill line and mix
- Drink the entire solution in the container. It is best to complete drinking the solution within 20 minutes.
- Refill the container with 16 oz of water to the fill line and drink it over the next 2 hours.
- Refill the container with 16 oz of water to the fill line and finish drinking it before going to bed.
Dose 2 - Next morning on the day of the colonoscopy (start at least 3 ½ hours prior to colonoscopy)
- Dissolve the powder of Dose 2 by adding water to the fill line on the jug.
- Shake the jug until all the powder is dissolved. The solution can be used with or without the addition of a flavor pack. Flavor packs are available in Cherry, Lemon-Lime, Orange and Pineapple. When dissolved in water to a volume of 2 liters, the solution is clear and colorless. The solution may be refrigerated after adding water. The solution should be used within 48 hours of reconstitution.
- Using the 16-oz container provided with the kit, drink all the solution in the jug at a rate of one 16-oz container every 20 minutes (this is four 16-oz containers over a period of one and a half hours).
- Complete drinking the solution at least 2 hours before the colonoscopy.
- Consume only clear liquids until 2 hours prior to colonoscopy. Thereafter, nothing should be consumed until the completion of colonoscopy.
2.3 Day-Before (1-Day) Regimen (Alternative Method)
The Day-Before regimen is the alternative dosing method for patients for whom the Split-Dosing regimen is inappropriate. Instruct patients to take two separate doses in conjunction with fluids as follows:
On the evening before the colonoscopy:
Dose 1 (Begin at least 3 ½ hours prior to bedtime)
- Dilute the 6-oz oral solution prior to use by pouring the entire contents of the bottle into the 16-oz mixing container and then filling the container with cool water to the fill line and mix.
- Drink the entire solution in the container. It is best to complete drinking the solution within 20 minutes.
- Refill the container with 16 oz of water to the fill line and drink it over the next 2 hours.
Dose 2 (Approximately 2 hours after starting Dose 1)
- Dissolve the powder of Dose 2 by adding water to the fill line on the jug.
- Shake the jug until all the powder is dissolved. The solution can be used with or without the addition of a flavor pack. Flavor packs are available in Cherry, Lemon-Lime, Orange and Pineapple. When dissolved in water to a volume of 2 liters, the solution is clear and colorless. The solution may be refrigerated after adding water. The solution should be used within 48 hours of reconstitution.
- Using the 16-oz container provided with the kit, drink all the solution in the jug at a rate of one 16-oz container every 20 minutes (This is four 16-oz containers over a period of one and a half hours).
- Refill the container with 16 oz of water to the fill line and finish drinking it before going to bed.
- Consume only clear liquids until 2 hours prior to colonoscopy. Thereafter, nothing should be consumed until the completion of colonoscopy.
3 DOSAGE FORMS AND STRENGTHS
Oral solution: One 6-oz bottle of oral solution containing 17.5 g of sodium sulfate, 3.13 g of potassium sulfate, and 1.6 g of magnesium sulfate.
For oral solution: One 2-L bottle with powder for solution containing 210 g of polyethylene glycol 3350 (PEG-3350), 5.6 g of sodium chloride, 2.86 g of sodium bicarbonate and 0.74 g of potassium chloride. An optional 1 g flavoring ingredient may be added.
4 CONTRAINDICATIONS
Suclear is contraindicated in patients with the following:
- Gastrointestinal obstruction or ileus
- Bowel perforation
- Gastric retention
- Toxic colitis or toxic megacolon
- Known allergies to any components of Suclear [see Description (11)]
5 WARNINGS AND PRECAUTIONS
5.1 Serious Fluid and Serum Chemistry Abnormalities
Advise all patients to hydrate adequately before, during, and after the use of Suclear. If a patient develops significant vomiting or signs of dehydration after taking Suclear, perform post-colonoscopy lab tests (electrolytes, creatinine, and BUN). Fluid and electrolyte disturbances can lead to serious adverse reactions including cardiac arrhythmias, seizures and renal impairment.
Patients with electrolyte abnormalities should have them corrected before treatment with Suclear. Consider the risks of prescribing Suclear in patients with certain medical conditions that predispose them (or who are using medications that increase the risk of) fluid and electrolyte disturbances or that may increase the risk of seizure, arrhythmias, and renal impairment. Use of alcohol may increase the risk of dehydration.
5.2 Cardiac Arrhythmias
There have been rare reports of serious arrhythmias associated with the use of osmotic laxative products for bowel preparation. Carefully consider the risks of prescribing Suclear for patients at increased risk of arrhythmias (e.g., patients with a history of prolonged QT, uncontrolled arrhythmias, recent myocardial infarction, unstable angina, congestive heart failure, or cardiomyopathy). Consider pre-dose and post-colonoscopy ECGs in patients at increased risk of serious cardiac arrhythmias.
5.3 Seizures
There have been reports of generalized tonic-clonic seizures associated with use of bowel preparation products in patients with no prior history of seizures. The seizure cases were associated with electrolyte abnormalities (e.g., hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) and low serum osmolality.
Consider the risks of prescribing Suclear in patients with a history of seizures and in patients at increased risk of seizure, such as patients taking medications that lower the seizure threshold (e.g., tricyclic antidepressants), patients withdrawing from alcohol or benzodiazepines, or patients with known or suspected hyponatremia.
5.4 Use in Patients with Renal Impairment
Consider the risks of prescribing Suclear in patients with impaired renal function or patients taking concomitant medications that may affect renal function (such as diuretics, angiotensin converting enzyme inhibitors, angiotensin receptor blockers, or non-steroidal anti-inflammatory drugs). Advise these patients of the importance of adequate hydration before and after the use of Suclear. Consider performing baseline and post-colonoscopy laboratory tests in these patients (e.g., electrolytes, creatinine, and BUN).
5.5 Colonic Mucosal Ulcerations and Ischemic Colitis
Osmotic laxatives may produce colonic mucosal aphthous ulcerations, and there have been reports of more serious cases of ischemic colitis requiring hospitalization. Concurrent use of stimulant laxatives and Suclear may increase these risks. The potential for mucosal ulcerations resulting from the bowel preparation should be considered when interpreting colonoscopy findings in patients with known or suspect inflammatory bowel disease.
5.6 Use in Patients with Significant Gastrointestinal Disease
If gastrointestinal obstruction or perforation is suspected, perform appropriate diagnostic studies to rule out these conditions before administering Suclear.
Patients with severe active ulcerative colitis may be at increased risk of exacerbation of their disease with Suclear.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
In randomized, multicenter, controlled clinical trials, the effectiveness of Suclear for colon cleansing was compared with two separate preparations containing two liters of polyethylene glycol plus electrolytes (Studies 1 and 2) [see Clinical Studies (14)]. Suclear and the comparator preparation were administered entirely the day before the colonoscopy in Study 1, whereas they were administered as split doses in Study 2. In addition to collection of spontaneously reported adverse reactions, patients were instructed to complete a symptom scale questionnaire that targeted expected adverse reactions (overall discomfort, stomach cramping, stomach bloating, and nausea). Patients also were asked separately to report vomiting episodes.
Table 1 presents the most common adverse reactions in Study 1 and Study 2.
|
Adverse Reaction | Study 1 (Day-Before Regimen) | Study 2 (Split-Dose Regimen) | ||
|---|---|---|---|---|
| Suclear (N=176) n (%) | Comparator 1†
(N=190) n (%) | Suclear (N=186) n (%) | Comparator 2‡
(N=185) n (%) |
|
|
#Include spontaneously reported adverse reactions as well as those reported in a symptom scale questionnaire. |
||||
|
*Abdominal pain also included reports of upper abdominal pain and abdominal tenderness. |
||||
|
†Comparator 1: PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution; and two bisacodyl 5-mg delayed-release tablets. |
||||
|
‡Comparator 2: PEG-3350, sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid for oral solution. |
||||
| Overall discomfort# | 122 (69) | 108 (57) | 116 (62) | 121 (65) |
| Abdominal distension# | 92 (52) | 85 (45) | 96 (52) | 112 (61) |
| Abdominal pain#* | 71 (40) | 78 (41) | 70 (38) | 79 (43) |
| Nausea# | 74 (42) | 75 (40) | 86 (46) | 72 (39) |
| Vomiting# | 19 (11) | 15 (8) | 26 (14) | 13 (7) |
| Headache | 1 (1) | 3 (2) | 3 (2) | 2 (1) |
Electrolyte Abnormalities
Table 2 presents the incidence of new laboratory abnormalities in Study 1 and Study 2.
| Laboratory Parameter (direction of change) | Study 1: Day-Before Regimen | Study 2: Split-Dose Regimen | ||
|---|---|---|---|---|
| Suclear % (n/N) | Comparator 1†
% (n/N) | Suclear % (n/N) | Comparator 2‡
% (n/N) |
|
|
†Comparator 1: PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution; and two bisacodyl 5-mg delayed-release tablets. |
||||
|
‡Comparator 2: PEG-3350, sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid for oral solution. |
||||
|
*Anion gap was calculated using the following formula: [Sodium (mEq/L)] - [Chloride (mEq/L)] + [Bicarbonate (mEq/L)]. Normal range was defined as 12±4, and high was defined as a calculated value >16. |
||||
|
**eGFR was calculated using the Cockcroft-Gault formula. |
||||
| Anion gap (high)* | 3% (5/155) | 5% (8/170) | 10% (17/166) | 8% (12/155) |
| Bicarbonate (low) | 3% (4/156) | 3% (5/167) | 4% (6/170) | 12% (20/161) |
| Bilirubin, total (high) | 8% (12/157) | 10% (17/170) | 11% (19/170) | 4% (6/162) |
| Calcium (high) | 9% (12/139) | 4% (5/139) | 4% (6/141) | 5% (7/144) |
| Chloride (low) | 1% (1/157) | 0% (0/171) | 1% (1/173) | 0% (0/163) |
| Creatinine (high) | 2% (3/145) | 3% (4/155) | 1% (2/167) | 1% (1/153) |
| Creatine kinase (high) | 7% (10/138) | 4% (6/151) | 7% (10/147) | 5% (7/143) |
| eGFR (low)** | 25% (21/84) | 14% (12/88) | 9% (9/101) | 11% (9/85) |
| Glucose (high) | 7% (10/146) | 3% (4/156) | 2% (3/160) | 3% (5/150) |
| Magnesium (low) | 1% (1/158) | 1% (1/169) | 0% (0/169) | 1% (1/163) |
| Osmolality (high) | 2% (3/139) | 5% (8/153) | 4% (6/151) | 8% (12/145) |
| Phosphate (low) | 0% (0/155) | 0% (0/168) | 3% (5/171) | 1% (2/160) |
| Potassium (low) | 4% (5/144) | 3% (4/160) | 4% (6/162) | 4% (7/159) |
| Sodium (low) | 0% (0/157) | 0% (0/169) | 1% (1/169) | 1% (1/163) |
| Uric acid (high) | 6% (8/143) | 7% (11/160) | 4% (7/163) | 3% (4/154) |
7 DRUG INTERACTIONS
7.1 Drugs That May Increase Risks Due to Fluid and Electrolyte Abnormalities
Certain medical conditions and concomitant medications may increase the risk for developing fluid and electrolyte disturbances or may increase the risk of seizure, arrhythmias, and prolonged QT in the setting of fluid and electrolyte abnormalities when taking Suclear. This includes patients receiving drugs which may be associated with hypokalemia (such as diuretics or corticosteroids, or drugs where hypokalemia is a particular risk, such as cardiac glycosides) or hyponatremia. Use caution when Suclear is used in patients on nonsteroidal anti-inflammatory drugs (NSAIDS) or drugs known to induce Antidiuretic Hormone Secretion (SIADH), such as tricyclic antidepressants, selective serotonin re-uptake inhibitors, antipsychotic drugs and carbamazepine, as these drugs may increase the risk of water retention and/or electrolyte imbalance. Consider additional patient evaluations as appropriate [see Warnings and Precautions (5)].
8 USE IN SPECIFIC POPULATIONS
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Suclear is administered to a nursing woman.
8.4 Pediatric Use
The safety and effectiveness of Suclear in pediatric patients have not been established.
8.5 Geriatric Use
Of the 362 patients who received Suclear in clinical trials, 90 (25%) were 65 years of age or older, and 29 (8%) were 75 years of age or older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
The safety of Suclear in patients with renal impairment has not been adequately evaluated. Due to increased risk for electrolyte abnormalities in this population and a potential for reduced clearance of drugs in this setting, consider the risks of using Suclear in patients with renal impairment [see Warnings and Precautions (5.4), Clinical Pharmacology (12.3)].
11 DESCRIPTION
Suclear (sodium sulfate, potassium sulfate and magnesium sulfate; and PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride) is an osmotic laxative that includes one 6-oz bottle of oral solution and one 2-L bottle of powder for oral solution.
The 6-oz bottle of oral solution contains 17.5 g of sodium sulfate, 3.13 g of potassium sulfate, and 1.6 g of magnesium sulfate. Inactive ingredients include sodium benzoate, NF, sucralose, malic acid, FCC, citric acid, USP, flavoring ingredients and purified water, USP. The solution is a clear to slightly hazy liquid. The solution is clear and colorless when diluted to a final volume of 16-oz with water. Each kit also contains a 16-oz mixing container.
The 2-L bottle contains a white powder for oral solution containing 210 g of PEG 3350, NF, 5.6 g of sodium chloride, USP, 2.86 g of sodium bicarbonate, USP and 0.74 g of potassium chloride, USP. Inactive ingredients include 1 g of an optional flavor ingredient. Flavor packs are available in Cherry, Lemon-Lime, Orange and Pineapple. The preparation can be used with or without the addition of a flavor pack. When dissolved in water to a volume of 2 L, the solution is isosmotic, clear and colorless.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The primary mode of action of Suclear is the osmotic effect of the unabsorbed polyethylene glycol (PEG) and sulfate salts. Sulfate salts provide sulfate anions, which are poorly absorbed. The osmotic effect of unabsorbed sulfate anions and the associated cations causes water to be retained within the gastrointestinal tract.
PEG is also a largely unabsorbed osmotic agent which causes water to be retained within the gastrointestinal tract.
12.2 Pharmacodynamics
The osmotic effect of the unabsorbed PEG or sulfate ions, when ingested, produces a copious watery diarrhea.
12.3 Pharmacokinetics
Oral sulfates: Following oral administration of oral sulfate solution, approximately 20% of dose undergoes systemic absorption, with the remainder of the dose excreted in feces. Renal elimination appears to be the predominant route of clearance for absorbed sulfate. After administration of Suclear (only the 6-oz bottle of sulfate salts) to six healthy volunteers, the time at which serum sulfate reached its highest point (Tmax) was approximately 5.5 hours after the first dose of sulfates. Serum sulfate levels declined with a half-life of 8.5 hours.
PEG3350: The pharmacokinetics of PEG3350 following administration of Suclear were not assessed. Available pharmacokinetic information for oral PEG3350 suggests that it is poorly absorbed, primarily excreted in feces, and, to the extent it is absorbed it is eliminated in urine.
Pharmacokinetic Studies in Patients with Hepatic Impairment or Renal Impairment
The disposition of sulfate after ingestion of one 6-oz bottle of the Suclear sulfate solution was studied in patients (N=6) with mild-moderate hepatic impairment [Child-Pugh grades A (n = 5) and B (n = 1)] and in patients (N=6) with moderate renal impairment (creatinine clearance of 30 to 49 mL/min). Following administration of one 6-oz dose of sulfates, the renal impairment group had the highest serum sulfate levels, followed by the hepatic impairment group, and then by healthy subjects. Renal impairment resulted in 43% higher mean Cmax than healthy subjects. Urinary excretion of sulfate over 30 hours in subjects who received two 6-oz doses of sulfates was similar between hepatic patients and normal volunteers, but was approximately 16% lower in moderate renal impairment patients than in healthy volunteers. The mean sulfate levels of all three groups returned to their respective baseline levels by Day 6 after dose initiation.
PK for PEG3350 at the recommended dose was not assessed in patients with organ impairment.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of Suclear. Studies to evaluate the possible impairment of fertility or mutagenic potential of Suclear have not been performed.
13.2 Animal Toxicology and/or Pharmacology
The sulfate salts of sodium, potassium, and magnesium contained in Suclear were administered orally (gavage) to rats and dogs up to 28 days up to a maximum daily dose of 5 g/kg/day (approximately 1.8 and 6 times for rats and dogs, respectively, the recommended human dose of 22 g/day or 0.44 g/kg based on the body surface area). In rats, the sulfate salts caused diarrhea and electrolyte and metabolic changes, including hypochloremia, hypokalemia, hyponatremia, lower serum osmolality, and high serum bicarbonate. Significant renal changes included increased fractional sodium excretion, increased urinary sodium and potassium excretion, and alkaline urine in both males and females. In addition, creatinine clearance was significantly decreased in females at the highest dose. No microscopic renal changes were seen. In dogs, the sulfate salts caused emesis, excessive salivation, excessive drinking of water, abnormal excreta (soft and/or mucoid feces and/or diarrhea) and increased urine pH and sodium excretion.
14 CLINICAL STUDIES
The colon cleansing efficacy of Suclear was evaluated in two randomized, single-blind (colonoscopist only), active-controlled, multicenter studies (Studies 1 and 2) in patients scheduled to undergo an elective colonoscopy. A total of 737 adult patients were included in the efficacy analysis: 366 from Study 1 and 371 from Study 2. Patients ranged in age from 21 to 86 years (mean age 57 years), and 54% were female. Self-identified race distribution was as follows: 82% White, 10% Black, and 8% other.
Patients randomized to Suclear in the two studies were treated with one of two dosing regimens:
- In Study 1, Suclear was given by “Day-Before” (evening before) dosing, where both doses were taken sequentially on the day before the colonoscopy, with the first dose taken at approximately 6:00 PM, followed by one 16-oz container (provided with the kit) of water, and the second dose taken 2 hours after starting the first dose. Patients were recommended to drink at least one additional 16-oz container of water on the evening prior to colonoscopy.
The comparator for Study 1 was a preparation containing two liters of PEG plus electrolytes (sodium chloride, sodium bicarbonate, and potassium chloride) for oral solution and two 5-mg bisacodyl tablets, administered the day before the procedure. All patients in both the Suclear and comparator group were limited to a clear liquid diet on the day before the procedure until after completion of the colonoscopy.
- In Study 2, Suclear was given by “Split-Dose” (evening before and day of) dosing, where the first dose was taken the evening before the colonoscopy (at approximately 6:00 PM), followed by one 16-oz container of water (with a recommendation to drink at least one additional 16-oz container of water on the evening prior to colonoscopy), and the second dose was taken the morning of the colonoscopy (at approximately 6:00 AM). The second dose had to be completed at least 2 hours prior to colonoscopy. Patients in the Suclear group were limited to a clear liquid diet on the day before the procedure until after completion of the colonoscopy.
The comparator for Study 2 was a preparation containing two liters of PEG plus electrolytes (sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid) for oral solution, administered as split doses. Patients receiving Suclear were limited to a clear liquid diet on the day before the procedure until completion of the colonoscopy. Patients receiving the comparator were allowed to have a normal breakfast, a light lunch, clear soup and/or plain yogurt for dinner the day prior to colonoscopy. They were limited to a clear liquid diet from the time the preparation was started until after completion of the colonoscopy.
The primary efficacy endpoint in both studies was the proportion of patients with successful colon cleansing as assessed by the colonoscopists, who were not informed about the type of preparation received. Successful colon cleansing was defined as bowel preparations that were graded excellent (no more than small bits of adherent feces/fluid) or good (small amounts of feces or fluid not interfering with the exam) by the colonoscopist. Failed colon cleansing was defined as bowel preparations that were graded fair (enough feces or fluid to prevent a completely reliable exam) or poor (large amounts of fecal residue, additional cleansing required). The proportion of patients with successful colon cleansing was numerically similar between treatment groups for both studies. See Tables 3 and 4.
| Suclear | Comparator 1‡ | Difference between treatment groups | |||
|---|---|---|---|---|---|
| % (n/N) | 95% CI | % (n/N) | 95% CI | Difference | 95% CI |
|
†Successful colon cleansing was defined as excellent (no more than small bits of adherent feces/fluid) or good (small amounts of feces or fluid not interfering with the exam) by the blinded colonoscopist. |
|||||
|
‡Comparator 1: PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution; and two bisacodyl 5-mg delayed-release tablets. |
|||||
| 90% (158/176) | (84%, 94%) | 84% (157/188) | (77%, 89%) | 6% | (-1%, 13%) |
|
†Successful colon cleansing was defined as excellent (no more than small bits of adherent feces/fluid) or good (small amounts of feces or fluid not interfering with the exam) by the blinded colonoscopist. |
|||||
|
‡Comparator 2: PEG-3350, sodium sulfate, sodium chloride, potassium chloride, sodium ascorbate, and ascorbic acid for oral solution. |
|||||
| Suclear | Comparator 2‡ | Difference between treatment groups | |||
| % (n/N) | 95% CI | % (n/N) | 95% CI | Difference | 95% CI |
| 94% (173/185) | (89%, 97%) | 94% (173/185) | (89%, 97%) | 0% | (-5%, 5%) |
16 HOW SUPPLIED/STORAGE AND HANDLING
Suclear NDC 52268-901-01
Each Suclear kit contains:
- One 6-oz (177-mL) bottle of oral solution
- One 16-oz mixing container
- One 2-L bottle with powder for oral solution
- Four flavor packs (1 gram each Cherry, Lemon-Lime, Orange and Pineapple flavors)
17 PATIENT COUNSELING INFORMATION
See FDA-Approved Patient Labeling (Medication Guide).
- Ask patients if they have trouble swallowing or are prone to regurgitation or aspiration. Patients with impaired gag reflex and patients prone to regurgitation or aspiration should be observed during administration of Suclear solution.
- Instruct patients that each bottle needs to be diluted in water before ingestion and that they need to drink additional water according to the instructions. Direct ingestion of the undiluted solution may increase the risk of nausea, vomiting, and dehydration.
- Inform patients that oral medications may not be absorbed properly if they are taken within one hour of starting each dose of Suclear.
- Tell patients not to take other laxatives while they are taking Suclear.
Distributed by Braintree Laboratories, Inc. Braintree, MA 02185
Medication Guide
SuclearTM (Soo-kleer)
(sodium sulfate, potassium sulfate and magnesium sulfate oral solution; and PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution)
Read this Medication Guide before you start taking Suclear. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is the most important information I should know about Suclear?
Suclear can cause serious side effects, including:
Serious loss of body fluid (dehydration) and changes in blood salts (electrolytes) in your blood. These changes can cause:
- abnormal heartbeats that can cause death
- seizures. This can happen even if you have never had a seizure.
- kidney problems
Your risk of having fluid loss and changes in blood salts with Suclear is higher if you:
- have heart problems
- have kidney problems
- take water pills (diuretics) or non-steroidal anti-inflammatory drugs (NSAIDS)
Tell your healthcare provider right away if you have any of these symptoms of a loss of too much body fluid (dehydration) while taking Suclear:
- vomiting that prevents you from keeping down the additional prescribed amount of water listed in the Instructions for Use in the “Patient Instructions for Use” booklet
- dizziness
- urinating less often than normal
- headache
See “What are the possible side effects of Suclear?” for more information about side effects.
What is Suclear?
Suclear is a prescription medicine used by adults to clean the colon before a colonoscopy. Suclear cleans your colon by causing you to have diarrhea. Cleaning your colon helps your healthcare provider see the inside of your colon more clearly during your colonoscopy.
It is not known if Suclear is safe and effective in children.
Who should not take Suclear?
Do not take Suclear if your healthcare provider has told you that you have:
- a blockage in your intestine (bowel obstruction)
- an opening in the wall of your stomach or intestine (bowel perforation)
- problems with food and fluid emptying from your stomach (gastric retention)
- a very dilated intestine (toxic megacolon)
- an allergy to any of the ingredients in Suclear. See the end of this leaflet for a complete list of ingredients in Suclear.
What should I tell my healthcare provider before taking Suclear?
Before you take Suclear, tell your healthcare provider if you:
- have heart problems
- have a history of seizures
- have kidney problems
- have ulcerative colitis
- have stomach or bowel problems
- have problems with swallowing or gastric reflux
- have gout
- are withdrawing from drinking alcohol
- have a low blood salt (sodium) level
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Suclear will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Suclear passes into your breast milk. You and your healthcare provider should decide if you will take Suclear while breastfeeding.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Suclear may affect how other medicines work. Medicines taken by mouth may not be absorbed properly when taken within 1 hour before the start of each dose of Suclear.
Especially tell your healthcare provider if you take:
- medicines for blood pressure or heart problems
- medicines for kidney problems
- medicines for seizures
- water pills (diuretics)
- non-steroidal anti-inflammatory medicines (NSAID) pain medicine s
- laxatives
- medicines for depression or mental health problems
Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure if you are taking any of the medicines listed above.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take Suclear?
See the Instructions for Use in the “Patient Instructions for Use” booklet for dosing instructions. You must read, understand, and follow these instructions to take Suclear the right way.
- Take Suclear exactly as your healthcare provider tells you to take it. Your healthcare provider will prescribe the Split-Dosing option or the Day-Before Dosing option, depending on the scheduling of your colonoscopy.
- Suclear comes in a kit. The contents of each bottle in the Suclear Kit must be mixed with water (diluted) before drinking. Do not take Suclear oral solution or powder that has not been mixed with water, it may increase your risk of nausea, vomiting, and fluid loss (dehydration). It is important for you to drink the additional prescribed amount of water listed in the Instructions for Use to help prevent fluid loss (dehydration).
- Do not take other laxatives while taking Suclear.
-
Do not eat solid foods or drink milk or alcohol on the day before your colonoscopy and until after your colonoscopy. Drink only clear liquids:
- the day before your colonoscopy
- while taking Suclear
- after taking Suclear until 2 hours before your colonoscopy.
Do not eat or drink anything 2 hours before your colonoscopy.
- Drink clear liquids before, during, and after you take Suclear to avoid fluid loss (dehydrated). Examples of clear liquids are:
- water
- clear fruit juices without pulp including apple, white grape, or white cranberry
- strained limeade or lemonade
- coffee or tea (Do not use any dairy or non-dairy creamer)
- clear broth
- clear soda
- gelatin (without added fruit or topping)
- popsicles without pieces of fruit or fruit pulp
Do not eat or drink anything colored red or purple.
What are the possible side effects of Suclear?
Suclear can cause serious side effects, including:
- See “What is the most important information I should know about Suclear?”
-
changes in certain blood tests. Your healthcare provider may do blood tests after you take Suclear to check your blood for changes. Tell your healthcare provider if you have any symptoms of too much fluid loss, including:
- vomiting
- nausea
- bloating
- dizziness
- stomach (abdominal) cramping
- headache
- urinate less than usual
- trouble drinking clear liquid
- ulcers of the bowel or bowel problems (ischemic colitis). Tell your healthcare provider right away if you have severe stomach-area (abdomen) pain or rectal bleeding.
The most common side effects of Suclear include:
- discomfort
- stomach-area (abdomen) bloating, pain, and tenderness
- nausea
- vomiting
- headache
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Suclear. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Suclear?
- Store Suclear at room temperature, between 68ºF to 77°F (20ºC to 25°C).
Keep Suclear and all medicines out of the reach of children.
General information about the safe and effective use of Suclear.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Suclear for a condition for which it was not prescribed. Do not give Suclear to other people, even if they are going to have the same procedure you are. It may harm them.
This Medication Guide summarizes the most important information about Suclear. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Suclear that is written for health professionals.
For more information, go to www.braintreelabs.com or call 1-800-874-6756.
What are the ingredients in Suclear?
Active ingredients:
Oral solution: sodium sulfate, potassium sulfate, and magnesium sulfate.
Powder for solution: polyethylene glycol 3350, sodium chloride, sodium bicarbonate, potassium chloride.
Inactive ingredients:
Oral solution: sodium benzoate, sucralose, malic acid, citric acid, flavoring ingredients, purified water.
Powder for solution: Cherry, Lemon-Lime, Orange and Pineapple flavor packs.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Braintree Laboratories, Inc.
Braintree, MA 02185, USA
Issued: 01/2013
Principal Display Panel – Dose 1 6oz Bottle Label
NDC #52268-902-01 For use with Suclear™ kit only
DOSE 1
(sodium sulfate, potassium sulfate,
magnesium sulfate) Oral Solution
(17.5g/3.13g/1.6g)
Dispense the enclosed Medication Guide to each patient.
This bottle contains 6 ounces (177 mL) of liquid bowel prep.
See complete instructions in the enclosed booklet or on the box
before using.
DIRECTIONS:
Dilute the solution concentration prior to use.
See enclosed booklet for complete dosage
and administration instructions.
Keep this and other drugs out of
reach of children. Store at 25°C
(77°F); excursions permitted to
15-30°C (59-86°F).
Distributed by Braintree Laboratories, Braintree, MA Rx only Rev January, 2013
Principal Display Panel – Dose 2 2L Bottle Label
NDC #52268-903-01
For use with Suclear™ kit only
DOSE 2
(PEG-3350, sodium chloride, sodium bicarbonate
and potassium chloride) for Oral Solution
Dispense the enclosed Medication Guide to each patient.
See complete instructions in the enclosed booklet or on the box
before using.
DIRECTIONS: add 1 flavor pack. Add drinking water to the top
of the fill line on the bottle. Shake. Drink one (16-oz) glass of solution
every 20 minutes. Drink all the solution.
Distributed by Braintree Laboratories, Braintree, MA Rx only Rev January, 2013
Principal Display Panel – Flavor Packs Labels
Pineapple
FLAVOR
PACK
For use with Suclear only.
Net Wt. 1.0 g Rev. 1/13
Lemon-
Lime
FLAVOR
PACK
For use with Suclear only.
Net Wt. 1.0 g Rev. 1/13
Orange
FLAVOR
PACK
For use with Suclear only.
Net Wt. 1.0 g Rev. 1/13
Cherry
FLAVOR
PACK
For use with Suclear only.
Net Wt. 1.0 g Rev. 1/13
Principal Display Panel - Carton Label
NDC #52268-901-01
Dispense the enclosed Medication Guide to each patient.
SuclearTM
(sodium sulfate, potassium sulfate, and magnesium sulfate oral solution; and PEG-3350, sodium chloride, sodium bicarbonate and potassium chloride for oral solution)
THIS CARTON CONTAINS:
Full Prescribing Information and one Patient Instruction Booklet with the Medication Guide
DOSE 1
One 6-ounce (177 mL) bottle of sodium sulfate 17.5 g, potassium sulfate 3.13 g, and magnesium sulfate 1.6 g Oral Solution
One 16-ounce mixing cup
DOSE 2
One 2-liter bottle of PEG-3350 210 g, sodium bicarbonate 2.86 g, sodium chloride 5.6 g, and potassium chloride 0.74 g for Oral Solution
DILUTE AS DIRECTED PRIOR TO USE
NOTE: both doses are required for a complete prep.
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F).
Rev January, 2013
Rx only
Braintree LABORATORIES INC
| SUCLEAR
sodium sulfate, potassium sulfate, magnesium sulfate, polyethylene glycol 3350, sodium chloride, sodium bicarbonate and potassium chloride kit |
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
|
|||||||||||||||||||||||
| Labeler - Braintree Laboratories, Inc. (107904591) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Braintree Laboratories, Inc. | 617357954 | MANUFACTURE(52268-901, 52268-902, 52268-903) , ANALYSIS(52268-901, 52268-902, 52268-903) , LABEL(52268-901, 52268-902, 52268-903) | |