COLD AND ALLERGY- phenylephrine hcl and chlorpheniramine maleate tablet
Chain Drug Consortium
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
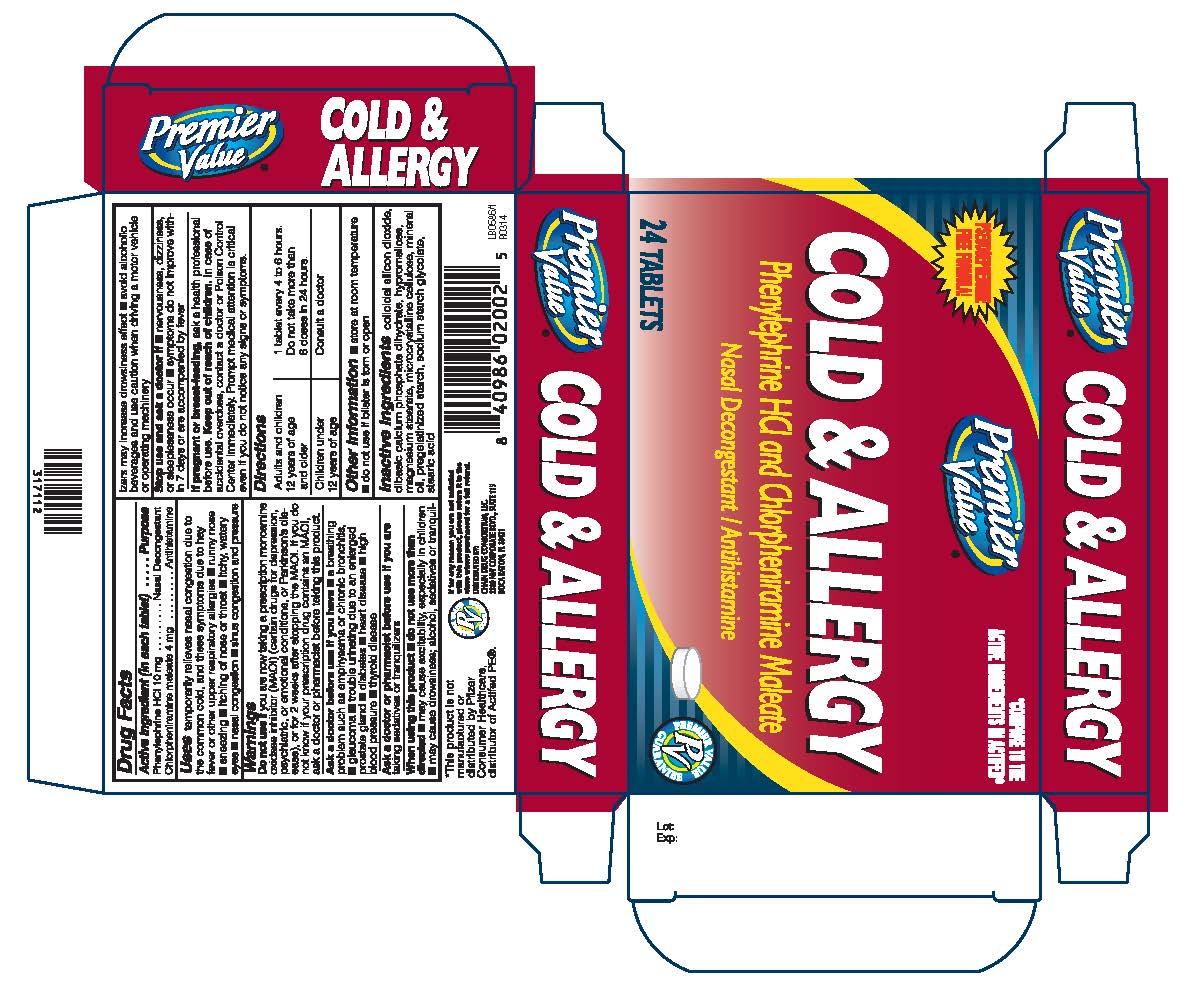
Drug Facts
Uses
temporarily relieves nasal congestion due to the common cold, and these symptoms due to hay fever or other upper respiratory allergiesrunny nose ■ sneezing ■ itching of nose or throat ■ itchy, watery
eyes ■ nasal congestion ■ sinus congestion and pressure
Warnings
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
a breathing problem such as emphysema or chronic bronchitis ■ glaucoma trouble urinating due to an enlarged prostate gland ■diabetes heart diseasehigh blood pressurethyroid disease
When using this product
do not use more than directed ■ may cause excitability, especially in children ■ may cause
drowsiness; alcohol, sedatives or tranquilzers may increase drowsiness effect ■avoid alcoholic beverages and use caution when driving a motor vehicle or operating machinery
Keep Out of Reach of Children
In case of accidental overdose, contact a doctor or Poison Control Center immediately. Prompt medical attention is critical even if you do not notice any signs or symptoms
Directions
Adults and children 12 years of age and older: 1 tablet every 4 to 6 hours. do not take more than 6 doses in 24 hours
Children under 12 years of age: Consult a doctor
| COLD AND ALLERGY
phenylephrine hcl and chlorpheniramine maleate tablet |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Chain Drug Consortium (101668460) |
| Registrant - Chain Drug Consortium (101668460) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| A&Z Pharmaceutical, Inc. | 926820705 | label(68016-233) , manufacture(68016-233) , pack(68016-233) , relabel(68016-233) , repack(68016-233) | |