Label: HYDROXYZINE PAMOATE capsule
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-1854-0, 54868-1854-1, 54868-1854-3, 54868-2892-0, view more54868-2892-3, 54868-2892-4, 54868-2892-5, 54868-2892-6 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0185-0613, 0185-0615
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 25, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
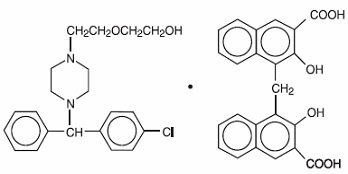
Hydroxyzine pamoate is a light yellow, practically odorless powder practically insoluble in water and methanol and freely soluble in dimethylformamide. It is chemically designated as (±)-2-[2-[4-(p-Chloro-α-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol 4,4’-methylenebis[3-hydroxy-2-naphthoate] (1:1) [10246-75-0] and can be structurally represented as follows:
C21H27CIN2O2•C23H16O6
M.W. 763.27
Each capsule, for oral administration, contains hydroxyzine pamoate equivalent to hydroxyzine hydrochloride 25 mg or 50 mg. In addition, each capsule contains the following inactive ingredients: colloidal silicon dioxide, D&C yellow No. 10, FD&C green No. 3, FD&C yellow No. 6, gelatin, hydroxypropyl cellulose, lactose monohydrate, magnesium stearate, propylene glycol, sodium lauryl sulfate, sodium starch glycolate and titanium dioxide.
-
CLINICAL PHARMACOLOGY
Hydroxyzine pamoate capsules are unrelated chemically to the phenothiazines, reserpine, meprobamate or the benzodiazepines.
Hydroxyzine pamoate capsules are not a cortical depressant, but their action may be due to a suppression of activity in certain key regions of the subcortical area of the central nervous system. Primary skeletal muscle relaxation has been demonstrated experimentally. Bronchodilator activity and antihistaminic and analgesic effects have been demonstrated experimentally and confirmed clinically.
An antiemetic effect, both by the apomorphine test and the veriloid test, has been demonstrated. Pharmacological and clinical studies indicate that hydroxyzine in therapeutic dosage does not increase gastric secretion or acidity and in most cases has mild antisecretory activity.
Hydroxyzine is rapidly absorbed from the gastrointestinal tract and hydroxyzine pamoate capsules clinical effects are usually noted within 15 to 30 minutes after oral administration.
-
INDICATIONS
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested.
Useful in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses and in histamine-mediated pruritus.
As a sedative when used as premedication and following general anesthesia, hydroxyzine may potentiate meperidine (Demerol®) and barbiturates, so their use in pre-anesthetic adjunctive therapy should be modified on an individual basis. Atropine and other belladonna alkaloids are not affected by the drug. Hydroxyzine is not known to interfere with the action of digitalis in any way and it may be used concurrently with this agent.
The effectiveness of hydroxyzine as an antianxiety agent for long term use, that is, more than 4 months, has not been assessed by systematic clinical studies. The physician should reassess periodically the usefulness of the drug for the individual patient.
-
CONTRAINDICATIONS
Hydroxyzine, when administered to the pregnant mouse, rat and rabbit, induced fetal abnormalities in the rat and mouse at doses substantially above the human therapeutic range. Clinical data in human beings are inadequate to establish safety in early pregnancy. Until such data are available, hydroxyzine is contraindicated in early pregnancy.
Hydroxyzine pamoate is contraindicated for patients who have shown a previous hypersensitivity to any component of this medication.
- WARNINGS
-
PRECAUTIONS
THE POTENTIATING ACTION OF HYDROXYZINE MUST BE CONSIDERED WHEN THE DRUG IS USED IN CONJUNCTION WITH CENTRAL NERVOUS SYSTEM DEPRESSANTS SUCH AS NARCOTICS, NON-NARCOTIC ANALGESICS AND BARBITURATES. Therefore, when central nervous system depressants are administered concomitantly with hydroxyzine their dosage should be reduced.
Since drowsiness may occur with use of the drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery while taking hydroxyzine pamoate capsules. Patients should be advised against the simultaneous use of other CNS depressant drugs and cautioned that the effect of alcohol may be increased.
Geriatric Use
A determination has not been made whether controlled clinical studies of hydroxyzine pamoate capsules included sufficient numbers of subjects aged 65 and over to define a difference in response from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function and of concomitant disease or other drug therapy.
The extent of renal excretion of hydroxyzine pamoate capsules has not been determined. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selections.
Sedating drugs may cause confusion and over sedation in the elderly; elderly patients generally should be started on low doses of hydroxyzine pamoate capsules and observed closely.
-
ADVERSE REACTIONS
Side effects reported with the administration of hydroxyzine pamoate capsules are usually mild and transitory in nature.
Central Nervous System
Drowsiness is usually transitory and may disappear in a few days of continued therapy or upon reduction of the dose. Involuntary motor activity including rare instances of tremor and convulsions have been reported, usually with doses considerably higher than those recommended. Clinically significant respiratory depression has not been reported at recommended doses.
In post-marketing experience, the following additional undesirable effects have been reported:
Body as a Whole: allergic reaction
Nervous System: headache
Psychiatric: hallucination
Skin and Appendages: pruritus, rash, urticaria
-
OVERDOSAGE
The most common manifestation of overdosage of hydroxyzine pamoate capsules is hypersedation. Other reported signs and symptoms were convulsions, stupor, nausea and vomiting. As in the management of overdosage with any drug, it should be borne in mind that multiple agents may have been taken.
If vomiting has not occurred spontaneously, it should be induced. Immediate gastric lavage is also recommended. General supportive care, including frequent monitoring of the vital signs and close observation of the patient, is indicated. Hypotension, though unlikely, may be controlled with intravenous fluids and vasopressors.
Do not use epinephrine as hydroxyzine counteracts its pressor action. Caffeine and Sodium Benzoate Injection USP, may be used to counteract central nervous system depressant effects.
There is no specific antidote. It is doubtful that hemodialysis would be of any value in the treatment of overdosage with hydroxyzine. However, if other agents such as barbiturates have been ingested concomitantly, hemodialysis may be indicated. There is no practical method to quantitate hydroxyzine in body fluids or tissue after its ingestion or administration.
-
DOSAGE AND ADMINISTRATION
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested: in adults, 50 mg to 100 mg q.i.d.; children under 6 years, 50 mg daily in divided doses; and over 6 years, 50 mg to 100 mg daily in divided doses.
For use in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses and in histamine-mediated pruritus: in adults: 25 mg t.i.d. or q.i.d.; children under 6 years, 50 mg daily in divided doses; and over 6 years, 50 mg to 100 mg daily in divided doses.
As a sedative when used as a premedication and following general anesthesia: 50 mg to 100 mg in adults and 0.6 mg/kg in children. When treatment is initiated by the intramuscular route of administration, subsequent doses may be administered orally.
As with all medications, the dosage should be adjusted according to the patient’s response to therapy.
-
HOW SUPPLIED
Hydroxyzine Pamoate Capsules USP for oral administration are available as:
25 mg: (equivalent to 25 mg hydroxyzine hydrochloride) are light green/dark green capsules imprinted “E 613” and supplied as:
Bottles of 15
NDC 54868-2892-4
Bottles of 20
NDC 54868-2892-5
Bottles of 30
NDC 54868-2892-3
Bottles of 100
NDC 54868-2892-0
Bottles of 120
NDC 54868-2892-6
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure, as required.
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Manufactured for
Sandoz Inc.
Princeton, NJ 08540
Manufactured by
Epic Pharma, LLC
Laurelton, NY 11413
Rev. 09/09
MF0613REV09/09
OS7127
Sandoz Inc.
Princeton, NJ 08540
OS8905
Rev. 09/09
Distributed by:
Physicians Total Care, Inc.
Tulsa, OK 74146
- HydrOXYzine Pamoate Capsules USP, 25 mg Capsules
- HydrOXYzine Pamoate Capsules USP, 50 mg Capsules
-
INGREDIENTS AND APPEARANCE
HYDROXYZINE PAMOATE
hydroxyzine pamoate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-2892(NDC:0185-0613) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROXYZINE PAMOATE (UNII: M20215MUFR) (HYDROXYZINE - UNII:30S50YM8OG) HYDROXYZINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color GREEN (light green/dark green) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code E613 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-2892-0 100 in 1 BOTTLE 2 NDC:54868-2892-3 30 in 1 BOTTLE, PLASTIC 3 NDC:54868-2892-4 15 in 1 BOTTLE, PLASTIC 4 NDC:54868-2892-5 20 in 1 BOTTLE, PLASTIC 5 NDC:54868-2892-6 120 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA087479 02/09/2000 HYDROXYZINE PAMOATE
hydroxyzine pamoate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-1854(NDC:0185-0615) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROXYZINE PAMOATE (UNII: M20215MUFR) (HYDROXYZINE - UNII:30S50YM8OG) HYDROXYZINE HYDROCHLORIDE 50 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color GREEN (white) Score no score Shape CAPSULE Size 19mm Flavor Imprint Code E615 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-1854-0 100 in 1 BOTTLE 2 NDC:54868-1854-1 30 in 1 BOTTLE, PLASTIC 3 NDC:54868-1854-3 60 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA086183 05/14/1996 06/30/2012 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel(54868-2892, 54868-1854) , repack(54868-2892, 54868-1854)