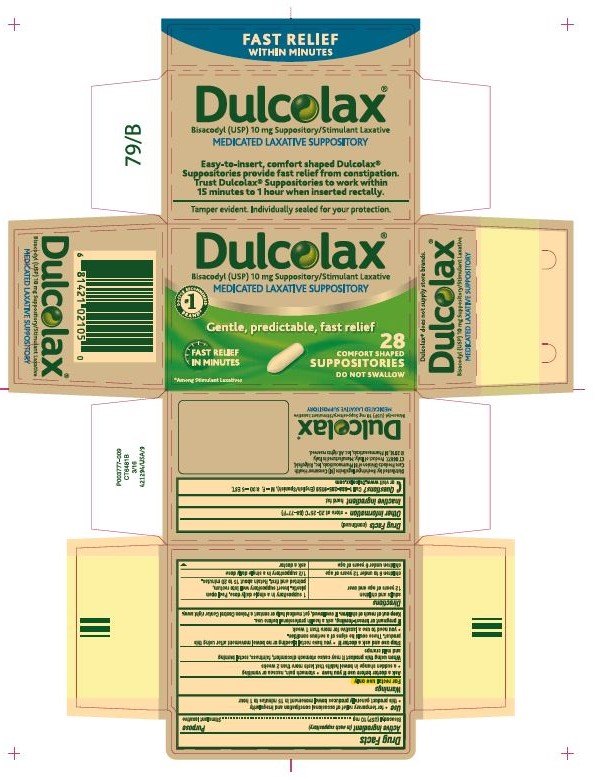
DULCOLAX- bisacodyl suppository
Boehringer Ingelheim Pharmaceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
| Active ingredient (in each suppository) | Purpose |
| Bisacodyl (USP) 10 mg .................................... | Stimulant laxative |
Use
- for temporary relief of occasional constipation and irregularity
- this product generally produces bowel movement in 15 minutes
to 1 hour
Warnings
For rectal use only
Ask a doctor before use if you have
- stomach pain, nausea or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
When using
this product it may cause stomach discomfort, faintness,
rectal burning and mild cramps
Stop use and ask a doctor if
- you have rectal bleeding or no bowel movement after using
this product. These could be signs of a serious condition.
- you need to use a laxative for more than 1 week
If pregnant or
breast-feeding, ask a health professional before
use.
Keep out of reach
of children. If swallowed, get medical help or contact
a Poison Control Center right away.
Directions
| adults and children
12 years of age and over | 1 suppository in
a single daily dose. Peel open plastic. Insert suppository well into
rectum, pointed end first. Retain about 15 to 20 minutes. |
| children 6 to under
12 years of age | 1/2 suppository
in a single daily dose |
| children under
6 years of age | ask a doctor |
Other information
- store at 20-25°C (68-77°F)
Inactive ingredient hard fat
Questions? call 1-888-285-9159 (English/Spanish), M – F, 8:30 – 5 EST, or
visit www.Dulcolax.com
Distributed by: Boehringer Ingelheim (BI)
Consumer Health Care Products Division of BI Pharmaceuticals, Inc.,
Ridgefield, CT 06877. Product of Italy. Manufactured in Italy. © 2016,
BI Pharmaceuticals, Inc. All rights reserved.
Dulcolax® 10 mg Suppository Carton
NDC: 0597-0052-28
Boehringer Ingelheim Pharmaceuticals, Inc.