SSD- silver sulfadiazine cream
Par Pharmaceutical Inc.
----------
SSD™ (1% Silver Sulfadiazine Cream)
SSD AF™ (1% Silver Sulfadiazine Cream)
DESCRIPTION
SSD (1% Silver Sulfadiazine) Cream and SSD AF (1% Silver Sulfadiazine) Cream are topical antibacterial preparations which have as their active antimicrobial ingredient silver sulfadiazine. The active moiety is contained within an opaque, white, water miscible cream base.
Each 1000 grams of SSD/SSD AF Cream contains 10 grams of silver sulfadiazine
Inactive Ingredients: cetyl alcohol (SSD Cream only), isopropyl myristate, polyoxyl 40 stearate, propylene glycol, purified water, stearyl alcohol, sodium hydroxide, sorbitan monooleate, white petrolatum; with 0.3% methyl paraben, as a preservative.
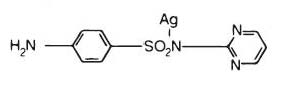
Silver sulfadiazine has an emprical formula of C10H9AgN4O2S, molecular weight of 357.14 and structural formula as shown:
CLINICAL PHARMACOLOGY
Silver sulfadiazine has broad antimicrobial activity. It is bactericidal for many gram-negative and gram-positive bacteria as well as being effective against yeast. Results from in vitro testing are listed below. Sufficient data have been obtained to demonstrate that silver sulfadiazine will inhibit bacteria that are resistant to other antimicrobial agents and that the compound is superior to sulfadiazine.
Studies utilizing radioactive micronized silver sulfadiazine, electron microscopy, and biochemical techniques have revealed that the mechanism of action of silver sulfadiazine on bacteria differs from silver nitrate and sodium sulfadiazine. Silver sulfadiazine acts only on the cell wall to produce its bactericidal effect.
|
Genus and Species |
50 ug/mL |
100 ug/mL |
|
Pseudomonas aeruginosa |
130/130 |
130/130 |
|
Pseudomonas maltophilia |
7/7 |
7/7 |
|
Enterobacter species |
48/50 |
50/50 |
|
Enterobacter cloacae |
24/24 |
24/24 |
|
Klebsiella species |
53/54 |
54/54 |
|
Escherichia coli |
63/63 |
63/63 |
|
Serratia species |
27/28 |
28/28 |
|
Proteus mirabilis |
53/53 |
53/53 |
|
Morganella morganii |
10/10 |
10/10 |
|
Providencia rettgeri |
2/2 |
2/2 |
|
Proteus vulgaris |
2/2 |
2/2 |
|
Providencia species |
1/1 |
1/1 |
|
Citrobacter species |
10/10 |
10/10 |
|
Acinetobacter calcoaceticus |
10/11 |
11/11 |
|
Stahylococcus aureus |
100/101 |
101/101 |
|
Staphylococcus epidermidis |
51/51 |
51/51 |
|
B-Hemolytic Streptococcus |
4/4 |
4/4 |
|
Enterococcus species |
52/53 |
53/53 |
|
Corynebacterium diphtheriae |
2/2 |
2/2 |
|
Clostridium perfringens |
0/2 |
2/2 |
|
Candida albicans |
43/50 |
50/50 |
Silver sulfadiazine is not a carbonic anhydrase inhibitor and may be useful in situations where such agents are contraindicated.
INDICATIONS AND USAGE
Silver Sulfadiazine Cream is a topical antimicrobial drug indicated as an adjunct for the prevention and treatment of wound sepsis in patients with second and third degree burns.
CONTRAINDICATIONS
Silver Sulfadiazine Cream is contraindicated in patients who are hypersensitive to silver sulfadiazine or any of the other ingredients in the preparation.
Because sulfonamide therapy is known to increase the possibility of kernicterus, Silver Sulfadiazine Cream should not be used on pregnant women approaching or at term, on premature infants, or on newborn infants during the first 2 months of life.
WARNINGS
There is potential cross-sensitivity between silver sulfadiazine and other sulfonamides. If allergic reactions attributable to treatment with silver sulfadiazine occur, continuation of therapy must be weighed against the potential hazards of the particular allergic reaction.
Fungal proliferation in and below the eschar may occur. However, the incidence of clinically reported fungal superinfection is low.
The use of Silver Sulfadiazine Cream in some cases of glucose-6-phosphate dehydrogenase-deficient individuals may be hazardous, as hemolysis may occur.
PRECAUTIONS
General: If hepatic and renal functions become impaired and elimination of drug decreases, accumulation may occur and discontinuation of Silver Sulfadiazine Cream should be weighed against the therapeutic benefit being achieved.
In considering the use of topical proteolytic enzymes in conjunction with Silver Sulfadiazine Cream, the possibility should be noted that silver may inactivate such enzymes.
Laboratory Tests: In the treatment of burn wounds involving extensive areas of the body, the serum sulfa concentrations may approach adult therapeutic levels (8 to 12mg %). Therefore, in these patients it would be advisable to monitor serum sulfa concentrations. Renal function should be carefully monitored and the urine should be checked for sulfa crystals.
Absorption of the propylene glycol vehicle has been reported to affect serum osmolality, which may affect the interpretation of laboratory tests.
Carcinogenesis and Mutagenesis and Impairment of Fertility
Long-term dermal toxicity studies of 24 months duration in rats and 18 months in mice with concentrations of silver sulfadiazine three to ten times the concentration in Silver Sulfadiazine Cream revealed no evidence of carcinogenicity.
Pregnancy: Pregnancy Category B. A reproductive study has been performed in rabbits at doses up to three to ten times the concentration of silver sulfadiazine in Silver Sulfadiazine Cream and has revealed no evidence of harm to the fetus due to silver sulfadiazine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly justified, especially in pregnant women approaching or at term. (See CONTRAINDICATIONS)
Nursing Mothers
It is not known whether Silver Sulfadiazine Cream is excreted in human milk. However, sulfonamides are known to be excreted in human milk, and all sulfonamides derivatives are known to increase the possibility of kernicterus. Because of the possibility for serious adverse reactions in nursing infants from sulfonamides, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
ADVERSE REACTIONS
Several cases of transient leukopenia have been reported in patients receiving silver sulfadiazine therapy. Leukopenia associated with silver sulfadiazine administration is primarily characterized by decreased neutrophil count. Maximal white blood cell depression occurs within two to four days of initiation of therapy. Rebound to normal leukocyte levels follows onset within two to three days. Recovery is not influenced by continuation of silver sulfadiazine therapy. The incidence of leukopenia in various reports averages about 20%. A higher incidence has been seen in patients treated concurrently with cimetidine.
Other infrequently occurring events include skin necrosis, erythema multiforme, skin discoloration, burning sensation, rashes, and interstitial nephritis.
Reduction in bacterial growth after application of topical antibacterial agents has been reported to permit spontaneous healing of deep partial thickness burns by preventing conversion of the partial thickness to full thickness by sepsis. However, reduction in bacterial colonization has caused delayed separation, in some cases necessitating escharotomy in order to prevent contracture.
Absorption of silver sulfadiazine varies depending upon the percent of body surface area and the extent of the tissue damage. Although few have been reported, it is possible that any adverse reaction associated with sulfonamides may occur. Some of the reactions which have been associated with sulfonamides are as follows: blood dyscrasias, including agranulocytosis, aplastic anemia, thrombocytopenia, leucopenia and hemolytic anemia, dermatologic and and allergic reactions, including Stevens-Johnson syndrome and exfoliative dermatitis, gastrointestinal reactions, hepatitis and hepatocellular necrosis, CNS reactions, and toxic nephrosis.
DOSAGE AND ADMINISTRATION
FOR TOPICAL USE ONLY - NOT FOR OPHTHALMIC USE:
Prompt institution of appropriate regimens for care of the burned patient is of prime importance and includes the control of shock and pain. The burn wounds are then cleansed and debrided; and Silver Sulfadiazine Cream is applied under sterile conditions. The burn areas should be covered with Silver Sulfadiazine Cream at all times. The cream should be applied once to twice daily to a thickness of approximately 1/16 inch. Whenever necessary, the cream should be reapplied to any areas from which it has been removed due to patient activity. Administration may be accomplished in minimal time because dressings are not required. However, if individual patient requirements make dressings necessary, they may be used.
Reapply immediately after hydrotherapy.
Treatment with Silver Sulfadiazine Cream should be continued until satisfactory healing has occurred or until the burn site is ready for grafting. The drug should not be withdrawn from the therapeutic regimen while there remains the possibility of infection except if a significant adverse reaction occurs.
HOW SUPPLIED
SSD™ (1% Silver Sulfadiazine) Cream: white to off-white cream.
|
50 gram jar |
|
|
400 gram jar |
|
|
25 gram tube |
|
|
85 gram tube |
|
SSD AF™ (1% Silver Sulfadiazine) Cream: white to off-white cream.
|
400 gram jar |
|
Store at controlled room temperature 15° - 30°C (59° - 86°F)
SSD is a trademark of Dr. Reddy's Laboratories Louisiana, LLC
Manufactured for
Par Pharmaceutical Companies, Inc.
Spring Valley, NY 10977
Manufactured by
Dr. Reddy's Laboratories Louisiana, LLC
Shreveport, LA 71106
Rev. 05/10
| SSD
silver sulfadiazine cream |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| SSD
silver sulfadiazine cream |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Par Pharmaceutical Inc. (092733690) |
| Registrant - Dr. Reddy's Laboratories Inc. DBA Dr. Reddy's Laboratories Louisiana, LLC (830397282) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Dr. Reddy's Laboratories Inc. DBA Dr. Reddy's Laboratories Louisiana, LLC | 830397282 | MANUFACTURE(49884-600, 49884-601) | |