Label: ZITHROMAX- azithromycin dihydrate tablet, film coated
ZITHROMAX- azithromycin dihydrate powder, for suspension
- NDC Code(s): 0069-3051-01, 0069-3051-07, 0069-3051-75, 0069-3080-30
- Packager: Pfizer Laboratories Div Pfizer Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated September 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZITHROMAX safely and effectively. See full prescribing information for ZITHROMAX.
ZITHROMAX® (azithromycin) 600 mg tablets, for oral use
ZITHROMAX® (azithromycin) for oral suspension
Initial U.S. Approval: 1991RECENT MAJOR CHANGES
Warnings and Precautions, Cardiovascular Death (5.5)
11/2021
INDICATIONS AND USAGE
ZITHROMAX is a macrolide antibacterial indicated for mild to moderate infections caused by designated, susceptible bacteria:
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZITHROMAX and other antibacterial drugs, ZITHROMAX should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.3)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- •
- Serious (including fatal) allergic and skin reactions. Discontinue ZITHROMAX and initiate appropriate therapy if reaction occurs. (5.1)
- •
- Hepatotoxicity: Discontinue azithromycin immediately if signs and symptoms of hepatitis occur. (5.2)
- •
- Infantile Hypertrophic Pyloric Stenosis (IHPS): Following the use of azithromycin in neonates (treatment up to 42 days of life), IHPS has been reported. Direct parents and caregivers to contact their physician if vomiting or irritability with feeding occurs. (5.3)
- •
- Prolongation of QT interval and cases of torsades de pointes have been reported. This risk which can be fatal should be considered in patients with certain cardiovascular disorders including known QT prolongation or history torsades de pointes, those with proarrhythmic conditions, and with other drugs that prolong the QT interval. (5.4)
- •
- Cardiovascular Death: Some observational studies have shown an approximately two-fold increased short-term potential risk of acute cardiovascular death in adults exposed to azithromycin relative to other antibacterial drugs, including amoxicillin. Consider balancing this potential risk with treatment benefits when prescribing ZITHROMAX. (5.5)
- •
- Clostridioides difficile-Associated Diarrhea: Evaluate patients if diarrhea occurs. (5.6)
- •
- ZITHROMAX may exacerbate muscle weakness in persons with myasthenia gravis. (5.7)
ADVERSE REACTIONS
The most common adverse reactions are diarrhea (5%), nausea (3%), abdominal pain (3%), or vomiting, (no percent given). (6)
- To report SUSPECTED ADVERSE REACTIONS, contact Pfizer Inc at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 11/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Sexually Transmitted Diseases
1.2 Mycobacterial Infections
1.3 Usage
2 DOSAGE AND ADMINISTRATION
2.1 Sexually Transmitted Diseases
2.2 Mycobacterial Infections
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Hypersensitivity
4.2 Hepatic Dysfunction
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
5.2 Hepatotoxicity
5.3 Infantile Hypertrophic Pyloric Stenosis (IHPS)
5.4 QT Prolongation
5.5 Cardiovascular Death
5.6 Clostridioides difficile-Associated Diarrhea (CDAD)
5.7 Exacerbation of Myasthenia Gravis
5.8 Use in Sexually Transmitted Infections
5.9 Development of Drug-Resistant Bacteria
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
6.3 Laboratory Abnormalities
7 DRUG INTERACTIONS
7.1 Nelfinavir
7.2 Warfarin
7.3 Potential Drug-Drug Interaction with Macrolides
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology
14 CLINICAL STUDIES
14.1 Clinical Studies in Patients with Advanced HIV Infection for the Prevention and Treatment of Disease Due to Disseminated Mycobacterium avium Complex (MAC)
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
ZITHROMAX is a macrolide antibacterial drug indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the specific conditions listed below.
1.1 Sexually Transmitted Diseases
Non-gonococcal urethritis and cervicitis due to Chlamydia trachomatis
1.2 Mycobacterial Infections
Prophylaxis of Disseminated Mycobacterium avium complex (MAC) Disease
ZITHROMAX, taken alone or in combination with rifabutin at its approved dose, is indicated for the prevention of disseminated MAC disease in persons with advanced HIV infection [see Dosage and Administration (2)].
Treatment of Disseminated MAC Disease
ZITHROMAX, taken in combination with ethambutol, is indicated for the treatment of disseminated MAC infections in persons with advanced HIV infection [see Use in Specific Populations (8.4) and Clinical Studies (14.1)].
1.3 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZITHROMAX and other antibacterial drugs, ZITHROMAX should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
-
2 DOSAGE AND ADMINISTRATION
[see Indications and Usage (1)]
ZITHROMAX for oral suspension (single dose 1 g packet) can be taken with or without food after constitution. However, increased tolerability has been observed when tablets are taken with food.
Zithromax for oral suspension (single dose 1 g packet) is not for pediatric use. For pediatric suspension see the prescribing information for ZITHROMAX (azithromycin for oral suspension) 100 mg/5 mL and 200 mg/5 mL bottles.
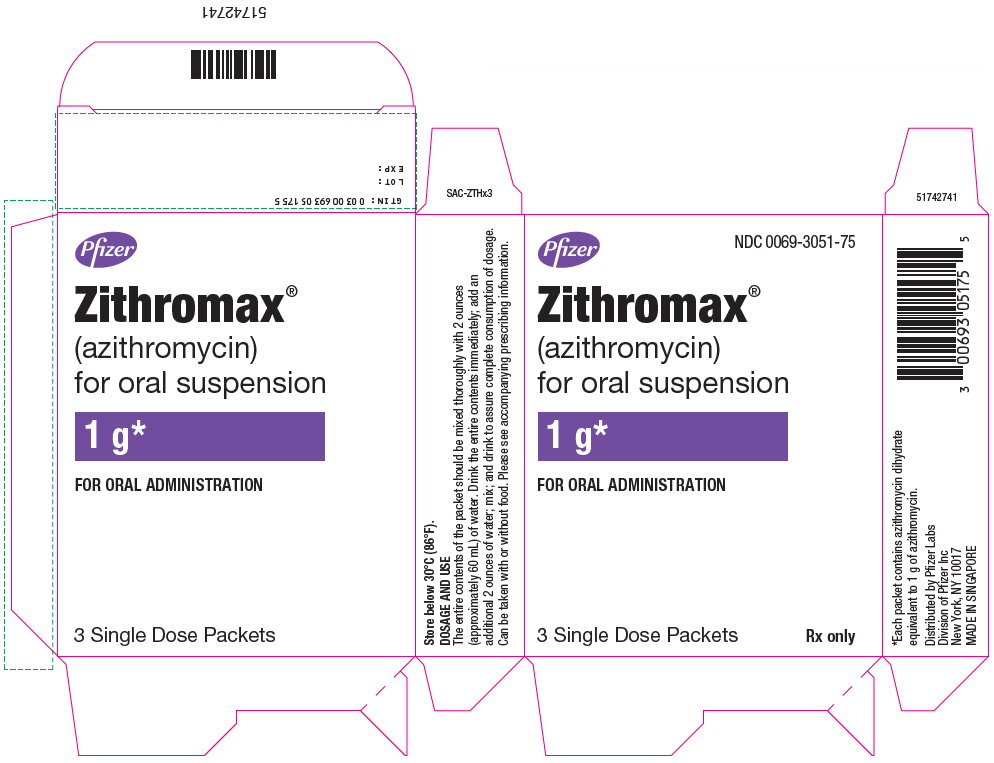
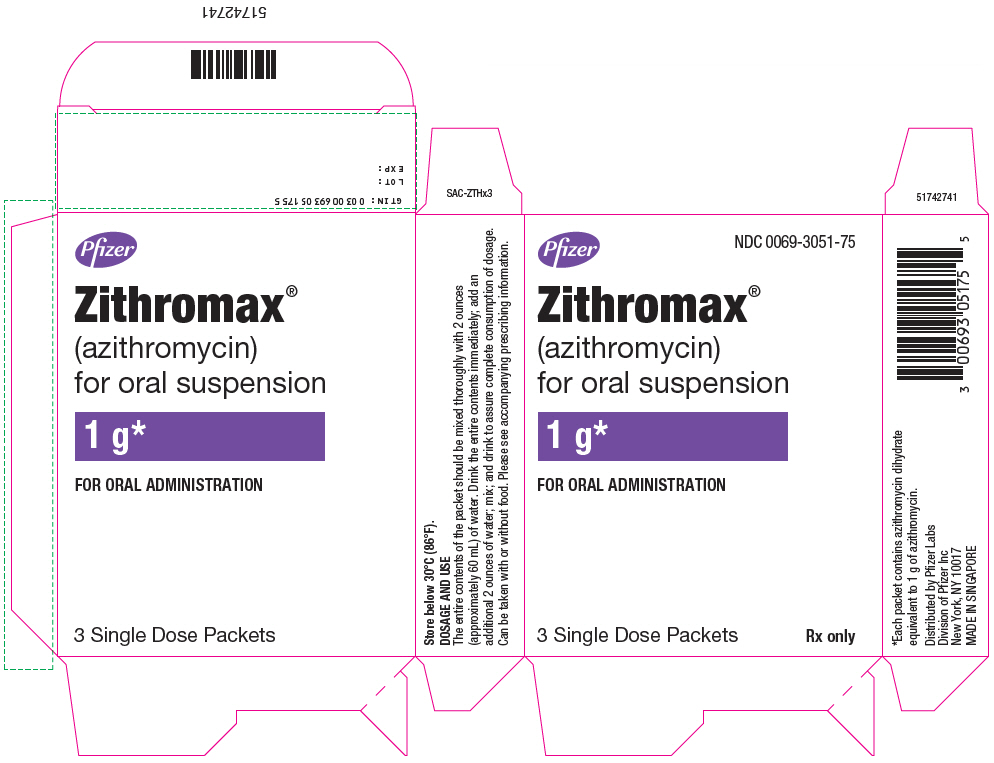
Directions for administration of ZITHROMAX for oral suspension in the single dose packet (1 g): The entire contents of the packet should be mixed thoroughly with two ounces (approximately 60 mL) of water. Drink the entire contents immediately; add an additional two ounces of water, mix, and drink to ensure complete consumption of dosage. The single dose packet should not be used to administer doses other than 1000 mg of azithromycin.
2.1 Sexually Transmitted Diseases
The recommended dose of ZITHROMAX for the treatment of non-gonococcal urethritis and cervicitis due to C. trachomatis is a single 1 gram (1000 mg) dose of ZITHROMAX. This dose can be administered as one single dose packet (1 g).
2.2 Mycobacterial Infections
Prevention of Disseminated MAC Infections
The recommended dose of ZITHROMAX for the prevention of disseminated Mycobacterium avium complex (MAC) disease is: 1200 mg taken once weekly. This dose of ZITHROMAX may be combined with the approved dosage regimen of rifabutin.
Treatment of Disseminated MAC Infections
ZITHROMAX should be taken at a daily dose of 600 mg, in combination with ethambutol at the recommended daily dose of 15 mg/kg. Other antimycobacterial drugs that have shown in vitro activity against MAC may be added to the regimen of azithromycin plus ethambutol at the discretion of the physician or health care provider.
-
3 DOSAGE FORMS AND STRENGTHS
ZITHROMAX 600 mg tablets (engraved on front with "PFIZER" and on back with "308") are supplied as white, modified oval-shaped, film-coated tablets containing azithromycin dihydrate equivalent to 600 mg azithromycin. These are packaged in bottles of 30 tablets.
ZITHROMAX for oral suspension 1000 mg/5 mL is supplied in single-dose packets containing azithromycin dihydrate equivalent to 1 gram of azithromycin.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Serious allergic reactions, including angioedema, anaphylaxis, and dermatologic reactions including Acute Generalized Exanthematous Pustulosis (AGEP), Stevens-Johnson Syndrome, and toxic epidermal necrolysis, have been reported rarely in patients on azithromycin therapy. [see Contraindications (4.1)]
Fatalities have been reported. Cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) have also been reported. Despite initially successful symptomatic treatment of the allergic symptoms, when symptomatic therapy was discontinued, the allergic symptoms recurred soon thereafter in some patients without further azithromycin exposure. These patients required prolonged periods of observation and symptomatic treatment. The relationship of these episodes to the long tissue half-life of azithromycin and subsequent prolonged exposure to antigen is presently unknown.
If an allergic reaction occurs, the drug should be discontinued and appropriate therapy should be instituted. Physicians should be aware that allergic symptoms may reappear when symptomatic therapy is discontinued.
5.2 Hepatotoxicity
Abnormal liver function, hepatitis, cholestatic jaundice, hepatic necrosis, and hepatic failure have been reported, some of which have resulted in death. Discontinue azithromycin immediately if signs and symptoms of hepatitis occur.
5.3 Infantile Hypertrophic Pyloric Stenosis (IHPS)
Following the use of azithromycin in neonates (treatment up to 42 days of life), IHPS has been reported. Direct parents and caregivers to contact their physician if vomiting or irritability with feeding occurs.
5.4 QT Prolongation
Prolonged cardiac repolarization and QT interval, imparting a risk of developing cardiac arrhythmia and torsades de pointes, have been seen with treatment with macrolides, including azithromycin. Cases of torsades de pointes have been spontaneously reported during postmarketing surveillance in patients receiving azithromycin. Providers should consider the risk of QT prolongation which can be fatal when weighing the risks and benefits of azithromycin for at-risk groups including:
- •
- patients with known prolongation of the QT interval, a history of torsades de pointes, congenital long QT syndrome, bradyarrhythmias or uncompensated heart failure
- •
- patients on drugs known to prolong the QT interval
- •
- patients with ongoing proarrhythmic conditions such as uncorrected hypokalemia or hypomagnesemia, clinically significant bradycardia, and in patients receiving Class IA (quinidine, procainamide) or Class III (dofetilide, amiodarone, sotalol) antiarrhythmic agents.
Elderly patients may be more susceptible to drug-associated effects on the QT interval.
5.5 Cardiovascular Death
Some observational studies have shown an approximately two-fold increased short-term potential risk of acute cardiovascular death in adults exposed to azithromycin relative to other antibacterial drugs, including amoxicillin. The five-day cardiovascular mortality observed in these studies ranged from 20 to 400 per million azithromycin treatment courses. This potential risk was noted to be greater during the first five days of azithromycin use and does not appear to be limited to those patients with preexisting cardiovascular diseases. The data in these observational studies are insufficient to establish or exclude a causal relationship between acute cardiovascular death and azithromycin use. Consider balancing this potential risk with treatment benefits when prescribing ZITHROMAX.
5.6 Clostridioides difficile-Associated Diarrhea (CDAD)
CDAD has been reported with use of nearly all antibacterial agents, including ZITHROMAX, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.7 Exacerbation of Myasthenia Gravis
Exacerbations of symptoms of myasthenia gravis and new onset of myasthenic syndrome have been reported in patients receiving azithromycin therapy.
5.8 Use in Sexually Transmitted Infections
ZITHROMAX, (single dose 1 g packet) at the recommended dose, should not be relied upon to treat gonorrhea or syphilis. Antibacterial agents used in high doses for short periods of time to treat non-gonococcal urethritis may mask or delay the symptoms of incubating gonorrhea or syphilis. All patients with sexually transmitted urethritis or cervicitis should have a serologic test for syphilis and appropriate cultures for gonorrhea performed at the time of diagnosis. Appropriate antibacterial therapy and follow-up tests for these diseases should be initiated if infection is confirmed.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in labeling:
- •
- Hypersensitivity [see Warnings and Precautions (5.1)]
- •
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- •
- Infantile Hypertrophic Pyloric Stenosis (IHPS) [see Warnings and Precautions (5.3)]
- •
- QT Prolongation [see Warnings and Precautions (5.4)]
- •
- Cardiovascular Death [see Warnings and Precautions (5.5)]
- •
- Clostridioides difficile-Associated Diarrhea (CDAD) [see Warnings and Precautions (5.6)]
- •
- Exacerbation of Myasthenia Gravis [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, most of the reported adverse reactions were mild to moderate in severity and were reversible upon discontinuation of the drug. Approximately 0.7% of the patients from the multiple-dose clinical trials discontinued ZITHROMAX (azithromycin) therapy because of treatment-related adverse reactions. Serious adverse reactions included angioedema and cholestatic jaundice. Most of the adverse reactions leading to discontinuation were related to the gastrointestinal tract, e.g., nausea, vomiting, diarrhea, or abdominal pain. [see Clinical Studies (14.2)]
Multiple-dose regimen
Overall, the most common adverse reactions in adult patients receiving a multiple-dose regimen of ZITHROMAX were related to the gastrointestinal system with diarrhea/loose stools (5%), nausea (3%), and abdominal pain (3%) being the most frequently reported.
No other adverse reactions occurred in patients on the multiple-dose regimen of ZITHROMAX with a frequency greater than 1%. Adverse reactions that occurred with a frequency of 1% or less included the following:
Cardiovascular: Palpitations and chest pain.
Gastrointestinal: Dyspepsia, flatulence, vomiting, melena, and cholestatic jaundice.
Genitourinary: Monilia, vaginitis, and nephritis.
Nervous System: Dizziness, headache, vertigo, and somnolence.
General: Fatigue.
Allergic: Rash, photosensitivity, and angioedema.
Chronic therapy with 1200 mg weekly regimen
The nature of adverse reactions seen with the 1200 mg weekly dosing regimen for the prevention of Mycobacterium avium infection in severely immunocompromised HIV-infected patients were similar to those seen with short-term dosing regimens. [see Clinical Studies (14)]
Chronic therapy with 600 mg daily regimen combined with ethambutol
The nature of adverse reactions seen with the 600 mg daily dosing regimen for the treatment of Mycobacterium avium complex infection in severely immunocompromised HIV-infected patients were similar to those seen with short term dosing regimens. Five percent of patients experienced reversible hearing impairment in the pivotal clinical trial for the treatment of disseminated MAC in patients with AIDS. Hearing impairment has been reported with macrolide antibiotics, especially at higher doses. Other treatment-related adverse reactions occurring in >5% of subjects and seen at any time during a median of 87.5 days of therapy include: abdominal pain (14%), nausea (14%), vomiting (13%), diarrhea (12%), flatulence (5%), headache (5%), and abnormal vision (5%). Discontinuations from treatment due to laboratory abnormalities or adverse reactions considered related to study drug occurred in 8 of 88 (9.1%) of subjects.
Single 1 gram dose regimen
Overall, the most common adverse reactions in patients receiving a single-dose regimen of 1 gram of ZITHROMAX were related to the gastrointestinal system and were more frequently reported than in patients receiving the multiple-dose regimen.
Adverse reactions that occurred in patients on the single 1 gram dosing regimen of ZITHROMAX with a frequency of 1% or greater included diarrhea/loose stools (7%), nausea (5%), abdominal pain (5%), vomiting (2%), dyspepsia (1%), and vaginitis (1%).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of azithromycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions reported with azithromycin during the postmarketing period in adult and/or pediatric patients for which a causal relationship may not be established include:
Allergic: Arthralgia, edema, urticaria, and angioedema.
Cardiovascular: Arrhythmias, including ventricular tachycardia, and hypotension. There have been reports of QT prolongation, torsades de pointes, and cardiovascular death.
Gastrointestinal: Anorexia, constipation, dyspepsia, flatulence, vomiting/diarrhea pseudomembranous colitis, pancreatitis, oral candidiasis, pyloric stenosis, and tongue discoloration.
General: Asthenia, paresthesia, fatigue, malaise, and anaphylaxis.
Genitourinary: Interstitial nephritis, acute renal failure, and vaginitis.
Hematopoietic: Thrombocytopenia.
Liver/Biliary: Abnormal liver function, hepatitis, cholestatic jaundice, hepatic necrosis, and hepatic failure. [see Warnings and Precautions (5.2)]
Nervous System: Convulsions, dizziness/vertigo, headache, somnolence, hyperactivity, nervousness, agitation, and syncope.
Psychiatric: Aggressive reaction and anxiety.
Skin/Appendages: Pruritus, and serious skin reactions including erythema multiforme, AGEP, Stevens-Johnson Syndrome, toxic epidermal necrolysis, and DRESS.
Special Senses: Hearing disturbances including hearing loss, deafness, and/or tinnitus, and reports of taste/smell perversion and/or loss.
6.3 Laboratory Abnormalities
Significant abnormalities (irrespective of drug relationship) occurring during the clinical trials were reported as follows:
- •
- With an incidence of 1–2%, elevated serum creatine phosphokinase, potassium, ALT (SGPT), GGT, and AST (SGOT).
- •
- With an incidence of less than 1%, leukopenia, neutropenia, decreased platelet count, elevated serum alkaline phosphatase, bilirubin, BUN, creatinine, blood glucose, LDH, and phosphate.
When follow-up was provided, changes in laboratory tests appeared to be reversible.
In multiple-dose clinical trials involving more than 3000 patients, 3 patients discontinued therapy because of treatment-related liver enzyme abnormalities and 1 because of a renal function abnormality.
In a phase 1 drug interaction study performed in normal volunteers, 1 of 6 subjects given the combination of azithromycin and rifabutin, 1 of 7 given rifabutin alone, and 0 of 6 given azithromycin alone developed a clinically significant neutropenia (<500 cells/mm3).
Laboratory abnormalities seen in clinical trials for the prevention of disseminated Mycobacterium avium disease in severely immunocompromised HIV-infected patients. [see Clinical Studies (14)]
Chronic therapy (median duration: 87.5 days, range: 1–229 days) that resulted in laboratory abnormalities in >5% of subjects with normal baseline values in the pivotal trial for treatment of disseminated MAC in severely immunocompromised HIV-infected patients treated with azithromycin 600 mg daily in combination with ethambutol include: a reduction in absolute neutrophils to <50% of the lower limit of normal (10/52, 19%) and an increase to five times the upper limit of normal in alkaline phosphatase (3/35, 9%). These findings in subjects with normal baseline values are similar when compared to all subjects for analyses of neutrophil reductions (22/75, 29%) and elevated alkaline phosphatase (16/80, 20%). Causality of these laboratory abnormalities due to the use of study drug has not been established.
-
7 DRUG INTERACTIONS
7.1 Nelfinavir
Co-administration of nelfinavir at steady-state with a single oral dose of azithromycin resulted in increased azithromycin serum concentrations. Although a dose adjustment of azithromycin is not recommended when administered in combination with nelfinavir, close monitoring for known adverse reactions of azithromycin, such as liver enzyme abnormalities and hearing impairment, is warranted. [see Adverse Reactions (6)]
7.2 Warfarin
Spontaneous postmarketing reports suggest that concomitant administration of azithromycin may potentiate the effects of oral anticoagulants such as warfarin, although the prothrombin time was not affected in the dedicated drug interaction study with azithromycin and warfarin. Prothrombin times should be carefully monitored while patients are receiving azithromycin and oral anticoagulants concomitantly.
7.3 Potential Drug-Drug Interaction with Macrolides
Interactions with digoxin, colchicine or phenytoin have not been reported in clinical trials with azithromycin. No specific drug interaction studies have been performed to evaluate potential drug-drug interaction. However, drug interactions have been observed with other macrolide products. Until further data are developed regarding drug interactions when digoxin, colchicine or phenytoin are used with azithromycin careful monitoring of patients is advised.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from published literature and postmarketing experience over several decades with azithromycin use in pregnant women have not identified any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes (see Data).
Developmental toxicity studies with azithromycin in rats, mice, and rabbits showed no drug-induced fetal malformations at doses up to 3, 2, and 1 times, respectively, an adult human daily dose of 600 mg based on body surface area. Decreased viability and delayed development were observed in the offspring of pregnant rats administered azithromycin from day 6 of pregnancy through weaning at a dose equivalent to 3 times an adult human daily dose of 600 mg based on body surface area (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Available data from published observational studies, case series, and case reports over several decades do not suggest an increased risk for major birth defects, miscarriage, or adverse maternal or fetal outcomes with azithromycin use in pregnant women. Limitations of these data include the lack of randomization and inability to control for confounders such as underlying maternal disease and maternal use of concomitant medications.
Animal Data
Azithromycin administered during the period of organogenesis did not cause fetal malformations in rats and mice at oral doses up to 200 mg/kg/day (moderately maternally toxic). Based on body surface area, this dose is approximately 3 (rats) and 2 (mice) times an adult human daily dose of 600 mg. In rabbits administered azithromycin at oral doses of 10, 20, and 40 mg/kg/day during organogenesis, reduced maternal body weight and food consumption were observed in all groups; no evidence of fetotoxicity or teratogenicity was observed at these doses, the highest of which is approximately equal to an adult human daily dose of 600 mg based on body surface area.
In a pre- and postnatal development study, azithromycin was administered orally to pregnant rats from day 6 of pregnancy until weaning at doses of 50 or 200 mg/kg/day. Maternal toxicity (reduced food consumption and body weight gain; increased stress at parturition) was observed at the higher dose. Effects in the offspring were noted at 200 mg/kg/day during the postnatal development period (decreased viability, delayed developmental landmarks). These effects were not observed in a pre- and postnatal rat study when up to 200 mg/kg/day of azithromycin was given orally beginning on day 15 of pregnancy until weaning.
8.2 Lactation
Risk Summary
Azithromycin is present in human milk (see Data). Non-serious adverse reactions have been reported in breastfed infants after maternal administration of azithromycin (see Clinical Considerations). There are no available data on the effects of azithromycin on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ZITHROMAX and any potential adverse effects on the breastfed infant from ZITHROMAX or from the underlying maternal condition.
Clinical Considerations
Advise women to monitor the breastfed infant for diarrhea, vomiting, or rash.
Data
Azithromycin breastmilk concentrations were measured in 20 women after receiving a single 2 g oral dose of azithromycin during labor. Breastmilk samples collected on days 3 and 6 postpartum as well as 2 and 4 weeks postpartum revealed the presence of azithromycin in breastmilk up to 4 weeks after dosing. In another study, a single dose of azithromycin 500 mg was administered intravenously to 8 women prior to incision for cesarean section. Breastmilk (colostrum) samples obtained between 12 and 48 hours after dosing revealed that azithromycin persisted in breastmilk up to 48 hours.
8.4 Pediatric Use
In controlled clinical studies, azithromycin has been administered to pediatric patients ranging in age from 6 months to 12 years. For information regarding the use of ZITHROMAX (azithromycin for oral suspension) in the treatment of pediatric patients, [see Indications and Usage (1) and Dosage and Administration (2)] of the prescribing information for ZITHROMAX (azithromycin for oral suspension) 100 mg/5 mL and 200 mg/5 mL bottles.
HIV-Infected Pediatric Patients: The safety and efficacy of azithromycin for the prevention or treatment of MAC in HIV-infected children have not been established. Safety data are available for 72 children 5 months to 18 years of age (mean 7 years) who received azithromycin for treatment of opportunistic infections. The mean duration of therapy was 242 days (range 3–2004 days) at doses of <1 to 52 mg/kg/day (mean 12 mg/kg/day). Adverse reactions were similar to those observed in the adult population, most of which involved the gastrointestinal tract. Treatment-related reversible hearing impairment in children was observed in 4 subjects (5.6%). Two (2.8%) children prematurely discontinued treatment due to adverse reactions: one due to back pain and one due to abdominal pain, hot and cold flushes, dizziness, headache, and numbness. A third child discontinued due to a laboratory abnormality (eosinophilia). The protocols upon which these data are based specified a daily dose of 10–20 mg/kg/day (oral and/or IV) of azithromycin.
8.5 Geriatric Use
In multiple-dose clinical trials of oral azithromycin, 9% of patients were at least 65 years of age (458/4949) and 3% of patients (144/4949) were at least 75 years of age. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Elderly patients may be more susceptible to development of torsades de pointes arrhythmias than younger patients. [see Warnings and Precautions (5.4)]
ZITHROMAX 600 mg tablets contain 2.1 mg of sodium per tablet. ZITHROMAX for oral suspension 1 gram single-dose packets contain 37.0 mg of sodium per packet.
Geriatric Patients with Opportunistic Infections, Including (MAC) Disease: Safety data are available for 30 patients (65–94 years old) treated with azithromycin at doses >300 mg/day for a mean of 207 days. These patients were treated for a variety of opportunistic infections, including MAC. The adverse reaction were generally similar to that seen in younger patients, except for a higher incidence of adverse reactions relating to the gastrointestinal system and to reversible impairment of hearing. [see Dosage and Administration (2)]
- 10 OVERDOSAGE
-
11 DESCRIPTION
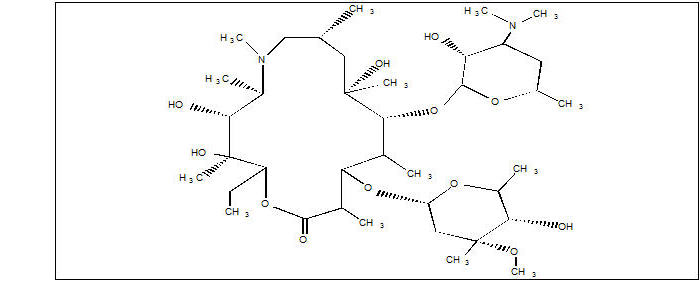
ZITHROMAX (azithromycin tablets and oral suspension) contains the active ingredient azithromycin, a macrolide antibacterial drug, for oral administration. Azithromycin has the chemical name (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-2-ethyl- 3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-11-[[3,4,6- trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy]-1-oxa-6-azacyclopentadecan-15-one. Azithromycin is derived from erythromycin; however, it differs chemically from erythromycin in that a methyl-substituted nitrogen atom is incorporated into the lactone ring. Its molecular formula is C38H72N2O12, and its molecular weight is 749.0. Azithromycin has the following structural formula:
Azithromycin, as the dihydrate, is a white crystalline powder with a molecular formula of C38H72N2O12∙2H2O and a molecular weight of 785.0.
ZITHROMAX tablets contain azithromycin dihydrate equivalent to 600 mg azithromycin. They also contain the following inactive ingredients: dibasic calcium phosphate anhydrous, pregelatinized starch, sodium croscarmellose, magnesium stearate, sodium lauryl sulfate, and an aqueous film coat consisting of hypromellose, titanium dioxide, lactose, and triacetin.
ZITHROMAX for oral suspension is supplied in a single-dose packet containing azithromycin dihydrate equivalent to 1 g azithromycin. It also contains the following inactive ingredients: colloidal silicon dioxide, sodium phosphate tribasic, anhydrous; spray dried artificial banana flavor, spray dried artificial cherry flavor, and sucrose.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
Based on animal models of infection, the antibacterial activity of azithromycin appears to correlate with the ratio of area under the concentration-time curve to minimum inhibitory concentration (AUC/MIC) for certain pathogens (S. pneumoniae and S. aureus). The principal pharmacokinetic/pharmacodynamic parameter best associated with clinical and microbiological cure has not been elucidated in clinical trials with azithromycin.
Cardiac Electrophysiology
QTc interval prolongation was studied in a randomized, placebo-controlled parallel trial in 116 healthy subjects who received either chloroquine (1000 mg) alone or in combination with oral azithromycin (500 mg, 1000 mg, and 1500 mg once daily). Co-administration of azithromycin increased the QTc interval in a dose- and concentration- dependent manner. In comparison to chloroquine alone, the maximum mean (95% upper confidence bound) increases in QTcF were 5 (10) ms, 7 (12) ms and 9 (14) ms with the co-administration of 500 mg, 1000 mg and 1500 mg azithromycin, respectively.
12.3 Pharmacokinetics
The pharmacokinetic parameters of azithromycin in plasma after dosing as per labeled recommendations in healthy young adults and asymptomatic HIV-positive adults (age 18–40 years old) are portrayed in the following chart:
MEAN (CV%) PK PARAMETER DOSE/DOSAGE FORM
(serum, except as indicated)Subjects Day No. Cmax
( mcg/mL)Tmax
(hr)C24
(mcg/mL)AUC
(mcg∙hr/mL)T½
(hr)Urinary Excretion
(% of dose)500 mg/250 mg capsule
12
1
0.41
2.5
0.05
2.6*
–
4.5
and 250 mg on Days 2–5
12
5
0.24
3.2
0.05
2.1*
–
6.5
1200 mg/600 mg tablets
12
1
0.66
2.5
0.074
6.8†
40
–
%CV
(62%)
(79%)
(49%)
(64%)
(33%)
600 mg tablet/day
7
1
0.33
2.0
0.039
2.4*
%CV
25%
(50%)
(36%)
(19%)
7
22
0.55
2.1
0.14
5.8*
84.5
-
%CV
(18%)
(52%)
(26%)
(25%)
-
600 mg tablet/day (leukocytes)
7
22
252
10.9
146
4763*
82.8
-
%CV
(49%)
(28%)
(33%)
(42%)
-
-
With a regimen of 500 mg on Day 1 and 250 mg/day on Days 2–5, Cmin and Cmax remained essentially unchanged from Day 2 through Day 5 of therapy. However, without a loading dose, azithromycin Cmin levels required 5 to 7 days to reach steady state.
In asymptomatic HIV-positive adult subjects receiving 600 mg ZITHROMAX tablets once daily for 22 days, steady state azithromycin serum levels were achieved by Day 15 of dosing.
The high values in adults for apparent steady-state volume of distribution (31.1 L/kg) and plasma clearance (630 mL/min) suggest that the prolonged half-life is due to extensive uptake and subsequent release of drug from tissues.
Absorption
The 1 gram single-dose packet is bioequivalent to four 250 mg azithromycin capsule.
When the oral suspension of azithromycin was administered with food, the Cmax increased by 46% and the AUC by 14%.
The absolute bioavailability of two 600 mg tablets was 34% (CV=56%). Administration of two 600 mg tablets with food increased Cmax by 31% (CV=43%) while the extent of absorption (AUC) was unchanged (mean ratio of AUCs=1.00; CV=55%).
Distribution
The serum protein binding of azithromycin is variable in the concentration range approximating human exposure, decreasing from 51% at 0.02 µg/mL to 7% at 2 µg/mL.
The antibacterial activity of azithromycin is pH related and appears to be reduced with decreasing pH. However, the extensive distribution of drug to tissues may be relevant to clinical activity.
Azithromycin has been shown to penetrate into tissues in humans, including skin, lung, tonsil, and cervix. Extensive tissue distribution was confirmed by examination of additional tissues and fluids (bone, ejaculum, prostate, ovary, uterus, salpinx, stomach, liver, and gallbladder). As there are no data from adequate and well-controlled studies of azithromycin treatment of infections in these additional body sites, the clinical importance of these tissue concentration data is unknown.
Azithromycin concentrates in phagocytes and fibroblasts as demonstrated by in vitro incubation techniques. Using such methodology, the ratio of intracellular to extracellular concentration was >30 after one hr of incubation. In vivo studies suggest that concentration in phagocytes may contribute to drug distribution to inflamed tissues.
Following oral administration of a single 1200 mg dose (two 600 mg tablets), the mean maximum concentration in peripheral leukocytes was 140 µg/mL. Concentration remained above 32 µg/mL, for approximately 60 hr. The mean half-lives for 6 males and 6 females were 34 hr and 57 hr, respectively. Leukocyte-to-plasma Cmax ratios for males and females were 258 (±77%) and 175 (±60%), respectively, and the AUC ratios were 804 (±31%) and 541 (±28%) respectively. The clinical relevance of these findings is unknown.
Following oral administration of multiple daily doses of 600 mg (1 tablet/day) to asymptomatic HIV-positive adults, mean maximum concentration in peripheral leukocytes was 252 µg/mL (±49%). Trough concentrations in peripheral leukocytes at steady-state averaged 146 µg/mL (±33%). The mean leukocyte-to-serum Cmax ratio was 456 (±38%) and the mean leukocyte to serum AUC ratio was 816 (±31%). The clinical relevance of these findings is unknown.
Metabolism
In vitro and in vivo studies to assess the metabolism of azithromycin have not been performed.
Elimination
Plasma concentrations of azithromycin following single 500 mg oral and IV doses declined in a polyphasic pattern resulting in an average terminal half-life of 68 hr. Biliary excretion of azithromycin, predominantly as unchanged drug, is a major route of elimination. Over the course of a week, approximately 6% of the administered dose appears as unchanged drug in urine.
Specific Populations
Patients with Renal Impairment
Azithromycin pharmacokinetics was investigated in 42 adults (21 to 85 years of age) with varying degrees of renal impairment. Following the oral administration of a single 1.0 g dose of azithromycin (4 × 250 mg capsules), the mean Cmax and AUC0–120 increased by 5.1% and 4.2%, respectively, in subjects with GFR 10 to 80 mL/min compared to subjects with normal renal function (GFR >80 mL/min). The mean Cmax and AUC0–120 increased 61% and 35%, respectively, in subjects with end-stage renal disease (GFR <10 mL/min) compared to subjects with normal renal function (GFR >80 mL/min).
Patients with Hepatic Impairment
The pharmacokinetics of azithromycin in subjects with hepatic impairment has not been established.
Male and Female Patients
There are no significant differences in the disposition of azithromycin between male and female subjects. No dosage adjustment is recommended on the basis of gender.
Geriatric Patients
Pharmacokinetic parameters in older volunteers (65 to 85 years old) were similar to those in younger volunteers (18 to 40 years old) for the 5-day therapeutic regimen. Dosage adjustment does not appear to be necessary for older patients with normal renal and hepatic function receiving treatment with this dosage regimen. [see Geriatric Use (8.5)]
Pediatric Patients
For information regarding the pharmacokinetics of ZITHROMAX (azithromycin for oral suspension) in pediatric patients, see the prescribing information for ZITHROMAX (azithromycin for oral suspension) 100 mg/5 mL and 200 mg/5 mL bottles.
Drug Interaction Studies
Drug interaction studies were performed with azithromycin and other drugs likely to be co-administered. The effects of co-administration of azithromycin on the pharmacokinetics of other drugs are shown in Table 1 and the effects of other drugs on the pharmacokinetics of azithromycin are shown in Table 2.
Co-administration of azithromycin at therapeutic doses had a modest effect on the pharmacokinetics of the drugs listed in Table 1. No dosage adjustment of drugs listed in Table 1 is recommended when co-administered with azithromycin.
Co-administration of azithromycin with efavirenz or fluconazole had a modest effect on the pharmacokinetics of azithromycin. Nelfinavir significantly increased the Cmax and AUC of azithromycin. No dosage adjustment of azithromycin is recommended when administered with drugs listed in Table 2. [see Drug Interactions (7.3)]
Table 1. Drug Interactions: Pharmacokinetic Parameters for Co-administered Drugs in the Presence of Azithromycin Co-administered Drug Dose of Co-administered Drug Dose of Azithromycin n Ratio (with/without azithromycin) of Co-administered Drug Pharmacokinetic Parameters (90% CI); No Effect = 1.00 Mean Cmax Mean AUC - *
- - 90% Confidence interval not reported
Atorvastatin
10 mg/day for 8 days
500 mg/day orally on days 6–8
12
0.83
(0.63 to 1.08)1.01
(0.81 to 1.25)Carbamazepine
200 mg/day for 2 days, then 200 mg twice a day for 18 days
500 mg/day orally for days 16–18
7
0.97
(0.88 to 1.06)0.96
(0.88 to 1.06)Cetirizine
20 mg/day for 11 days
500 mg orally on day 7, then 250 mg/day on days 8–11
14
1.03
(0.93 to 1.14)1.02
(0.92 to 1.13)Didanosine
200 mg orally twice a day for 21 days
1,200 mg/day orally on days 8–21
6
1.44
(0.85 to 2.43)1.14
(0.83 to 1.57)Efavirenz
400 mg/day for 7 days
600 mg orally on day 7
14
1.04*
0.95*
Fluconazole
200 mg orally single dose
1,200 mg orally single dose
18
1.04
(0.98 to 1.11)1.01
(0.97 to 1.05)Indinavir
800 mg three times a day for 5 days
1,200 mg orally on day 5
18
0.96
(0.86 to 1.08)0.90
(0.81 to 1.00)Midazolam
15 mg orally on day 3
500 mg/day orally for 3 days
12
1.27
(0.89 to 1.81)1.26
(1.01 to 1.56)Nelfinavir
750 mg three times a day for 11 days
1,200 mg orally on day 9
14
0.90
(0.81 to 1.01)0.85
(0.78 to 0.93)Sildenafil
100 mg on days 1 and 4
500 mg/day orally for 3 days
12
1.16
(0.86 to 1.57)0.92
(0.75 to 1.12)Theophylline
4 mg/kg IV on days 1, 11, 25
500 mg orally on day 7, 250 mg/day on days 8–11
10
1.19
(1.02 to 1.40)1.02
(0.86 to 1.22)Theophylline
300 mg orally BID ×15 days
500 mg orally on day 6, then 250 mg/day on days 7–10
8
1.09
(0.92 to 1.29)1.08
(0.89 to 1.31)Triazolam
0.125 mg on day 2
500 mg orally on day 1, then 250 mg/day on day 2
12
1.06*
1.02*
Trimethoprim/
Sulfamethoxazole160 mg/800 mg/day orally for 7 days
1,200 mg orally on day 7
12
0.85
(0.75 to 0.97)/
0.90
(0.78 to 1.03)0.87
(0.80 to 0.95/
0.96
(0.88 to 1.03)Zidovudine
500 mg/day orally for 21 days
600 mg/day orally for 14 days
5
1.12
(0.42 to 3.02)0.94
(0.52 to 1.70)Zidovudine
500 mg/day orally for 21 days
1,200 mg/day orally for 14 days
4
1.31
(0.43 to 3.97)1.30
(0.69 to 2.43)Table 2. Drug Interactions: Pharmacokinetic Parameters for Azithromycin in the Presence of Co-administered Drugs. [see Drug Interactions (7.3)] Co-administered Drug Dose of Co-administered Drug Dose of Azithromycin n Ratio (with/without co-administered drug) of Azithromycin Pharmacokinetic Parameters (90% CI); No Effect = 1.00 Mean Cmax Mean AUC - *
- - 90% Confidence interval not reported
Efavirenz
400 mg/day for 7 days
600 mg orally on day 7
14
1.22
(1.04 to 1.42)0.92*
Fluconazole
200 mg orally single dose
1,200 mg orally single dose
18
0.82
(0.66 to 1.02)1.07
(0.94 to 1.22)Nelfinavir
750 mg three times a day for 11 days
1,200 mg orally on day 9
14
2.36
(1.77 to 3.15)2.12
(1.80 to 2.50)12.4 Microbiology
Mechanism of Action
Azithromycin acts by binding to the 23S rRNA of the 50S ribosomal subunit of susceptible microorganisms inhibiting bacterial protein synthesis and impeding the assembly of the 50S ribosomal subunit.
Resistance
The most frequently encountered mechanism of resistance to azithromycin is modification of the 23S rRNA target, most often by methylation. Ribosomal modifications can determine cross resistance to other macrolides, lincosamides, and streptogramin B (MLSB phenotype). The mechanism of acquired mutational resistance in isolates of Mycobacterium avium complex (i.e., 23S rRNA genemutation) is the same for both clarithromycin and azithromycin.
Antimicrobial Activity
Azithromycin has been shown to be active against the following microorganisms, both in vitro and in clinical infections. [see Indications and Usage (1)]
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential. Azithromycin has shown no mutagenic potential in standard laboratory tests: mouse lymphoma assay, human lymphocyte clastogenic assay, and mouse bone marrow clastogenic assay. In fertility studies conducted in male and female rats, oral administration of azithromycin for 64 to 66 days (males) or 15 days (females) prior to and during cohabitation resulted in decreased pregnancy rate at 20 and 30 mg/kg/day when both males and females were treated with azithromycin. This minimal effect on pregnancy rate (approximately 12% reduction compared to concurrent controls) did not become more pronounced when the dose was increased from 20 to 30 mg/kg/day (approximately 0.3 to 0.5 times the adult human daily dose of 600 mg based on body surface area) and it was not observed when only one animal in the mated pair was treated. There were no effects on any other reproductive parameters, and there were no effects on fertility at 10 mg/kg/day. The relevance of these findings to patients being treated with azithromycin at the doses and durations recommended in the prescribing information is uncertain.
13.2 Animal Toxicology
Phospholipidosis (intracellular phospholipid accumulation) has been observed in some tissues of mice, rats, and dogs given multiple doses of azithromycin. It has been demonstrated in numerous organ systems (e.g., eye, dorsal root ganglia, liver, gallbladder, kidney, spleen, and/or pancreas) in dogs and rats treated with azithromycin at doses which, expressed on the basis of body surface area, are similar to or less than the highest recommended adult human dose. This effect has been shown to be reversible after cessation of azithromycin treatment. Based on the pharmacokinetic data, phospholipidosis has been seen in the rat (50 mg/kg/day dose) at the observed maximal plasma concentration of 1.3 mcg/mL (1.6 times the observed Cmax of 0.821 mcg/mL at the adult dose of 2 g.) Similarly, it has been shown in the dog (10 mg/kg/day dose) at the observed maximal serum concentration of 1 mcg/mL (1.2 times the observed Cmax of 0.821 mcg/mL at the adult dose of 2 g).
Phospholipidosis was also observed in neonatal rats dosed for 18 days at 30 mg/kg/day, which is less than the pediatric dose of 60 mg/kg based on the surface area. It was not observed in neonatal rats treated for 10 days at 40 mg/kg/day with mean maximal serum concentrations of 1.86 mcg/mL, approximately 1.5 times the Cmax of 1.27 mcg/mL at the pediatric dose. Phospholipidosis has been observed in neonatal dogs (10 mg/kg/day) at maximum mean whole blood concentrations of 3.54 mcg/mL, approximately 3 times the pediatric dose Cmax.
The significance of the finding for animals and for humans is unknown.
-
14 CLINICAL STUDIES
14.1 Clinical Studies in Patients with Advanced HIV Infection for the Prevention and Treatment of Disease Due to Disseminated Mycobacterium avium Complex (MAC)
[see Indications and Usage (1)]
Prevention of Disseminated MAC Disease
Two randomized, double-blind clinical trials were performed in patients with CD4 counts <100 cells/µL. The first trial (Study 155) compared azithromycin (1200 mg once weekly) to placebo and enrolled 182 patients with a mean CD4 count of 35 cells/mcgL. The second trial (Study 174) randomized 723 patients to either azithromycin (1200 mg once weekly), rifabutin (300 mg daily), or the combination of both. The mean CD4 count was 51 cells/mcgL. The primary endpoint in these trials was disseminated MAC disease. Other endpoints included the incidence of clinically significant MAC disease and discontinuations from therapy for drug-related side effects.
MAC bacteremia
In Study 155, 85 patients randomized to receive azithromycin and 89 patients randomized to receive placebo met the entrance criteria. Cumulative incidences at 6, 12, and 18 months of the possible outcomes are in the following table:
Cumulative Incidence Rate, %: Placebo (n=89)
Month
MAC Free and Alive
MAC
Adverse Experience
Lost to Follow-up
6
69.7
13.5
6.7
10.1
12
47.2
19.1
15.7
18.0
18
37.1
22.5
18.0
22.5
Cumulative Incidence Rate, %: Azithromycin (n=85)
Month
MAC Free and Alive
MAC
Adverse Experience
Lost to Follow-up
6
84.7
3.5
9.4
2.4
12
63.5
8.2
16.5
11.8
18
44.7
11.8
25.9
17.6
The difference in the one-year cumulative incidence rates of disseminated MAC disease (placebo – azithromycin) is 10.9%. This difference is statistically significant (p=0.037) with a 95% confidence interval for this difference of 0.8%, 20.9%. The comparable number of patients experiencing adverse events and the fewer number of patients lost to follow-up on azithromycin should be taken into account when interpreting the significance of this difference.
In Study 174, 223 patients randomized to receive rifabutin, 223 patients randomized to receive azithromycin, and 218 patients randomized to receive both rifabutin and azithromycin met the entrance criteria. Cumulative incidences at 6, 12, and 18 months of the possible outcomes are recorded in the following table:
Cumulative Incidence Rate, %: Rifabutin (n=223)
Month
MAC Free and Alive
MAC
Adverse Experience
Lost to Follow-up
6
83.4
7.2
8.1
1.3
12
60.1
15.2
16.1
8.5
18
40.8
21.5
24.2
13.5
Cumulative Incidence Rate, %: Azithromycin (n=223)
Month
MAC Free and Alive
MAC
Adverse Experience
Lost to Follow-up
6
85.2
3.6
5.8
5.4
12
65.5
7.6
16.1
10.8
18
45.3
12.1
23.8
18.8
Cumulative Incidence Rate, %: Azithromycin/Rifabutin Combination (n=218)
Month
MAC Free and Alive
MAC
Adverse Experience
Lost to Follow-up
6
89.4
1.8
5.5
3.2
12
71.6
2.8
15.1
10.6
18
49.1
6.4
29.4
15.1
Comparing the cumulative one-year incidence rates, azithromycin monotherapy is at least as effective as rifabutin monotherapy. The difference (rifabutin – azithromycin) in the one-year rates (7.6%) is statistically significant (p=0.022) with an adjusted 95% confidence interval (0.9%, 14.3%). Additionally, azithromycin/rifabutin combination therapy is more effective than rifabutin alone. The difference (rifabutin – azithromycin/rifabutin) in the cumulative one-year incidence rates (12.5%) is statistically significant (p<0.001) with an adjusted 95% confidence interval of 6.6%, 18.4%. The comparable number of patients experiencing adverse events and the fewer number of patients lost to follow-up on rifabutin should be taken into account when interpreting the significance of this difference.
In Study 174, sensitivity testing1 was performed on all available MAC isolates from subjects randomized to either azithromycin, rifabutin, or the combination. The distribution of MIC values for azithromycin from susceptibility testing of the breakthrough isolates was similar between trial arms. As the efficacy of azithromycin in the treatment of disseminated MAC has not been established, the clinical relevance of these in vitro MICs as an indicator of susceptibility or resistance is not known.
Clinically Significant Disseminated MAC Disease
In association with the decreased incidence of bacteremia, patients in the groups randomized to either azithromycin alone or azithromycin in combination with rifabutin showed reductions in the signs and symptoms of disseminated MAC disease, including fever or night sweats, weight loss, and anemia.
Discontinuations from Therapy for Drug-Related Side Effects
In Study 155, discontinuations for drug-related toxicity occurred in 8.2% of subjects treated with azithromycin and 2.3% of those given placebo (p=0.121). In Study 174, more subjects discontinued from the combination of azithromycin and rifabutin (22.7%) than from azithromycin alone (13.5%; p=0.026) or rifabutin alone (15.9%; p=0.209).
Safety
As these patients with advanced HIV disease were taking multiple concomitant medications and experienced a variety of intercurrent illnesses, it was often difficult to attribute adverse reactions to study medication. Overall, the nature of adverse reactions seen on the weekly dosage regimen of azithromycin over a period of approximately one year in patients with advanced HIV disease were similar to that previously reported for shorter course therapies.
INCIDENCE OF ONE OR MORE TREATMENT-RELATED* ADVERSE REACTIONS† IN HIV INFECTED PATIENTS RECEIVING PROPHYLAXIS FOR DISSEMINATED MAC OVER APPROXIMATELY 1 YEAR Study 155 Study 174 Placebo Azithromycin 1200 mg weekly Azithromycin 1200 mg weekly Rifabutin 300 mg daily Azithromycin + Rifabutin (N=91) (N=89) (N=233) (N=236) (N=224) Mean Duration of Therapy (days)
303.8
402.9
315
296.1
344.4
Discontinuation of Therapy
2.3
8.2
13.5
15.9
22.7
Autonomic Nervous System
Mouth Dry
0
0
0
3.0
2.7
Central Nervous System
Dizziness
0
1.1
3.9
1.7
0.4
Headache
0
0
3.0
5.5
4.5
Gastrointestinal
Diarrhea
15.4
52.8
50.2
19.1
50.9
Loose Stools
6.6
19.1
12.9
3.0
9.4
Abdominal Pain
6.6
27
32.2
12.3
31.7
Dyspepsia
1.1
9
4.7
1.7
1.8
Flatulence
4.4
9
10.7
5.1
5.8
Nausea
11
32.6
27.0
16.5
28.1
Vomiting
1.1
6.7
9.0
3.8
5.8
General
Fever
1.1
0
2.1
4.2
4.9
Fatigue
0
2.2
3.9
2.1
3.1
Malaise
0
1.1
0.4
0
2.2
Musculoskeletal
Arthralgia
0
0
3.0
4.2
7.1
Psychiatric
Anorexia
1.1
0
2.1
2.1
3.1
Skin & Appendages
Pruritus
3.3
0
3.9
3.4
7.6
Rash
3.2
3.4
8.1
9.4
11.1
Skin discoloration
0
0
0
2.1
2.2
Special Senses
Tinnitus
4.4
3.4
0.9
1.3
0.9
Hearing Decreased
2.2
1.1
0.9
0.4
0
Uveitis
0
0
0.4
1.3
1.8
Taste Perversion
0
0
1.3
2.5
1.3
Adverse reactions related to the gastrointestinal tract were seen more frequently in patients receiving azithromycin than in those receiving placebo or rifabutin. In Study 174, 86% of diarrheal episodes were mild to moderate in nature with discontinuation of therapy for this reason occurring in only 9/233 (3.8%) of patients.
Changes in Laboratory Values
In these immunocompromised patients with advanced HIV infection, it was necessary to assess laboratory abnormalities developing on trial with additional criteria if baseline values were outside the relevant normal range.
PROPHYLAXIS AGAINST DISSEMINATED MAC ABNORMAL LABORATORY VALUES* Placebo Azithromycin 1200 mg weekly Rifabutin 300 mg daily Azithromycin & Rifabutin Hemoglobin
<8 g/dL
1/51
2%
4/170
2%
4/114
4%
8/107
8%
Platelet Count
<50 × 103/mm3
1/71
1%
4/260
2%
2/182
1%
6/181
3%
WBC Count
<1 × 103/mm3
0/8
0%
2/70
3%
2/47
4%
0/43
0%
Neutrophils
<500/mm3
0/26
0%
4/106
4%
3/82
4%
2/78
3%
SGOT
>5 × ULN†
1/41
2%
8/158
5%
3/121
3%
6/114
5%
SGPT
>5 × ULN
0/49
0%
8/166
5%
3/130
2%
5/117
4%
Alk Phos
>5 × ULN
1/80
1%
4/247
2%
2/172
1%
3/164
2%
Treatment of Disseminated MAC Disease
One randomized, double-blind clinical trial (Study 189) was performed in patients with disseminated MAC. In this trial, 246 HIV-infected patients with disseminated MAC received either azithromycin 250 mg daily (N=65), azithromycin 600 mg daily (N=91), or clarithromycin 500 mg twice a day (N=90), each administered with ethambutol 15 mg/kg daily, for 24 weeks. Blood cultures and clinical assessments were performed every 3 weeks through week 12 and monthly thereafter through week 24. After week 24, patients were switched to any open-label therapy at the discretion of the investigator and followed every 3 months through the last follow-up visit of the trial. Patients were followed from the baseline visit for a period of up to 3.7 years (median: 9 months). MAC isolates recovered during treatment or post-treatment were obtained whenever possible.
The primary endpoint was sterilization by week 24. Sterilization was based on data from the central laboratory, and was defined as two consecutive observed negative blood cultures for MAC, independent of missing culture data between the two negative observations. Analyses were performed on all randomized patients who had a positive baseline culture for MAC.
The azithromycin 250 mg arm was discontinued after an interim analysis at 12 weeks showed a significantly lower clearance of bacteremia compared to clarithromycin 500 mg twice a day. Efficacy results for the azithromycin 600 mg daily and clarithromycin 500 mg twice a day treatment regimens are described in the following table:
RESPONSE TO THERAPY OF PATIENTS TAKING ETHAMBUTOL AND EITHER AZITHROMYCIN 600 MG DAILY OR CLARITHROMYCIN 500 MG TWICE A DAY Azithromycin 600 mg daily Clarithromycin 500 mg twice a day *95.1% CI on difference Patients with positive culture at baseline
68
57
Week 24
Two consecutive negative blood cultures†
31/68 (46%)
32/57 (56%)
[-28, 7]
Mortality
16/68 (24%)
15/57 (26%)
[-18, 13]
The primary endpoint, rate of sterilization of blood cultures (two consecutive negative cultures) at 24 weeks, was lower in the azithromycin 600 mg daily group than in the clarithromycin 500 mg twice a day group.
Sterilization by Baseline Colony Count
Within both treatment groups, the sterilization rates at week 24 decreased as the range of MAC cfu/mL increased.
Azithromycin 600 mg (N=68) Clarithromycin 500 mg twice a day (N=57) groups stratified by MAC colony counts at baseline no. (%) subjects in stratified group sterile at week 24 no. (%) subjects in stratified group sterile at week 24 ≤10 cfu/mL
10/15 (66.7%)
12/17 (70.6%)
11–100 cfu/mL
13/28 (46.4%)
13/19 (68.4%)
101–1,000 cfu/mL
7/19 (36.8%)
5/13 (38.5%)
1,001–10,000 cfu/mL
1/5 (20.0%)
1/5 (20%)
>10,000 cfu/mL
0/1 (0.0%)
1/3 (33.3%)
Susceptibility Pattern of MAC Isolates
Susceptibility testing was performed on MAC isolates recovered at baseline, at the time of breakthrough on therapy or during post-therapy follow-up. The T100 radiometric broth method was employed to determine azithromycin and clarithromycin MIC values. Azithromycin MIC values ranged from <4 to >256 µg/mL and clarithromycin MICs ranged from <1 to >32 µg/mL. The individual MAC susceptibility results demonstrated that azithromycin MIC values could be 4 to 32-fold higher than clarithromycin MIC values.
During treatment and post-treatment follow-up for up to 3.7 years (median: 9 months) in Study 189, a total of 6/68 (9%) and 6/57 (11%) of the patients randomized to azithromycin 600 mg daily and clarithromycin 500 mg twice a day respectively, developed MAC blood culture isolates that had a sharp increase in MIC values. All twelve MAC isolates had azithromycin MICs ≥256 µg/mL and clarithromycin MICs >32 µg/mL. These high MIC values suggest development of drug resistance. However, at this time, specific breakpoints for separating susceptible and resistant MAC isolates have not been established for either macrolide.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
ZITHROMAX 600 mg tablets (engraved on front with "PFIZER" and on back with "308") are supplied as white, modified oval-shaped, film-coated tablets containing azithromycin dihydrate equivalent to 600 mg azithromycin. These are packaged in bottles of 30 tablets. ZITHROMAX tablets are supplied as follows:
Bottles of 30
NDC 0069-3080-30
Tablets should be stored at or below 30°C (86°F).
ZITHROMAX for oral suspension is supplied in single-dose packets containing azithromycin dihydrate equivalent to 1 gram of azithromycin as follows:
Boxes of 10 single-dose packets (1 g)
NDC 0069-3051-07
Boxes of 3 single-dose packets (1 g)
NDC 0069-3051-75
-
17 PATIENT COUNSELING INFORMATION
ZITHROMAX tablets may be taken with or without food. However, increased tolerability has been observed when tablets are taken with food.
ZITHROMAX for oral suspension in single 1 g packets can be taken with or without food after constitution.
Patients should also be cautioned not to take aluminum- and magnesium-containing antacids and azithromycin simultaneously.
The patient should be directed to discontinue azithromycin immediately and contact a physician if any signs of an allergic reaction occur.
Direct parents or caregivers to contact their physician if vomiting and irritability with feeding occurs in the infant.
Patients should be counseled that antibacterial drugs, including ZITHROMAX, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When ZITHROMAX is prescribed to treat bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by ZITHROMAX or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibacterial which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibacterials, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 600 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 1 g Packet Label
- PRINCIPAL DISPLAY PANEL - 1 g Packet Box
-
INGREDIENTS AND APPEARANCE
ZITHROMAX
azithromycin dihydrate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0069-3080 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AZITHROMYCIN DIHYDRATE (UNII: 5FD1131I7S) (AZITHROMYCIN ANHYDROUS - UNII:J2KLZ20U1M) AZITHROMYCIN ANHYDROUS 600 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) SODIUM LAURYL SULFATE (UNII: 368GB5141J) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color WHITE Score no score Shape OVAL Size 19mm Flavor Imprint Code PFIZER;308 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-3080-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 06/12/1996 09/30/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050730 06/12/1996 09/30/2018 ZITHROMAX
azithromycin dihydrate powder, for suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0069-3051 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AZITHROMYCIN DIHYDRATE (UNII: 5FD1131I7S) (AZITHROMYCIN ANHYDROUS - UNII:J2KLZ20U1M) AZITHROMYCIN ANHYDROUS 1 g Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM PHOSPHATE, TRIBASIC, ANHYDROUS (UNII: SX01TZO3QZ) SUCROSE (UNII: C151H8M554) Product Characteristics Color Score Shape Size Flavor BANANA, CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-3051-07 10 in 1 BOX 09/28/1994 1 NDC:0069-3051-01 1 in 1 PACKET; Type 0: Not a Combination Product 2 NDC:0069-3051-75 3 in 1 BOX 09/28/1994 2 NDC:0069-3051-01 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050693 09/28/1994 Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) Registrant - Pfizer Inc (113480771) Establishment Name Address ID/FEI Business Operations Pfizer Ireland Pharmaceuticals 985052076 ANALYSIS(0069-3051, 0069-3080) , API MANUFACTURE(0069-3051, 0069-3080) Establishment Name Address ID/FEI Business Operations Pfizer Inc 943955690 ANALYSIS(0069-3051, 0069-3080) Establishment Name Address ID/FEI Business Operations Pfizer Asia Manufacturing Pte Ltd 936889401 API MANUFACTURE(0069-3080, 0069-3051) , ANALYSIS(0069-3080, 0069-3051) Establishment Name Address ID/FEI Business Operations Eurofins Lancaster Laboratories, Inc 069777290 ANALYSIS(0069-3080, 0069-3051) Establishment Name Address ID/FEI Business Operations Viatris Pharmaceuticals LLC 829084552 PACK(0069-3080, 0069-3051) , ANALYSIS(0069-3080, 0069-3051) , MANUFACTURE(0069-3080) Establishment Name Address ID/FEI Business Operations Alembic Pharmaceuticals Limited 650574671 MANUFACTURE(0069-3080)