TEPADINA- thiotepa injection, powder, for solution
ADIENNE SA
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use TEPADINA® safely and effectively. See full prescribing information for TEPADINA®.
TEPADINA® (thiotepa) for injection, for intravenous, intracavitary, or intravesical use. Initial U.S. Approval: 1959 WARNING: SEVERE MYELOSUPPRESSION, CARCINOGENICITY
resulting infection or bleeding. Monitor hematologic laboratory parameters. Hematopoietic progenitor
|
FULL PRESCRIBING INFORMATION
WARNING: SEVERE MYELOSUPPRESSION, CARCINOGENICITY
- TEPADINA may cause severe marrow suppression, and high doses may cause marrow ablation with resulting infection or bleeding. Monitor hematologic laboratory parameters. Hematopoietic progenitor (stem) cell transplantation (HSCT) is required to prevent potentially fatal complications of the prolonged myelosuppression after high doses of TEPADINA [see Warnings and Precautions (5.1)]
- TEPADINA should be considered potentially carcinogenic in humans [see Warnings and Precautions (5.7)]
1 INDICATIONS AND USAGE
1.1 Class 3 Beta-Thalassemia
TEPADINA is indicated to reduce the risk of graft rejection when used in conjunction with high-dose busulfan and cyclophosphamide as a preparative regimen for allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) for pediatric patients with class 3 beta-thalassemia [see Clinical Studies (14)].
1.2 Adenocarcinoma of the Breast or Ovary
TEPADINA is indicated for treatment of adenocarcinoma of the breast or ovary.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dose of TEPADINA in pediatric patients is two administrations of 5 mg/kg given intravenously approximately 12 hours apart on Day -6 before allogeneic HSCT in conjunction with high-dose busulfan and cyclophosphamide as outlined in Table 1. See Prescribing Information for cyclophosphamide and busulfan for information on these drugs.The recommended dose of TEPADINA in pediatric patients is two administrations of 5 mg/kg given intravenously approximately 12 hours apart on Day -6 before allogeneic HSCT in conjunction with high-dose busulfan and cyclophosphamide as outlined in Table 1. See Prescribing Information for cyclophosphamide and busulfan for information on these drugs.
|
Table 1: Dosage Regimen For Allogeneic HSCT In Pediatric Patients With Class 3 Beta-Thalassemia |
|||||||||||
|
Day prior to transplantationDay prior to transplantation |
|||||||||||
| Treatment |
DayDay -10-10 |
DayDay -9-9 |
DayDay -8-8 |
DayDay -7-7 |
DayDay -6-6 |
DayDay -5-5 |
DayDay -4-4 |
DayDay -3-3 |
DayDay -2-2 |
DayDay -1-1 |
DayDay -0-0 |
|
Busulfan IV weight-based dose *Busulfan IV weight-based dose *Busulfan IV weight-based dose *Busulfan IV weight-based dose * | ▲ | ▲ | ▲ | ▲ | |||||||
|
TEPADINA IV 5 mg/kg twiceTEPADINA IV 5 mg/kg twiceTEPADINA IV 5 mg/kg twiceTEPADINA IV 5 mg/kg twice
| ▲ | ||||||||||
|
Cyclophosphamide IV 40 mg/kg/dayCyclophosphamide IV 40 mg/kg/day | ▲ | ▲ | ▲ | ▲ | |||||||
|
Stem cell InfusionStem cell InfusionStem cell InfusionStem cell Infusion
| ▲ | ||||||||||
| *Busulfan IV weight-based dose: 1.0 mg/kg every 6 hours for patients less than 9 kg; 1.2 mg/kg every 6 hours for patients 9 to 16 kg; 1.1 mg/kg every 6 hours for patients 16.1 to 23 kg; 0.95 mg/kg every 6 hours for patients 23.1 to 34 kg; 0.8 mg/kg every 6 hours for patients more than 34 kg. | |||||||||||
Infuse TEPADINA via a central venous catheter over 3 hours using an infusion set equipped with a 0.2 micron in-line filter. Prior to and following each infusion, flush the catheter with approximately 5 ml sodium chloride 0.9% solution for injection.Infuse TEPADINA via a central venous catheter over 3 hours using an infusion set equipped with a 0.2 micron in-line filter. Prior to and following each infusion, flush the catheter with approximately 5 ml sodium chloride 0.9% solution for injection.
TEPADINA is excreted through the skin of patients receiving high-dose therapy. Take precautions to prevent skin toxicity [ ]. TEPADINA is excreted through the skin of patients receiving high-dose therapy. Take precautions to prevent skin toxicity [ see Warnings and Precautions ( 5.3)].
Adenocarcinoma of the Breast or Ovary
The recommended dose of TEPADINA for treatment of adenocarcinoma of the breast or ovary is 0.3 to 0.4 mg/kg intravenously. Doses should be given at 1 to 4 week intervals. Initially the higher dose in the given range is commonly administered. The maintenance dose should be adjusted weekly on the basis of pretreatment control blood counts and subsequent blood counts. Maintenance doses should not be administered more frequently than weekly.The recommended dose of TEPADINA for treatment of adenocarcinoma of the breast or ovary is 0.3 to 0.4 mg/kg intravenously. Doses should be given at 1 to 4 week intervals. Initially the higher dose in the given range is commonly administered. The maintenance dose should be adjusted weekly on the basis of pretreatment control blood counts and subsequent blood counts. Maintenance doses should not be administered more frequently than weekly.
Malignant Effusions
The recommended dose of TEPADINA for treatment of malignant effusions is 0.6 to 0.8 mg/kg intracavitary. Administration is usually effected through the same tubing which is used to remove the fluid from the cavity involved. Doses should be given at 1 to 4 week intervals. Initially the higher dose in the given range is commonly administered. The maintenance dose should be adjusted weekly on the basis of pretreatment control blood counts and subsequent blood counts. Maintenance doses should not be administered more frequently than weekly.The recommended dose of TEPADINA for treatment of malignant effusions is 0.6 to 0.8 mg/kg intracavitary. Administration is usually effected through the same tubing which is used to remove the fluid from the cavity involved. Doses should be given at 1 to 4 week intervals. Initially the higher dose in the given range is commonly administered. The maintenance dose should be adjusted weekly on the basis of pretreatment control blood counts and subsequent blood counts. Maintenance doses should not be administered more frequently than weekly.
Superficial Papillary Carcinoma of the Urinary Bladder
The recommended dose of TEPADINA for treatment of superficial papillary carcinoma of the urinary bladder is 60 mg in 30 to 60 mL of Sodium Chloride Injection into the bladder by catheter. The solution should be retained for 2 hours. If the patient finds it impossible to retain 60 mL for 2 hours, the dose may be given in a volume of 30 mL. The patient may be repositioned every 15 minutes for maximum area contact. The usual course of treatment is once a week for 4 weeks. The course may be repeated if necessary, but second and third courses must be given with caution since bone-marrow depression may be increased.The recommended dose of TEPADINA for treatment of superficial papillary carcinoma of the urinary bladder is 60 mg in 30 to 60 mL of Sodium Chloride Injection into the bladder by catheter. The solution should be retained for 2 hours. If the patient finds it impossible to retain 60 mL for 2 hours, the dose may be given in a volume of 30 mL. The patient may be repositioned every 15 minutes for maximum area contact. The usual course of treatment is once a week for 4 weeks. The course may be repeated if necessary, but second and third courses must be given with caution since bone-marrow depression may be increased.
2.2 Preparation Instructions
TEPADINA is a cytotoxic drug. Follow applicable special handling and disposal procedures1.
Reconstitution
Reconstitute TEPADINA 15 mg with 1.5 ml of sterile water for injection. Using a syringe fitted with a needle, aseptically
withdraw 1.5 ml of sterile water for injection. Inject the content of the syringe into the vial through the rubber stopper.
Remove the syringe and needle, and mix manually by repeated inversions.
Reconstitute TEPADINA 100 mg with 10 ml of sterile water for injection. Using a syringe fitted with a needle, aseptically
withdraw 10 ml of sterile water for injection. Inject the content of the syringe into the vial through the rubber stopper.
Remove the syringe and needle, and mix manually by repeated inversions.
The reconstituted solution is hypotonic and must be diluted in saline prior to administration. Reconstituted solutions, free of
visible particulate matter, may occasionally show opalescence; such solutions can still be used for further dilution.
If not used immediately after reconstitution, the product is stable for 8 hours when stored at 2°C to 8°C (36° to 46°F).
Dilution in the infusion bag
Prior to administration, dilute the reconstituted solution further with an appropriate volume of sodium chloride 0.9% solution
for injection to obtain a final TEPADINA concentration between 0.5 and 1 mg/mL. Dilute TEPADINA as recommended in
Table 2.
Table 2: Dilution of TEPADINA in the infusion bag
|
Calculated TEPADINA Dose |
Dilution Volume (Sodium Chloride 0.9% solution for injection) |
|
Less than 250 mg |
Appropriate volume to obtain a final concentration of 0.5 to 1 mg/mL |
|
250 mg to 500 mg |
500 mL or appropriate volume to obtain a final concentration of 0.5 to 1 mg/mL |
|
Greater than 500 mg |
1000 mL or appropriate volume to obtain a final concentration of 0.5 to 1 mg/mL |
After dilution the product is stable for 24 hours when stored at 2°C to 8°C (36° to 46°F) and for 4 hours when stored at 25°C
(77°F). From a microbiological point of view, the product should be used immediately.
Inspect the diluted solution visually for particulate matter and discoloration prior to administration. Use TEPADINA diluted
solutions only if free of visible particulate matter. Filter using a 0.2 micron filter prior to administration. Filtering does not
alter solution potency
3 DOSAGE FORMS AND STRENGTHS
- For injection, 15 mg, lyophilized white powder in single-dose vial for reconstitution
- For injection, 100 mg, lyophilized white powder in single-dose vial for reconstitution
4 CONTRAINDICATIONS
TEPADINA is contraindicated in:
- Patients with severe hypersensitivity to thiotepa [see Warnings and Precautions (5.2)]
- Concomitant use with live or attenuated vaccines [see Warnings and Precautions (5.4)].
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
The consequence of treatment with high doses of TEPADINA together with other chemotherapy at the recommended dose and schedule in the preparative regimen for class 3 beta- thalassemia is profound myelosuppression occurring in all patients. Do not begin the preparative regimen if a stem cell donor is not available. Monitor complete blood counts, and provide supportive care for infections, anemia and thrombocytopenia until there is adequate hematopoietic recovery.
For patients receiving TEPADINA for treatment of adenocarcinoma of the breast, adenocarcinoma of the ovary, malignant effusions and superficial papillary carcinoma of the urinary bladder, if the bone marrow has been compromised by prior irradiation or chemotherapy, or is recovering from chemotherapy, the risk of severe myelosuppression with TEPADINA may be increased. Perform periodic complete blood counts during the course of treatment with TEPADINA. Provide supportive care for infections, bleeding, and symptomatic anemia [see Adverse Reactions (6.1)].
5.2 Hypersensitivity
Clinically significant hypersensitivity reactions, including anaphylaxis, have occurred following administration of TEPADINA. If anaphylactic or other clinically significant allergic reaction occurs, discontinue treatment with TEPADINA, initiate appropriate therapy, and monitor until signs and symptoms resolve [see Contraindications (4), Adverse Reactions (6.1)].
5.3 Cutaneous Toxicity
TEPADINA and/or its active metabolites may be excreted in part via skin patients receiving high-dose therapy. Treatment with TEPADINA may cause skin discoloration, pruritus, blistering, desquamation, and peeling that may be more severe in the groin, axillae, skin folds, in the neck area, and under dressings. Instruct patients to shower or bathe with water at least twice daily through 48 hours after administration of TEPADINA. Change occlusive dressing and clean the covered skin at least twice daily through 48 hours after administration of TEPADINA. Change bed sheets daily during treatment.
Skin reactions associated with accidental exposure to TEPADINA may also occur. Wash the skin thoroughly with soap and water in case TEPADINA solution contacts the skin. Flush mucous membranes in case of TEPADINA contact with mucous membranes.
5.4 Concomitant Use of Live and Attenuated Vaccines
Do not administer live or attenuated viral or bacterial vaccines to a patient treated with TEPADINA until the immunosuppressive effects have resolved.
5.5 Hepatic Veno-Occlusive Disease
Hepatic veno-occlusive disease may occur in patients who have received high-dose TEPADINA in conjunction with busulfan and cyclophosphamide [see Adverse Reactions (6.1)]. Monitor by physical examination, serum transaminases and bilirubin daily through BMT Day +28, and provide supportive care to patients who develop hepatic veno-occlusive disease.
5.6 Central Nervous System Toxicity
Fatal encephalopathy has occurred in patients treated with high doses of thiotepa. Other central nervous system toxicities, such as headache, apathy, psychomotor retardation, disorientation, confusion, amnesia, hallucinations, drowsiness, somnolence, seizures, coma, inappropriate behaviour and forgetfulness have been reported to occur in a dose-dependent manner during or shortly after administration of high-dose thiotepa. In pediatric patients treated with TEPADINA at the recommended dose in combination with busulfan and cyclophosphamide, 8% developed central nervous system toxicity (seizures and intracranial hemorrhage). Do not exceed the recommended dose of TEPADINA. If severe or life-threatening central nervous system toxicity occurs, discontinue administration of TEPADINA and provide supportive care.
5.7 Carcinogenicity
Like many alkylating agents, thiotepa has been reported to be carcinogenic when administered to laboratory animals [see Nonclinical Toxicity (13.1)]. Carcinogenicity is shown most clearly in studies using mice, but there is some evidence of carcinogenicity in man. There is an increased risk of a secondary malignancy with use of TEPADINA.
5.8 Embryo-Fetal Toxicity
Based on the mechanism of action and findings in animals, TEPADINA can cause fetal harm when administered to a pregnant woman. There are no adequate and well-controlled studies of TEPADINA in pregnant women. Thiotepa given by the intraperitoneal (IP) route was teratogenic in mice at doses ≥ 1 mg/kg (3.2 mg/m 2), approximately 8-fold less than the maximum recommended human therapeutic dose (0.8 mg/kg, 27 mg/m 2), based on body-surface area. Thiotepa given by the IP route was teratogenic in rats at doses ≥ 3 mg/kg (21 mg/m 2), approximately equal to the maximum recommended human therapeutic dose, based on body-surface area. Thiotepa was lethal to rabbit fetuses at a dose of 3 mg/kg (41 mg/m 2), approximately two times the maximum recommended human therapeutic dose based on body-surface area.
Advise pregnant women of the potential risk to the fetus [ see Use in Specific Populations (8.1, 8.3)]. Advise females of reproductive potential to use highly effective contraception during and after treatment with TEPADINA for at least 6 months after therapy. Advise males of reproductive potential to use effective contraception during and after treatment with TEPADINA for at least 1 year after therapy [ see Use in Specific Populations (8.1, 8.3)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Myelosuppression [ see Warnings and Precautions ( 5.1)]
- Infection [ see Warnings and Precautions (5.1)]
- Hypersensitivity [ see Warnings and Precautions (5.2)].
- Cutaneous Toxicity [ see Warnings and Precautions (5.3)]
- Hepatic Veno-Occlusive Disease [ see Warnings and Precautions (5.5)]
- Central Nervous System Toxicity [ see Warnings and Precautions (5.6)]
- Carcinogenicity [ see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions With the Preparative Regimen for Class 3 Beta-Thalassemia
The safety of TEPADINA was evaluated by retrospective analysis of 76 pediatric patients with class 3 beta-thalassemia who underwent allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) using busulfan and cyclophosphamide with TEPADINA (n=25) or without TEPADINA (n=51) [ ]. Adverse reactions were abstracted retrospectively from the medical records. The safety of TEPADINA was evaluated by retrospective analysis of 76 pediatric patients with class 3 beta-thalassemia who underwent allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) using busulfan and cyclophosphamide with TEPADINA (n=25) or without TEPADINA (n=51) [ ]. Adverse reactions were abstracted retrospectively from the medical records.
Serious adverse events that occurred in the TEPADINA-treated and control cohort were, respectively: gastrointestinal hemorrhage (4% vs 2%), pneumonia (4% vs 0), seizure (4% vs 2%), subarachnoid hemorrhage (4% vs 0) and veno-occlusive disease (4% vs 2%). By 90 days after HSCT, grades 2 to 4 acute graft-versus-host disease was observed in 7 (28%) patients in the TEPADINA cohort and in 13 (26%) patients in the control cohort. By 1-year after transplantation, chronic graft-versus-host disease was observed in 8 (35%) of 23 evaluable patients in the TEPADINA cohort, and 7 (14%) of 49 evaluable patients in the control cohort.Serious adverse events that occurred in the TEPADINA-treated and control cohort were, respectively: gastrointestinal hemorrhage (4% vs 2%), pneumonia (4% vs 0), seizure (4% vs 2%), subarachnoid hemorrhage (4% vs 0) and veno-occlusive disease (4% vs 2%). By 90 days after HSCT, grades 2 to 4 acute graft-versus-host disease was observed in 7 (28%) patients in the TEPADINA cohort and in 13 (26%) patients in the control cohort. By 1-year after transplantation, chronic graft-versus-host disease was observed in 8 (35%) of 23 evaluable patients in the TEPADINA cohort, and 7 (14%) of 49 evaluable patients in the control cohort.
Adverse reactions occurring in at least 5% of patients treated with TEPADINA from start of the preparative regimen through 30 days after transplantation are shown in Table 3.Adverse reactions occurring in at least 5% of patients treated with TEPADINA from start of the preparative regimen through 30 days after transplantation are shown in Table 3.
| Table 3: Common Adverse Reactions (>5%) Occurring Through 30 Days After Transplantation In Patients With Class 3 Beta-Thalassemia Using Busulfan And Cyclophosphamide With Or Without TEPADINA in the Preparative Regimen | ||||
| Preparative Regimen of Busulfan and Cyclophosphamide | ||||
|
With TEPADINAWith TEPADINA N=25 patients (%)N=25 patients (%) |
Without TEPADINAWithout TEPADINA N=51 patients (%)N=51 patients (%) |
|||
|
Adverse ReactionAdverse Reaction |
Any GradeAny Grade | Grade 3-5 1 | Any Grade | Grade 3-5 1 |
| Mucositis 2 |
16 (64%)16 (64%)16 (64%)16 (64%) |
4 (16%)4 (16%)4 (16%)4 (16%) |
22 (43%)22 (43%)22 (43%)22 (43%) |
1 (2%)1 (2%)1 (2%)1 (2%) |
|
Cytomegalovirus InfectionCytomegalovirus InfectionCytomegalovirus InfectionCytomegalovirus Infection |
12 (48%)12 (48%)12 (48%)12 (48%) |
00 |
15 (29%)15 (29%)15 (29%)15 (29%) | 0 |
| Hemorrhage 3 |
7 (28%)7 (28%)7 (28%)7 (28%) |
2 (8%)2 (8%)2 (8%)2 (8%) |
12 (24%)12 (24%)12 (24%)12 (24%) |
3 (6%)3 (6%)3 (6%)3 (6%) |
| Diarrhea |
6 (24%)6 (24%)6 (24%)6 (24%) | 0 |
7 (14%)7 (14%)7 (14%)7 (14%) |
2 (4%)2 (4%)2 (4%)2 (4%) |
| Hematuria 4 |
5 (20%)5 (20%)5 (20%)5 (20%) | 0 |
10 (20%)10 (20%)10 (20%)10 (20%) |
3 (6%)3 (6%)3 (6%)3 (6%) |
| Rash 5 |
3 (12%)3 (12%)3 (12%)3 (12%) | 0 |
11 (22%)11 (22%)11 (22%)11 (22%) | 0 |
| Intracranial Hemorrhage 6 |
2 (8%)2 (8%)2 (8%)2 (8%) |
1 (4%)1 (4%)1 (4%)1 (4%) | 0 | 0 |
| Pseudomonas Infection |
2 (8%)2 (8%)2 (8%)2 (8%) | 0 | 0 | 0 |
|
Severe, life-threatening or fatal 1Severe, life-threatening or fatal Mucositis includes mouth hemorrhage, mucosal inflammation and stomatitis 2Mucositis includes mouth hemorrhage, mucosal inflammation and stomatitis Hemorrhage includes all hemorrhage terms 3Hemorrhage includes all hemorrhage terms Hematuria includes cystitis hemorrhagic and hematuria 4Hematuria includes cystitis hemorrhagic and hematuria Rash includes dermatitis exfoliative, palmar erythema, rash, rash maculo-papular, rash pruritic and skin toxicity 5Rash includes dermatitis exfoliative, palmar erythema, rash, rash maculo-papular, rash pruritic and skin toxicity 6Hemorrhage Intracranial includes hemorrhage intracranial and subarachnoid hemorrhage |
||||
All patients in the TEPADINA-treated and control cohorts developed profound cytopenias, including neutropenia, anemia, thrombocytopenia. Table 4 shows the selected chemistry abnormalities that occurred from start of the preparative regimen through 30 days after transplantation.All patients in the TEPADINA-treated and control cohorts developed profound cytopenias, including neutropenia, anemia, thrombocytopenia. Table 4 shows the selected chemistry abnormalities that occurred from start of the preparative regimen through 30 days after transplantation.
|
| Preparative Regimen of Busulfan and Cyclophosphamide | |||
|
With TEPADINAWith TEPADINA N=25 patients (%) |
Without TEPADINAWithout TEPADINA N=51 patients (%) |
|||
| Adverse Reaction | Any Grade | Grade 3-4 | Any Grade | Grade 3-4 |
| Elevated alanine aminotransferase |
22 (88%)22 (88%)22 (88%)22 (88%) |
6 (24%)6 (24%)6 (24%)6 (24%) |
49 (96%)49 (96%)49 (96%)49 (96%) |
14 (27%)14 (27%)14 (27%)14 (27%) |
| Elevated aspartate aminotransferase |
20 (80%)20 (80%)20 (80%)20 (80%) |
4 (16%)4 (16%)4 (16%)4 (16%) |
45 (88%)45 (88%)45 (88%)45 (88%) |
9 (18%)9 (18%)9 (18%)9 (18%) |
| Elevated total bilirubin |
20 (80%)20 (80%)20 (80%)20 (80%) |
4 (16%)4 (16%)4 (16%)4 (16%) |
39 (77%)39 (77%) |
2 (4%)2 (4%) |
Adverse Reactions with Treatment of adenocarcinoma of the breast, adenocarcinoma of the ovary, malignant effusions and superficial papillary carcinoma of the urinary bladder
: Nausea, vomiting, abdominal pain, anorexia. Gastrointestinal: Nausea, vomiting, abdominal pain, anorexia.
: Fatigue, weakness. Febrile reaction and discharge from a subcutaneous lesion may occur as the result of breakdown of tumor tissue. General: Fatigue, weakness. Febrile reaction and discharge from a subcutaneous lesion may occur as the result of breakdown of tumor tissue.
: Allergic reactions - rash, urticaria, laryngeal edema, asthma, anaphylactic shock, wheezing. Hypersensitivity Reactions: Allergic reactions - rash, urticaria, laryngeal edema, asthma, anaphylactic shock, wheezing.
: Contact dermatitis, pain at the injection site. Local Reactions: Contact dermatitis, pain at the injection site.
: Dizziness, headache, blurred vision. Neurologic: Dizziness, headache, blurred vision.
: Dysuria, urinary retention, chemical cystitis or hemorrhagic cystitis. Renal: Dysuria, urinary retention, chemical cystitis or hemorrhagic cystitis.
: Amenorrhea, interference with spermatogenesis. Reproductive: Amenorrhea, interference with spermatogenesis.
: Prolonged apnea has been reported when succinylcholine was administered prior to surgery, following combined use of thiotepa and other anticancer agents. It was theorized that this was caused by decrease of pseudocholinesterase activity caused by the anticancer drugs. Respiratory: Prolonged apnea has been reported when succinylcholine was administered prior to surgery, following combined use of thiotepa and other anticancer agents. It was theorized that this was caused by decrease of pseudocholinesterase activity caused by the anticancer drugs.
: Dermatitis, alopecia. Skin depigmentation has been reported following topical use. Skin: Dermatitis, alopecia. Skin depigmentation has been reported following topical use.
: Conjunctivitis. Special Senses: Conjunctivitis.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post approval use of TEPADINA in preparative regimens prior to allogeneic or autologous hematopoietic progenitor (stem) cell transplantation (HSCT) in adult and pediatric patients.
Blood and lymphatic system disorders: Febrile bone marrow aplasia.
Cardiac disorder: Bradycardia, cardiac failure congestive, cardio-respiratory arrest, pericardial effusion, pericarditis, right ventricular hypertrophy.
Congenital, familial and genetic disorders: Aplasia.
Ear and labyrinth disorders: Deafness.
Eye disorders: Blindness, eyelid ptosis, papilledema, strabismus.
Gastrointestinal disorders: Ascites, dysphagia, enterocolitis, gastritis, palatal disorder.
General disorders and administration site conditions: Device related infection, gait disturbance, malaise, multi-organ failure, pain.
Hepatobiliary disorders: Hepatomegaly.
Immune system disorders: Bone marrow transplant rejection, immunosuppression.
Infection and infestation: Acute sinusitis, bronchopulmonary aspergillosis, candida sepsis, enterococcal infection, Epstein-Barr virus infection, Escherichia sepsis, Fusarium infection, gastroenteritis, infection, lower respiratory tract infection fungal, lower respiratory tract infection viral, parainfluenza virus infection, Pneumonia legionella, relapsing fever, respiratory tract infection, sepsis, septic shock, Staphylococcal bacteremia, Staphylococcal infection, systemic candida, urinary tract infection.
Injury, poisoning and procedural complications: Refractoriness to platelet transfusion, subdural hematoma.
Investigations: Coagulation test abnormal, hemoglobin decreased, Klebsiella test positive, nuclear magnetic resonance imaging brain abnormal, transaminases increased, weight increased.
Metabolism and nutrition disorders: Hyponatremia.
Neoplasms benign, malignant and unspecified (incl. cysts and polyps): Breast cancer metastatic, central nervous system lymphoma, leukemia recurrent, lymphoma, malignant neoplasm progression, metastatic neoplasm, post transplant lymphoproliferative disorder.
Nervous system disorders: Aphasia, brain injury, bulbar palsy, central nervous system lesion, cerebral microangiopathy, cerebral ventricle dilatation, cerebrovascular accident, cognitive disorder, convulsion, coordination abnormal, encephalitis, encephalopathy, hemiplegia, hypotonia, leukoencephalopathy, memory impairment, motor dysfunction, neurotoxicity, quadriparesis, speech disorder, tremor, VIIth nerve paralysis, white matter lesion.
Psychiatric disorders: Delirium, depression, disorientation, suicidal ideation.
Renal and urinary disorders: Renal failure, nephropathy toxic.
Respiratory, thoracic and mediastinal disorders: Acute respiratory distress, aspiration, dyspnea exertional, interstitial lung disease, lung disorder, pneumonitis, pulmonary arteriopathy, pulmonary sepsis, pulmonary veno-occlusive disease, respiratory distress, respiratory failure, pulmonary hypertension.
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome and toxic epidermal necrolysis.
Vascular disorders: Capillary leak syndrome.
7 DRUG INTERACTIONS
7.1 Effect of Cytochrome CYP3A Inhibitors and Inducers
In vitro studies suggest that thiotepa is metabolized by CYP3A4 and CYP2B6 to its active metabolite TEPA. Avoid coadministration of strong CYP3A4 inhibitors (e.g., itraconazole, clarithromycin, ritonavir) and strong CYP3A4 inducers (e.g., rifampin, phenytoin) with TEPADINA due to the potential effects on efficacy and toxicity [see Clinical Pharmacology (12.2)]. Consider alternative medications with no or minimal potential to inhibit or induce CYP3A4. If concomitant use of strong CYP3A4 modulators cannot be avoided, closely monitor for adverse drug reactions.
7.2 Effect of TEPADINA on Cytochrome CYP2B6 Substrates
In vitro studies suggest that thiotepa inhibits CYP2B6. TEPADINA may increase the exposure of drugs that are substrates of CYP2B6 in patients; however, the clinical relevance of this in vitro interaction is unknown [see Clinical Pharmacology (12.2)].
The administration of thiotepa with cyclophosphamide in patients reduces the conversion of cyclophosphamide to the active metabolite, 4-hydroxycyclophosphamide; the effect appears sequence dependent with a greater reduction in the conversion to 4-hydroxycyclophosphamide when thiotepa is administered 1.5 hours prior to the intravenous administration of cyclophosphamide compared to administration of thiotepa after intravenous cyclophosphamide [see Clinical Pharmacology (12.2)]. The reduction in 4-hydroxycyclophosphamide levels may potentially reduce efficacy of cyclophosphamide treatment.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
TEPADINA can cause fetal harm when administered to a pregnant woman based on findings from animals and the drug’s mechanism of action [ see Clinical Pharmacology (12.1)]. Limited available data with TEPADINA use in pregnant women are insufficient to inform a drug-associated risk of major birth defects and miscarriage. In animal reproduction studies, administration of thiotepa to pregnant rats and rabbits during organogenesis produced teratogenic effects (alterations in embryo development, anomalies of the skeletal system of the fetus) at doses approximately 0.125 and 1 times, respectively, the maximum recommended human daily dose on a mg/m 2 basis. Thiotepa was lethal to rabbit fetuses at approximately 2 times the maximum recommended human therapeutic dose based on body-surface area [see Data]. Consider the benefits and risks of TEPADINA for the mother and possible risks to the fetus when prescribing TEPADINA to a pregnant woman.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Thiotepa given by the IP route in mice at doses ≥ 1 mg/kg (3.2 mg/m
2), approximately 8-fold less than the maximum recommended human therapeutic dose based on body-surface area, and in rats at doses ≥ 3 mg/kg (21 mg/m
2), approximately
equal to the maximum recommended human therapeutic dose based on body-surface area, resulted in various malformations
including neural tube defects, omphalocele, renal agenesis, atresia ani, limb and digit defects, cleft palate, micrognathia, other
skeletal anomalies in the skull, vertebrae and ribs, and reduced skeletal ossification. Thiotepa was lethal to rabbit fetuses at a
dose of 3 mg/kg (41 mg/m
2), approximately 2 times the maximum recommended human therapeutic dose based on bodysurface
area.
8.2 Lactation
Risk Summary
There is no information regarding the presence of thiotepa in human milk, the effects on the breastfed infant, or the effects on milk production.
Because of the potential for serious adverse reactions, including the potential for tumorigenicity shown for thiotepa in animal studies, advise patients not to breastfeed during TEPADINA treatment.
8.3 Females and Males of Reproductive Potential
Pregnancy testing
TEPADINA can cause fetal harm when administered to a pregnant female. Verify the pregnancy status of females of reproductive potential prior to initiating TEPADINA therapy.
Contraception
Females
Advise females of reproductive potential to avoid pregnancy during TEPADINA treatment and for at least 6 months after the final dose of TEPADINA. Advise females to immediately report pregnancy [seeUse in Specific Populations (8.1)].
Males
TEPADINA may damage spermatozoa and testicular tissue, resulting in possible genetic abnormalities. Males with female sexual partners of reproductive potential should use effective contraception during TEPADINA treatment and for at least 1 year after the final dose of TEPADINA [ see Nonclinical Toxicology (13.1)].
Infertility
Based on nonclinical findings, male and female fertility may be compromised by treatment with TEPADINA. Inform male patients about the possibility of sperm conservation before the start of therapy [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of TEPADINA for prevention of graft rejection in pediatric patients undergoing allogeneic HSCT for class 3 beta-thalassemia was established in one prospective study and one retrospective study [ see Clinical Studies (14)] that included 1 infant (1 month to 1 year), 23 children (2 to 11 years) and 13 adolescents (12 to 16 years) who received TEPADINA as part of their preparative regimen. Safety and effectiveness of TEPADINA in neonates have not been established.
Safety and effectiveness of TEPADINA for treatment of adenocarcinoma of the breast, adenocarcinoma of the ovary, malignant effusions and superficial papillary carcinoma of the urinary bladder in pediatric patients have not been established.
8.5 Geriatric Use
The safety and effectiveness of TEPADINA as a preparative regimen prior to allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) for patients with class 3 beta-thalassemia have not been established in geriatric patients. Clinical studies of TEPADINA for this indication did not include subjects aged 65 and over.
Clinical studies of TEPADINA for treatment of adenocarcinoma of the breast, adenocarcinoma of the ovary, malignant effusions and superficial papillary carcinoma of the urinary bladder did not include sufficient numbers of subjects aged 65 and over to determine whether elderly subjects respond differently from younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreasing hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
In patients with moderate (creatinine clearance (CLcr) of 30 mL/min to 59 mL/min) renal impairment, decreased renal excretion may result in increased plasma levels of thiotepa and TEPA [see Clinical Pharmacology (12.2)]. This may result in increased toxicity. Monitor patients with moderate to severe (CLcr < 30 mL/min) renal impairment for signs and symptoms of toxicity following treatment with TEPADINA for an extended period of time.
8.7 Hepatic Impairment
Thiotepa is extensively metabolized in the liver. Patients with moderate (bilirubin levels greater than 1.5 times to 3 times the upper limit of normal and any AST) hepatic impairment may have increased plasma levels of thiotepa [ see Clinical Pharmacology (12.2)] . This may result in toxicity. Monitor patients with moderate to severe (bilirubin levels greater than 3 times upper limit of normal and any AST) hepatic impairment for signs and symptoms of toxicity following treatment with TEPADINA for an extended period of time.
10 OVERDOSE
There is no experience with overdoses of thiotepa. The most important adverse reactions expected in case of overdose are myeloablation and pancytopenia [see Nonclinical Toxicology (13.2)]. There is no known antidote for thiotepa. Monitor the hematological status closely and provide vigorous supportive measures as medically indicated.
11 DESCRIPTION
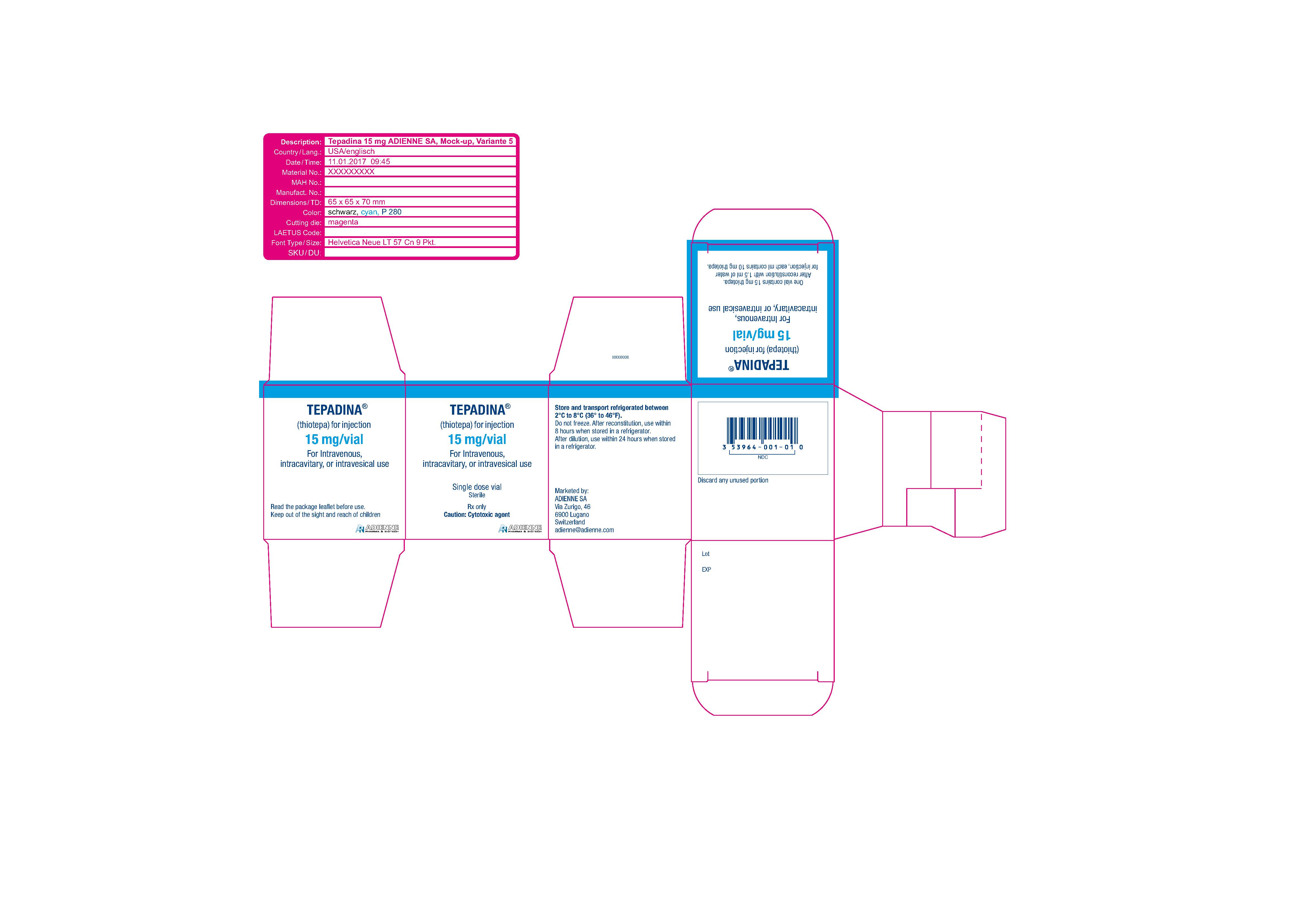
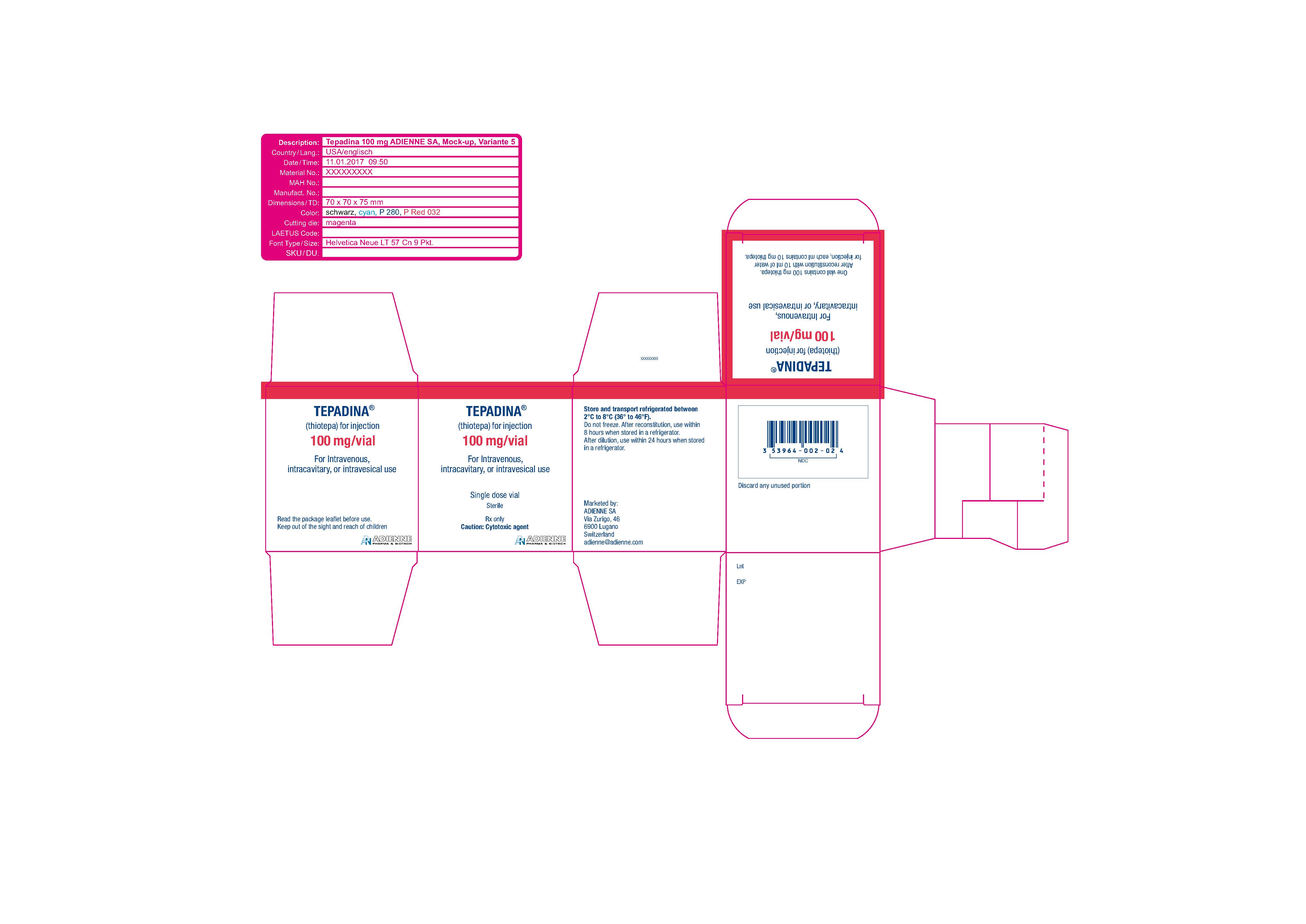
TEPADINA (thiotepa) is an alkylating agent. TEPADINA (thiotepa) for injection is supplied as a non-pyrogenic, sterile lyophilized white powder for intravenous, intracavitary, or intravesical use after reconstitution and dilution.
TEPADINA is available in a single-dose vial containing:
- 15 mg thiotepa. After reconstitution with 1.5 ml of water for injection, each ml contains 10 mg thiotepa.
- 100 mg thiotepa. After reconstitution with 10 ml of water for injection, each ml contains 10 mg thiotepa.
Thiotepa is a synthetic product with antitumor activity. The chemical name for thiotepa is Tris(1-aziridinyl)phosphine sulfide. Thiotepa has the following structural formula:
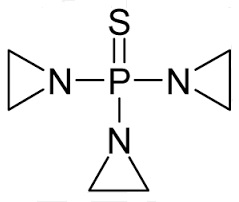
Thiotepa has the molecular formula C 6H 12N 3PS, and a molecular weight of 189.23, and it appears as fine, white crystalline flakes, with a melting range of 52°C to 57°C. It is soluble in water and organic solvents. When reconstituted with sterile water for injection, the resulting solution has a pH of approximately 5.5 to 7.5. Thiotepa is unstable in acid medium.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Thiotepa is a cytotoxic agent of the polyfunctional type, related chemically and pharmacologically to the nitrogen mustard. The radiomimetic action of thiotepa is believed to occur through the release of ethyleneimine radicals which, like irradiation, disrupt the bonds of DNA. One of the principle bond disruptions is initiated by alkylation of guanine at the N-7 position, which severs the linkage between the purine base and the sugar and liberates alkylated guanines.
12.2 Pharmacokinetics
Absorption
Thiotepa reached maximal concentrations close to the end infusion following an intravenous infusion.
Distribution
The binding of thiotepa to plasma proteins is approximately 10% to 20%. The mean volume of distribution (% coefficient of variation) of thiotepa was 30 L/m 2 (44%) or 1.2 L/kg (47%) following a single intravenous infusion of TEPADINA at a dose of 5 mg/kg over 3 hours in pediatric population. In adults administered intravenous thiotepa between 20 mg to 250 mg/m 2 as an intravenous bolus or infusion up to 4 hours, the mean volume of distribution of thiotepa ranged from 1.0 L/kg (30%) to 1.9 L/kg (17%).
Elimination
Following a single intravenous infusion over 3 hours of TEPADINA at a dose of 5 mg/kg in pediatric population, the estimated mean (% coefficient of variation) clearance of thiotepa was 0.58 L/hr/kg (60%) or 13.8 L/hr/m 2 (52%). The mean terminal elimination half-life was 1.7 hours (64%) for thiotepa and 4 hours (29%) for its major active metabolite, N,N',N''-triethylenephosphoramide (TEPA) in pediatric population. In adults administered intravenous thiotepa between 20 mg to 250 mg/m 2 as an intravenous bolus or infusion up to 4 hours, the mean thiotepa clearance ranged from 14.6 L/hr/m 2 (23%) to 27.9 L/hr/m 2 (69%). In adult population, the mean terminal elimination half-life ranged from 1.4 hours (7%) to 3.7 hours (14%) for thiotepa and from 4.9 hours to 17.6 hours (20%) for TEPA.
Metabolism
Thiotepa undergoes hepatic metabolism. In vitro data suggests that CYP3A4 and CYP2B6 may be responsible for the metabolism of thiotepa to TEPA, a major active metabolite.
Excretion
In adult and pediatric patients, urinary excretion of thiotepa accounted for less than 2% of the dose and TEPA accounted for 11% or less of the dose.
Specific Populations
Hepatic Impairment
The clearance of thiotepa following a single TEPADINA dose of 5 mg/kg in pediatric population with mild hepatic impairment was similar to the clearance observed in patients with normal liver function administered thiotepa.
The exposure (as measured by area under the curve (AUC)) of thiotepa increased by 1.6-fold and 1.8-fold following administration of multiple thiotepa doses of 7 mg/kg administered every 2 days with cyclophosphamide in two adult patients who had liver metastases with moderate hepatic impairment compared to the exposure observed in one patient with normal hepatic function . The effect of severe hepatic impairment on thiotepa exposure is unknown.
Renal Impairment
The exposure (as measured by AUC) of thiotepa increased by 1.4-fold and TEPA increased by 2.6-fold following administration of multiple doses of 120 mg/m 2/day in one patient with moderate renal impairment (CLcr = 38 mL/min) administered cyclophosphamide plus thiotepa plus carboplatin, compared to exposure of thiotepa in patients with normal renal function. The effects of severe renal impairment or end-stage renal disease on thiotepa exposure are unknown.
Drug Interactions
The clinical relevance of in vitro inhibition of the cytochrome P450 enzymes described below is unknown, but it cannot be excluded that the systemic exposure of thiotepa or medicinal products that are substrates for these enzymes may be affected with concomitant administration with TEPADINA.
Effect of Cytochrome P450 Modulators on Thiotepa
In vitro data demonstrates that CYP3A4 and CYP2B6 inhibitors decrease the metabolism of thiotepa [see Drug Interactions (7.1)].
Effect of Thiotepa on Cytochrome P450 2B6
In vitro data demonstrates that thiotepa inhibits CYP2B6.
Effect of Thiotepa on Cyclophosphamide
The administration of thiotepa 1.5 hours prior to intravenous cyclophosphamide in patients administered cyclophosphamide plus thiotepa plus carboplatin decreased the AUC of 4-hydroxycyclophosphamide by 26% and maximal concentrations of 4-hydroxycyclophosphamide by 62%, compared to administration of cyclophosphamide prior to thiotepa.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In mice, repeated intraperitoneal (IP) administration of thiotepa (1.15 or 2.3 mg/kg three times per week for 52 or 43 weeks, respectively) produced a significant increase in the combined incidence of squamous-cell carcinomas of the skin, preputial gland, and ear canal, and combined incidence of lymphoma and lymphocytic leukemia. In other studies in mice, repeated IP administration of thiotepa (4 or 8 mg/kg three times per week for 4 weeks followed by a 20 week observation period or 1.8 mg/kg three times per week for 4 weeks followed by a 35 week observation period) resulted in an increased incidence of lung tumors. In rats, repeated IP administration of thiotepa (0.7 or 1.4 mg/kg three times per week for 52 or 34 weeks, respectively) produced significant increases in the incidence of squamous-cell carcinomas of the skin or ear canal, combined hematopoietic neoplasms, and uterine adenocarcinomas. Thiotepa given intravenously (IV) to rats (1 mg/kg once per week for 52 weeks) produced an increased incidence of malignant tumors (abdominal cavity sarcoma, lymphosarcoma myelosis, seminoma, fibrosarcoma, salivary gland hemangioendothelioma, mammary sarcoma, pheochromocytoma) and benign tumors.
The lowest reported carcinogenic dose in mice (1.15 mg/kg, 3.68 mg/m 2) is approximately 7-fold less than the maximum recommended human therapeutic dose based on body-surface area. The lowest reported carcinogenic dose in rats (0.7 mg/kg, 4.9 mg/m 2) is approximately 6-fold less than the maximum recommended human therapeutic dose based on body-surface area.
Thiotepa was mutagenic in in vitro assays in Salmonella typhimurium, E coli, Chinese hamster lung and human lymphocytes. Chromosomal aberrations and sister chromatid exchanges were observed in vitro with thiotepa in bean root tips, human lymphocytes, Chinese hamster lung, and monkey lymphocytes.
Mutations were observed with oral thiotepa in mouse at doses > 2.5 mg/kg (8 mg/m 2). The mouse micronucleus test was positive with intraperitoneal administration of > 1 mg/kg (3.2 mg/m 2). Other positive in vivo chromosomal aberration or mutation assays included Drosophila melanogaster, Chinese hamster marrow, murine marrow, monkey lymphocyte, and murine germ cell.
Thiotepa impaired fertility in male mice at oral or intraperitoneal doses ≥ 0.7 mg/kg (2.24 mg/m 2), approximately 12-fold less than the maximum recommended human therapeutic dose based on body-surface area. Thiotepa (0.5 mg) inhibited implantation in female rats when instilled into the uterine cavity. Thiotepa interfered with spermatogenesis in mice at IP doses ≥ 0.5 mg/kg (1.6 mg/m 2), approximately 17-fold less than the maximum recommended human therapeutic dose based on body-surface area. Thiotepa interfered with spermatogenesis in hamsters at an IP dose of 1 mg/kg (4.1 mg/m 2), approximately 7-fold less than the maximum recommended human therapeutic dose based on body-surface area.
14 CLINICAL STUDIES
TEPADINA was evaluated in a retrospective study of pediatric patients with class 3 beta-thalassemia who underwent allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) from a human leukocyte antigen (HLA)-identical sibling donor. Twenty-five patients (10 male and 15 female) of median age 10 years (range, 5-16 years) were treated with intravenous busulfan at weight-based dosing from Day -10 to Day -7 pretransplant [ see Table 1]), intravenous TEPADINA 5 mg/kg twice on Day -6, intravenous cyclophosphamide 40 mg/kg/day on Day -5 to Day -2, and marrow infusion on Day 0. All patients had also received precytoreduction with hydroxyurea, azathioprine and fludarabine prior to start of the preparative regimen.
Efficacy was based on the incidence of graft rejection (primary or late rejection). The incidence of graft rejection in these 25 patients using TEPADINA was 0% (95% CI: [0, 0.12]). Of the 51 patients who received the same preparative regimen, historically, without TEPADINA, the incidence of graft rejection reported was 25.5% (95% CI: [0.13, 0.37]).
15 REFERENCES
1. OSHA Hazardous Drugs. OSHA. [Accessed from http://www.osha.gov/SLTC/hazardousdrugs/index.html].
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
Hypersensitivity
Counsel patients on the signs and symptoms of hypersensitivity and to seek immediate emergency assistance if they develop any of these signs and symptoms [see Warnings and Precautions (5.2)].
Myelosuppression
Inform patients of the possibility of developing low blood cell counts and the need for hematopoietic progenitor cell infusion. Instruct patients to immediately report to their healthcare provider if bleeding or fever occurs [see Warnings and Precautions (5.1)].
Females and Males of Reproductive Potential
TEPADINA can cause fetal harm. Advise females receiving TEPADINA to avoid pregnancy during TEPADINA treatment and for at least 6 months after the final dose of TEPADINA [see Warnings and Precautions (5.8)].
Advise males with female sexual partners of reproductive potential to use effective contraception during TEPADINA treatment and for at least 1 year after the final dose of TEPADINA [see Use in Specific Populations (8.3)].
Advise females to report pregnancy immediately [see Warnings and Precautions (5.8)].
Advise patients that TEPADINA can produce infertility. Inform male patients about the possibility of sperm conservation before the start of therapy [see Use in Specific Populations (8.3)].
Lactation
Advise patients to avoid breastfeeding while receiving TEPADINA [see Use in Specific Populations (8.2)].
Secondary malignancies
Inform patients that TEPADINA can increase the risk of secondary malignancy [see Warnings and Precautions (5.7)].
Marketed by:
ADIENNE SA
Via Zurigo, 46
6900 Lugano
Switzerland
(Logo)
Revision Date:01/2017
| TEPADINA
thiotepa injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| TEPADINA
thiotepa injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - ADIENNE SA (486304103) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Cenexi-Laboratoires Thissen S.A | 370088959 | analysis(53964-002, 53964-001) , manufacture(53964-002, 53964-001) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| IDT Australia Limited | 743859243 | manufacture(53964-002, 53964-001) , analysis(53964-002, 53964-001) | |