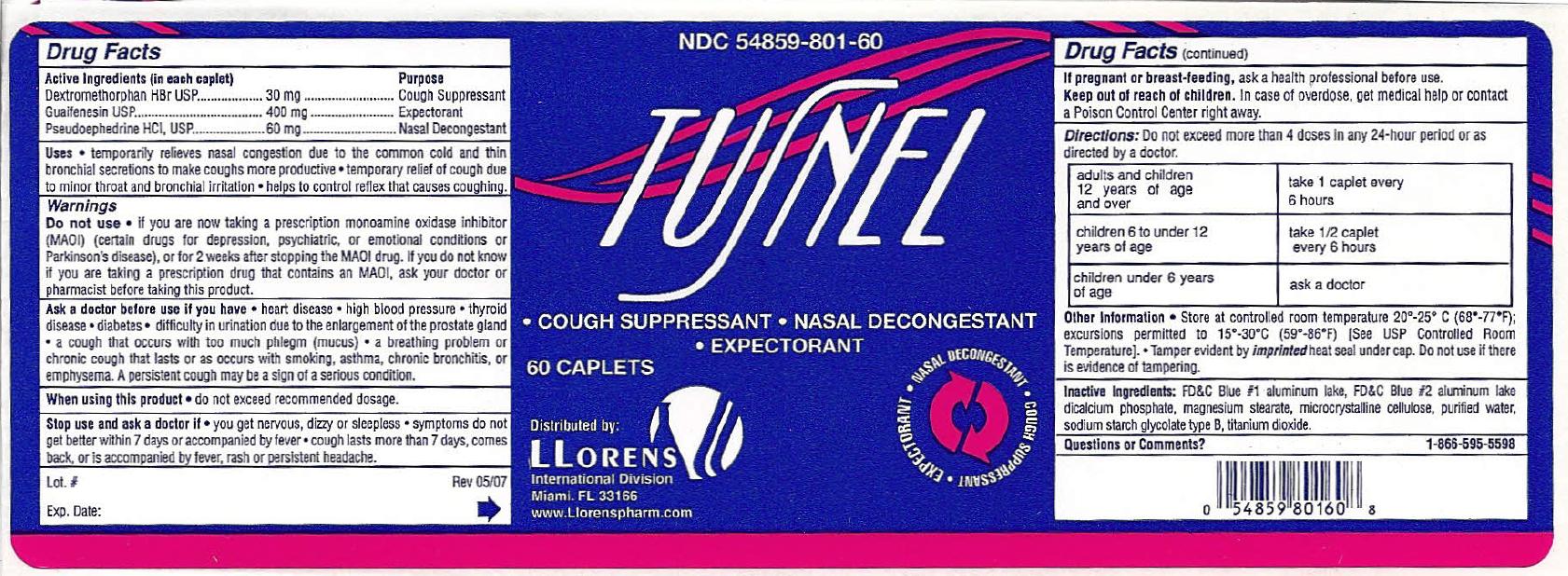
Label: TUSNEL- dextromethorphan hbr, guaifenesin, pseudoephedrine hcl tablet
- NDC Code(s): 54859-801-60
- Packager: LLORENS PHARMACEUTICALS INTERNATIONAL DIVISION
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 23, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients (In each tablet) Purpose
Dextromethorphan HBr ................... 30 mg ................... Cough Suppressant
Guaifenesin .................................. 400 mg .................. Expectorant
Pseudoephedrine HCl .................... 60 mg .................... Nasal Decongestant
- PURPOSE
-
WARNINGS
Warnings
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- difficulty in urination due to the enlargement of the prostate gland
- a cough that occurs with too much phlegm (mucus)
- a breathing problem or chronic cough that lasts or as occurs with smoking, asthma, chronic bronchitis, or emphysema. A persistent cough may be a sign of a serious condition.
When using this product do not exceed recommended dosage.
Stop use and ask a doctor if
- you get nervous, dizzy or sleepless
- symptoms do not get better within 7 days or accompanied by fever
- cough lasts more than 7 days, comes back or is accompanied by fever, rash or persistent headache
-
DO NOT USE
Do not use
- if you are now taking a prescription monamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if you are taking a prescription drug that contains on MAOI, ask your doctor or pharmacist before taking this product.
- if you are now taking a prescription monamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if you are taking a prescription drug that contains on MAOI, ask your doctor or pharmacist before taking this product.
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- DOSAGE & ADMINISTRATION
- INDICATIONS & USAGE
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TUSNEL
dextromethorphan hbr, guaifenesin, pseudoephedrine hcl tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54859-801 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 60 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) WATER (UNII: 059QF0KO0R) SODIUM STARCH GLYCOLATE TYPE B POTATO (UNII: 27NA468985) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape OVAL Size 20mm Flavor Imprint Code TUSNEL Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54859-801-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 07/01/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/01/2007 Labeler - LLORENS PHARMACEUTICALS INTERNATIONAL DIVISION (037342305)