TUSSIN DM- guaifenesin and dextromethorphan hydrobromide solution
Bio-Pharm, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Tussin DM
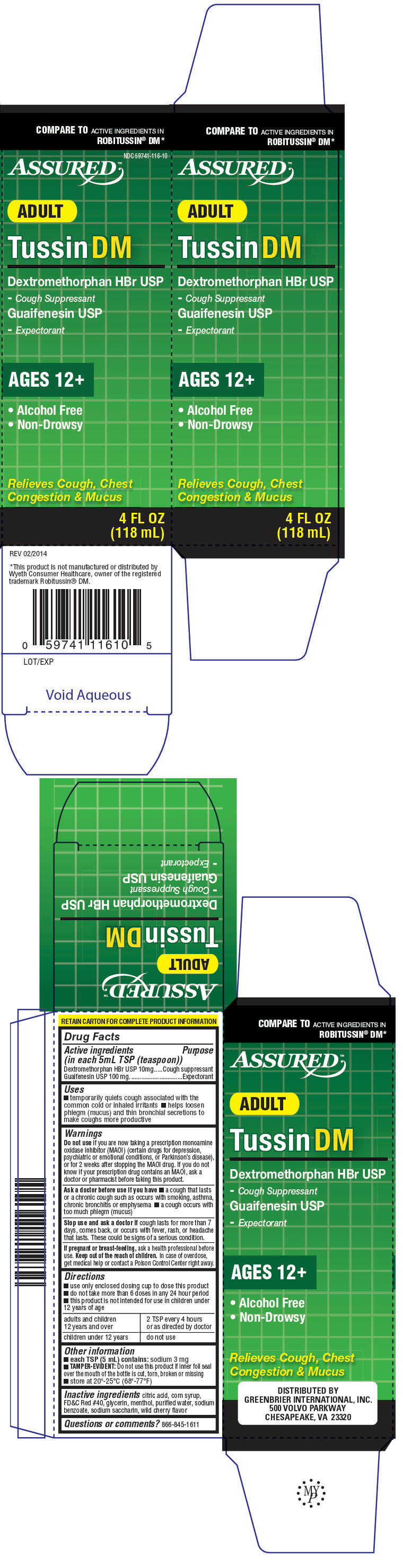
Uses
- temporarily quiets cough associated with the common cold or inhaled irritants
- helps loosen phlegm (mucus) and thin bronchial secretions to make coughs more productive
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- a cough that lasts or a chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
- a cough occurs with too much phlegm (mucus)
Directions
- use only enclosed dosing cup to dose this product
- do not take more than 6 doses in any 24 hour period
- this product is not intended for use in children under 12 years of age
| adults and children 12 years and over | 2 TSP every 4 hours or as directed by doctor |
| children under 12 years | do not use |
Other information
- each TSP (5 mL) contains: sodium 3 mg
- TAMPER-EVIDENT: Do not use this product if inner foil seal over the mouth of the bottle is cut, torn, broken or missing
- store at 20°-25°C (68°-77°F)
Inactive ingredients
citric acid, corn syrup, FD&C Red #40, glycerin, menthol, purified water, sodium benzoate, sodium saccharin, wild cherry flavor
PRINCIPAL DISPLAY PANEL - 118 mL Bottle Carton
COMPARE TO ACTIVE INGREDIENTS IN
ROBITUSSIN® DM*
NDC 59741-116-10
ASSURED™
ADULT
Tussin DM
Dextromethorphan HBr USP
- Cough Suppressant
Guaifenesin USP
- Expectorant
AGES 12+
- Alcohol Free
- Non-Drowsy
Relieves Cough, Chest
Congestion & Mucus
4 FL OZ
(118 mL)
| TUSSIN DM
guaifenesin and dextromethorphan hydrobromide solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Bio-Pharm, Inc. (801652546) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bio-Pharm, Inc. | 801652546 | MANUFACTURE(59741-116) , ANALYSIS(59741-116) , PACK(59741-116) , LABEL(59741-116) | |