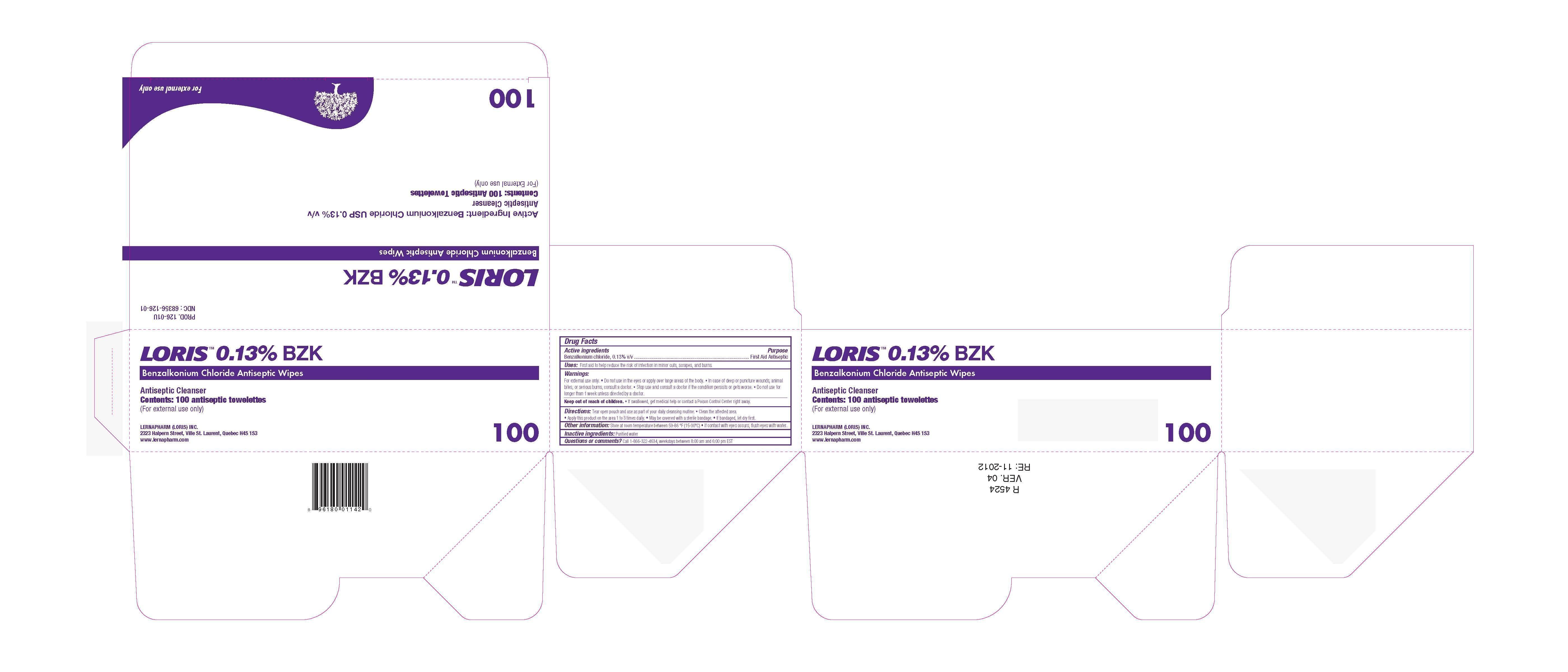
Label: LORIS BZK ANTISEPTIC WIPES- benzalkonium chloride swab
- NDC Code(s): 68356-126-01, 68356-126-02, 68356-126-03, 68356-126-04
- Packager: LernaPharm Loris Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
-
Warnings
For External Use Only
- Directions
- Other Information
- Inactive Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LORIS BZK ANTISEPTIC WIPES
benzalkonium chloride swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68356-126 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68356-126-02 2.6 g in 1 PACKET; Type 0: Not a Combination Product 11/21/2012 2 NDC:68356-126-04 2.6 g in 1 PACKET; Type 0: Not a Combination Product 11/21/2012 3 NDC:68356-126-01 100 in 1 BOX 11/21/2012 3 2.6 g in 1 PACKET; Type 0: Not a Combination Product 4 NDC:68356-126-03 3 in 1 PACKAGE 11/21/2012 4 2.5 g in 1 APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 11/21/2012 Labeler - LernaPharm Loris Inc. (206940905) Establishment Name Address ID/FEI Business Operations LernaPharm Loris Inc. 206940905 manufacture(68356-126) , label(68356-126)