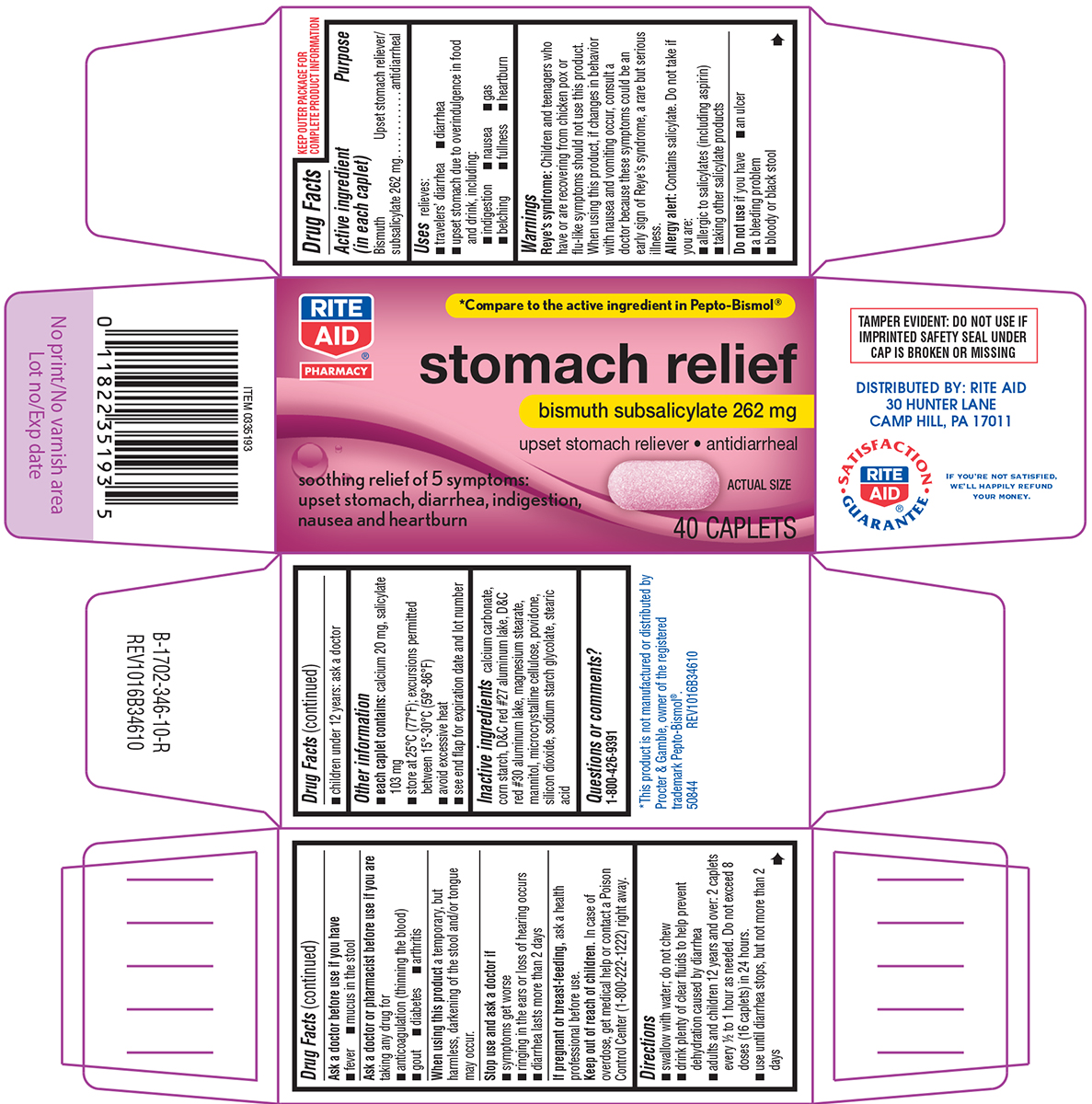
STOMACH RELIEF- bismuth subsalicylate tablet
Rite Aid Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Rite Aid 44-346-Delisted
Uses
relieves:
- travelers' diarrhea
- diarrhea
- upset stomach due to overindulgence in food and drink, including:
- indigestion
- nausea
- gas
- belching
- fullness
- heartburn
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Contains salicylate. Do not take if you are:
- allergic to salicylates (including aspirin)
- taking other salicylate products
Ask a doctor or pharmacist before use if you are
taking any drug for
- anticoagulation (thinning the blood)
- gout
- diabetes
- arthritis
Directions
- swallow with water; do not chew
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- adults and children 12 years and over: 2 caplets every ½ to 1 hour as needed. Do not exceed 8 doses (16 caplets) in 24 hours.
- use until diarrhea stops, but not more than 2 days
- children under 12 years: ask a doctor
Other information
- each caplet contains: calcium 20 mg, salicylate 103 mg
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- avoid excessive heat
- see end flap for expiration date and lot number
Inactive ingredients
calcium carbonate, corn starch, D&C red #27 aluminum lake, D&C red #30 aluminum lake, magnesium stearate, mannitol, microcrystalline cellulose, povidone, silicon dioxide, sodium starch glycolate, stearic acid
Principal Display Panel
RITE
AID®
PHARMACY
*Compare to the active ingredient in Pepto-Bismol®
stomach relief
bismuth subsalicylate 262 mg
upset stomach reliever • antidiarrheal
soothing relief of 5 symptoms:
upset stomach, diarrhea, indigestion,
nausea and heartburn
ACTUAL SIZE
40 CAPLETS
TAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER
CAP IS BROKEN OR MISSING
DISTRIBUTED BY: RITE AID
30 HUNTER LANE
CAMP HILL, PA 17011
SATISFACTION
GUARANTEE
IF YOU'RE NOT SATISFIED,
WE'LL HAPPILY REFUND
YOUR MONEY.
*This product is not manufactured or distributed by
Procter & Gamble, owner of the registered
trademark Pepto-Bismol®.
50844 REV1016B34610
Rite Aid 44-346
| STOMACH RELIEF
bismuth subsalicylate tablet |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Rite Aid Corporation (014578892) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(11822-0346) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(11822-0346) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | PACK(11822-0346) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | PACK(11822-0346) | |