LOVASTATIN- lovastatin tablet
REMEDYREPACK INC.
----------
DESCRIPTION
Lovastatin is a cholesterol lowering agent isolated from a strain of Aspergillus terreus. After oral ingestion, lovastatin, which is an inactive lactone, is hydrolyzed to the correspondingform. This is a principal metabolite and an inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase. This enzyme catalyzes the conversion of HMG-CoA to mevalonate, which is an early and rate limiting step in the biosynthesis of cholesterol.
Lovastatin is [1S-[16-oxo-2H-pyran-2-yl)ethyl]-1-naphthalenyl 2-methylbutanoate. The molecular formula of lovastatin is C24H36O5 and its molecular weight is 404.55. Its structural formula is:
Lovastatin, USP is a white, nonhygroscopic crystalline powder that is insoluble in water and sparingly soluble in ethanol, methanol, and acetonitrile.
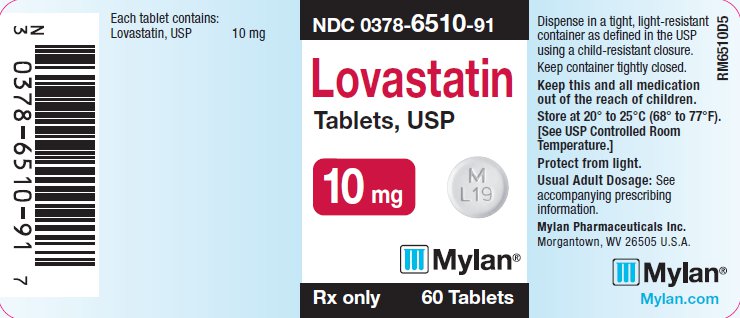
Lovastatin Tablets, USP are supplied as 10 mg, 20 mg and 40 mg tablets for oral administration. In addition to the active ingredient lovastatin, each tablet contains the following inactive ingredients: butylated hydroxyanisole, lactose monohydrate, magnesium stearate, microcrystalline cellulose and pregelatinized starch. Lovastatin Tablets, 20 mg also contain D&C Yellow No. 10 Aluminum Lake. Lovastatin Tablets, 40 mg also contain D&C Red No. 30 Aluminum Lake and D&C Yellow No. 10 Aluminum Lake.
CLINICAL PHARMACOLOGY
The involvement of low density lipoprotein cholesterol (LDL-C) in atherogenesis has been well documented in clinical and pathological studies, as well as in many animal experiments. Epidemiological and clinical studies have established that high LDL-C and low high density lipoprotein cholesterol (HDL-C) are both associated with coronary heart disease. However, the risk of developing coronary heart disease is continuous and graded over the range of cholesterol levels and many coronary events do occur in patients with total cholesterol (total-C) and LDL-C in the lower end of this range.
Lovastatin has been shown to reduce both normal and elevated LDL-C concentrations. LDL is formed from very low density lipoprotein (VLDL) and is catabolized predominantly by the high affinity LDL receptor. The mechanism of the LDL-lowering effect of lovastatin may involve both reduction of VLDL-C concentration, and induction of the LDL receptor, leading to reduced production and/or increased catabolism of LDL-C. Apolipoprotein B also falls substantially during treatment with lovastatin. Since each LDL particle contains one molecule of apolipoprotein B, and since little apolipoprotein B is found in other lipoproteins, this strongly suggests that lovastatin does not merely cause cholesterol to be lost from LDL, but also reduces the concentration of circulating LDL particles. In addition, lovastatin can produce increases of variable magnitude in HDL-C, and modestly reduces VLDL-C and plasma triglycerides (TG) (see
Tables I through III under Clinical Studies
). The effects of lovastatin on Lp(a), fibrinogen, and certain other independent biochemical risk markers for coronary heart disease are unknown.
Lovastatin is a specific inhibitor of HMG-CoA reductase, the enzyme which catalyzes the conversion of HMG-CoA to mevalonate. The conversion of HMG-CoA to mevalonate is an early step in the biosynthetic pathway for cholesterol.
PHARMACOKINETICS
Lovastatin is a lactone which is readily hydrolyzed in vivo to the correspondinga potent inhibitor of HMG-CoA reductase. Inhibition of HMG-CoA reductase is the basis for an assay in pharmacokinetic studies of themetabolites (active inhibitors) and, following base hydrolysis, active plus latent inhibitors (total inhibitors) in plasma following administration of lovastatin.
Following an oral dose of 14C-labeled lovastatin in man, 10% of the dose was excreted in urine and 83% in feces. The latter represents absorbed drug equivalents excreted in bile, as well as any unabsorbed drug. Plasma concentrations of total radioactivity (lovastatin plus 14C-metabolites) peaked at 2 hours and declined rapidly to about 10% of peak by 24 hours postdose. Absorption of lovastatin, estimated relative to an intravenous reference dose, in each of four animal species tested, averaged about 30% of an oral dose. In animal studies, after oral dosing, lovastatin had high selectivity for the liver, where it achieved substantially higher concentrations than in nontarget tissues. Lovastatin undergoes extensive first-pass extraction in the liver, its primary site of action, with subsequent excretion of drug equivalents in the bile. As a consequence of extensive hepatic extraction of lovastatin, the availability of drug to the general circulation is low and variable. In a single-dose study in four hypercholesterolemic patients, it was estimated that less than 5% of an oral dose of lovastatin reaches the general circulation as active inhibitors. Following administration of lovastatin tablets the coefficient of variation, based on between subject variability, was approximately 40% for the area under the curve (AUC) of total inhibitory activity in the general circulation.
Both lovastatin and itsmetabolite are highly bound (> 95%) to human plasma proteins. Animal studies demonstrated that lovastatin crosses the blood-brain and placental barriers.
The major active metabolites present in human plasma are theof lovastatin, its 6'-hydroxy derivative, and two additional metabolites. Peak plasma concentrations of both active and total inhibitors were attained within 2 to 4 hours of dose administration. While the recommended therapeutic dose range is 10 to 80 mg/day, linearity of inhibitory activity in the general circulation was established by a single-dose study employing lovastatin tablet dosages from 60 mg to as high as 120 mg. With a once a day dosing regimen, plasma concentrations of total inhibitors over a dosing interval achieved a steady-state between the second and third days of therapy and were about 1.5 times those following a single-dose. When lovastatin was given under fasting conditions, plasma concentrations of total inhibitors were on average about two-thirds those found when lovastatin was administered immediately after a standard test meal.
In a study of patients with severe renal insufficiency (creatinine clearance 10 to 30 mL/min), the plasma concentrations of total inhibitors after a single-dose of lovastatin were approximately 2-fold higher than those in healthy volunteers.
In a study including sixteen elderly patients between 70 to 78 years of age who received lovastatin 80 mg/day, the mean plasma level of HMG-CoA reductase inhibitory activity was increased approximately 45% compared with eighteen patients between 18 to 30 years of age (see
PRECAUTIONS: Geriatric Use
).
Although the mechanism is not fully understood, cyclosporine has been shown to increase the AUC of HMG-CoA reductase inhibitors. The increase in AUC for lovastatin and lovastatin acid is presumably due, in part, to inhibition of CYP3A4.
The risk of myopathy is increased by high levels of HMG-CoA reductase inhibitory activity in plasma. Potent inhibitors of CYP3A4 can raise the plasma levels of HMG-CoA reductase inhibitory activity and increase the risk of myopathy (see
WARNINGS: Myopathy/Rhabdomyolysis
and
PRECAUTIONS: Drug Interactions
).
Lovastatin is a substrate for cytochrome P450 isoform 3A4 (CYP3A4) (see
PRECAUTIONS: Drug Interactions
). Grapefruit juice contains one or more components that inhibit CYP3A4 and can increase the plasma concentrations of drugs metabolized by CYP3A4. In one study
1
, ten subjects consumed 200 mL of double strength grapefruit juice (one can of frozen concentrate diluted with one rather than three cans of water) three times daily for 2 days and an additional 200 mL double strength grapefruit juice together with and 30 and 90 minutes following a single-dose of 80 mg lovastatin on the third day. This regimen of grapefruit juice resulted in a mean increase in the serum concentration of lovastatin and itsmetabolite (as measured by the area under the concentration time curve) of 15-fold and 5-fold, respectively [as measured using a chemical assayhigh performance liquid chromatography]. In a second study, fifteen subjects consumed one 8 oz glass of single strength grapefruit juice (one can of frozen concentrate diluted with three cans of water) with breakfast for 3 consecutive days and a single-dose of 40 mg lovastatin in the evening of the third day. This regimen of grapefruit juice resulted in a mean increase in the plasma concentration (as measured by the area under the concentration time curve) of active and total HMG-CoA reductase inhibitory activity [using an enzyme inhibition assay both before (for active inhibitors) and after (for total inhibitors) base hydrolysis] of 1.34-fold and 1.36-fold, respectively, and of lovastatin and itsmetabolite [measured using a chemical assayliquid chromatography/tandem mass spectrometrydifferent from that used in the first
1
study] of 1.94-fold and 1.57-fold, respectively. The effect of amounts of grapefruit juice between those used in these two studies on lovastatin pharmacokinetics has not been studied.
1402.
CONTRAINDICATIONS
CONTRAINDICATIONS
Hypersensitivity to any component of this medication.
Active liver disease or unexplained persistent elevations of serum transaminases (see
WARNINGS
).
Pregnancy and Lactation
(See
PRECAUTIONS, Pregnancy
and
Nursing Mothers
.)
Atherosclerosis is a chronic process and the discontinuation of lipid lowering drugs during pregnancy should have little impact on the outcome of long-term therapy of primary hypercholesterolemia. Moreover, cholesterol and other products of the cholesterol biosynthesis pathway are essential components for fetal development, including synthesis of steroids and cell membranes. Because of the ability of inhibitors of HMG-CoA reductase such as lovastatin to decrease the synthesis of cholesterol and possibly other products of the cholesterol biosynthesis pathway, lovastatin is contraindicated during pregnancy and in nursing mothers. Lovastatin should be administered to women of childbearing age only when such patients are highly unlikely to conceive. If the patient becomes pregnant while taking this drug, lovastatin should be discontinued immediately and the patient should be apprised of the potential hazard to the fetus (see
PRECAUTIONS: Pregnancy
).
WARNINGS
Myopathy/Rhabdomyolysis
Lovastatin, like other inhibitors of HMG-CoA reductase, occasionally causes myopathy manifested as muscle pain, tenderness or weakness with creatine kinase (CK) above ten times the upper limit of normal (ULN). Myopathy sometimes takes the form of rhabdomyolysis with or without acute renal failure secondary to myoglobinuria, and rare fatalities have occurred. The risk of myopathy is increased by high levels of HMG-CoA reductase inhibitory activity in plasma.
As with other HMG-CoA reductase inhibitors, the risk of myopathy/rhabdomyolysis is dose related. In a clinical study (EXCEL) in which patients were carefully monitored and some interacting drugs were excluded, there was one case of myopathy among 4,933 patients randomized to lovastatin 20 mg to 40 mg daily for 48 weeks, and four among 1,649 patients randomized to 80 mg daily.
All patients starting therapy with lovastatin, or whose dose of lovastatin is being increased, should be advised of the risk of myopathy and told to report promptly any unexplained muscle pain, tenderness or weakness. Lovastatin therapy should be discontinued immediately if myopathy is diagnosed or suspected. In most cases, muscle symptoms and CK increases resolved when treatment was promptly discontinued. Periodic CK determinations may be considered in patients starting therapy with lovastatin or whose dose is being increased, but there is no assurance that such monitoring will prevent myopathy.
Many of the patients who have developed rhabdomyolysis on therapy with lovastatin have had complicated medical histories, including renal insufficiency usually as a consequence of long standing diabetes mellitus. Such patients merit closer monitoring. Therapy with lovastatin should be temporarily stopped a few days prior to elective major surgery and when any major medical or surgical condition supervenes.
Potent inhibitors of CYP3A4: Lovastatin, like several other inhibitors of HMG-CoA reductase, is a substrate of cytochrome P450 3A4 (CYP3A4). When lovastatin is used with a potent inhibitor of CYP3A4, elevated plasma levels of HMG-CoA reductase inhibitory activity can increase the risk of myopathy and rhabdomyolysis, particularly with higher doses of lovastatin.
The use of lovastatin concomitantly with the potent CYP3A4 inhibitors itraconazole, ketoconazole, erythromycin, clarithromycin, telithromycin, HIV protease inhibitors, nefazodone, or large quantities of grapefruit juice (> 1 quart daily) should be avoided. Concomitant use of other medicines labeled as having a potent inhibitory effect on CYP3A4 should be avoided unless the benefits of combined therapy outweigh the increased risk. If treatment with itraconazole, ketoconazole, erythromycin, clarithromycin or telithromycin is unavoidable, therapy with lovastatin should be suspended during the course of treatment.
Gemfibrozil, particularly with higher doses of lovastatin: The dose of lovastatin should not exceed 20 mg daily in patients receiving concomitant medication with gemfibrozil. The combined use of lovastatin with gemfibrozil should be avoided, unless the benefits are likely to outweigh the increased risks of this drug combination.
Other lipid lowering drugs (other fibrates or1 g/day of niacin): The dose of lovastatin should not exceed 20 mg daily in patients receiving concomitant medication with other fibrates or1 g/day of niacin. Caution should be used when prescribing other fibrates or lipid lowering doses (1 g/day) of niacin with lovastatin, as these agents can cause myopathy when given alone. The benefit of further alterations in lipid levels by the combined use of lovastatin with other fibrates or niacin should be carefully weighed against the potential risks of these combinations.
Cyclosporine or danazol, with higher doses of lovastatin: The dose of lovastatin should not exceed 20 mg daily in patients receiving concomitant medication with cyclosporine or danazol. The benefits of the use of lovastatin in patients receiving cyclosporine or danazol should be carefully weighed against the risks of these combinations.
Amiodarone or verapamil: The dose of lovastatin should not exceed 40 mg daily in patients receiving concomitant medication with amiodarone or verapamil. The combined use of lovastatin at doses higher than 40 mg daily with amiodarone or verapamil should be avoided unless the clinical benefit is likely to outweigh the increased risk of myopathy. The risk of myopathy/rhabdomyolysis is increased when either amiodarone or verapamil is used concomitantly with higher doses of a closely related member of the HMG-CoA reductase inhibitor class.
Prescribing recommendations for interacting agents are summarized in Table VI (see also
CLINICAL PHARMACOLOGY: Pharmacokinetics
,
PRECAUTIONS: Drug Interactions
and
DOSAGE AND ADMINISTRATION
).
Table VI Drug Interactions Associated with Increased Risk of Myopathy/Rhabdomyolysis
ItraconazoleAvoid lovastatinKetoconazoleErythromycinClarithromycinTelithromycinHIV protease inhibitorsNefazodoneGemfibrozilDo not exceed 20 mg lovastatin dailyOther fibratesLipid lowerding doses (g/day) of niacinCyclosporineDanazoleAmiodaroneDo not exceed 40 mg lovastatin dailyVerapamilGrapefruit juiceAvoid large quantities of grapefruit juice (> 1 quart daily)
ADVERSE REACTIONS
). When the drug was interrupted or discontinued in these patients, the transaminase levels usually fell slowly to pretreatment levels. The increases usually appeared 3 to 12 months after the start of therapy with lovastatin, and were not associated with jaundice or other clinical signs or symptoms. There was no evidence of hypersensitivity. In the EXCEL study (see
CLINICAL PHARMACOLOGY: Clinical Studies
), the incidence of persistent increases in serum transaminases over 48 weeks was 0.1% for placebo, 0.1% at 20 mg/day, 0.9% at 40 mg/day, and 1.5% at 80 mg/day in patients on lovastatin. However, in post-marketing experience with lovastatin, symptomatic liver disease has been reported rarely at all dosages (see
ADVERSE REACTIONS
).
In AFCAPS/TexCAPS, the number of participants with consecutive elevations of either alanine aminotransferase (ALT) or aspartate aminotransferase (AST) (> 3 times the upper limit of normal), over a median of 5.1 years of follow-up, was not significantly different between the lovastatin and placebo groups (18 [0.6%] vs. 11 [0.3%]). The starting dose of lovastatin was 20 mg/day; 50% of the lovastatin treated participants were titrated to 40 mg/day at Week 18. Of the 18 participants on lovastatin with consecutive elevations of either ALT or AST, 11 (0.7%) elevations occurred in participants taking 20 mg/day, while 7 (0.4%) elevations occurred in participants titrated to 40 mg/day. Elevated transaminases resulted in discontinuation of 6 (0.2%) participants from therapy in the lovastatin group (n = 3,304) and 4 (0.1%) in the placebo group (n = 3,301).
It is recommended that liver function tests be performed prior to initiation of therapy in patients with a history of liver disease, or when otherwise clinically indicated. It is recommended that liver function tests be performed in all patients prior to use of 40 mg or more daily and thereafter when clinically indicated. Patients who develop increased transaminase levels should be monitored with a second liver function evaluation to confirm the finding and be followed thereafter with frequent liver function tests until the abnormality(ies) returns to normal. Should an increase in AST or ALT of three times the upper limit of normal or greater persist, withdrawal of therapy with lovastatin is recommended.
The drug should be used with caution in patients who consume substantial quantities of alcohol and/or have a past history of liver disease. Active liver disease or unexplained transaminase elevations are contraindications to the use of lovastatin.
As with other lipid lowering agents, moderate (less than three times the upper limit of normal) elevations of serum transaminases have been reported following therapy with lovastatin (see
ADVERSE REACTIONS
). These changes appeared soon after initiation of therapy with lovastatin, were often transient, were not accompanied by any symptoms and interruption of treatment was not required.
- The risk of myopathy/rhabdomyolysis is increased by concomitant use of lovastatin with the following:
PRECAUTIONS
General
Lovastatin may elevate creatine phosphokinase and transaminase levels (see
WARNINGS
and
ADVERSE REACTIONS
). This should be considered in the differential diagnosis of chest pain in a patient on therapy with lovastatin.
Homozygous Familial Hypercholesterolemia
Lovastatin is less effective in patients with the rare homozygous familial hypercholesterolemia, possibly because these patients have no functional LDL receptors. Lovastatin appears to be more likely to raise serum transaminases (see
ADVERSE REACTIONS
) in these homozygous patients.
INFORMATION FOR PATIENTS
Patients should be advised about substances they should not take concomitantly with lovastatin and be advised to report promptly unexplained muscle pain, tenderness, or weakness (see list below and
WARNINGS: Myopathy/Rhabdomyolysis
). Patients should also be advised to inform other physicians prescribing a new medication that they are taking lovastatin.
DRUG INTERACTIONS
Drug Interactions
CYP3A4 Interactions
WARNINGS: Myopathy/Rhabdomyolysis
, and
CLINICAL PHARMACOLOGY: Pharmacokinetics
.
Itraconazole
Ketoconazole
Erythromycin
Clarithromycin
Telithromycin
HIV protease inhibitors
Nefazodone
Large quantities of grapefruit juice (> 1 quart daily)
Interactions With Lipid Lowering Drugs That Can Cause Myopathy When Given Alone
The risk of myopathy is also increased by the following lipid lowering drugs that are not potent CYP3A4 inhibitors, but which can cause myopathy when given alone.
See
WARNINGS: Myopathy/Rhabdomyolysis
.
Gemfibrozil
Other fibrates
Niacin (nicotinic acid) (1 g/day)
Other Drug Interactions
Cyclosporine or Danazol
The risk of myopathy/rhabdomyolysis is increased by concomitant administration of cyclosporine or danazol particularly with higher doses of lovastatin (see
WARNINGS: Myopathy/Rhabdomyolysis
CLINICAL PHARMACOLOGY: Pharmacokinetics).
Amiodarone or Verapamil
The risk of myopathy/rhabdomyolysis is increased when either amiodarone or verapamil is used concomitantly with a closely related member of the HMG-CoA reductase inhibitor class (see
WARNINGS: Myopathy/Rhabdomyolysis).
Coumarin Anticoagulants
In a small clinical trial in which lovastatin was administered to warfarin treated patients, no effect on prothrombin time was detected. However, another HMG-CoA reductase inhibitor has been found to produce a less than two second increase in prothrombin time in healthy volunteers receiving low doses of warfarin. Also, bleeding and/or increased prothrombin time have been reported in a few patients taking coumarin anticoagulants concomitantly with lovastatin. It is recommended that in patients taking anticoagulants, prothrombin time be determined before starting lovastatin and frequently enough during early therapy to insure that no significant alteration of prothrombin time occurs. Once a stable prothrombin time has been documented, prothrombin times can be monitored at the intervals usually recommended for patients on coumarin anticoagulants. If the dose of lovastatin is changed, the same procedure should be repeated. Lovastatin therapy has not been associated with bleeding or with changes in prothrombin time in patients not taking anticoagulants.
Propranolol
In normal volunteers, there was no clinically significant pharmacokinetic or pharmacodynamic interaction with concomitant administration of single doses of lovastatin and propranolol.
Digoxin
In patients with hypercholesterolemia, concomitant administration of lovastatin and digoxin resulted in no effect on digoxin plasma concentrations.
Oral Hypoglycemic Agents
In pharmacokinetic studies of lovastatin in hypercholesterolemic non-insulin-dependent diabetic patients, there was no drug interaction with glipizide or with chlorpropamide (see
CLINICAL PHARMACOLOGY: Clinical Studies).
Endocrine Function
HMG-CoA reductase inhibitors interfere with cholesterol synthesis and as such might theoretically blunt adrenal and/or gonadal steroid production. Results of clinical trials with drugs in this class have been inconsistent with regard to drug effects on basal and reserve steroid levels. However, clinical studies have shown that lovastatin does not reduce basal plasma cortisol concentration or impair adrenal reserve, and does not reduce basal plasma testosterone concentration. Another HMG-CoA reductase inhibitor has been shown to reduce the plasma testosterone response to HCG. In the same study, the mean testosterone response to HCG was slightly but not significantly reduced after treatment with lovastatin 40 mg daily for 16 weeks in 21 men. The effects of HMG-CoA reductase inhibitors on male fertility have not been studied in adequate numbers of male patients. The effects, if any, on the pituitary-gonadal axis in premenopausal women are unknown. Patients treated with lovastatin who develop clinical evidence of endocrine dysfunction should be evaluated appropriately. Caution should also be exercised if an HMG-CoA reductase inhibitor or other agent used to lower cholesterol levels is administered to patients also receiving other drugs (e.g., ketoconazole, spironolactone, cimetidine) that may decrease the levels or activity of endogenous steroid hormones.
CNS Toxicity
Lovastatin produced optic nerve degeneration (Wallerian degeneration of retinogeniculate fibers) in clinically normal dogs in a dose dependent fashion starting at 60 mg/kg/day, a dose that produced mean plasma drug levels about 30 times higher than the mean drug level in humans taking the highest recommended dose (as measured by total enzyme inhibitory activity). Vestibulocochlear Wallerian-like degeneration and retinal ganglion cell chromatolysis were also seen in dogs treated for 14 weeks at 180 mg/kg/day, a dose which resulted in a mean plasma drug level (Cmax) similar to that seen with the 60 mg/kg/day dose.
CNS vascular lesions, characterized by perivascular hemorrhage and edema, mononuclear cell infiltration of perivascular spaces, perivascular fibrin deposits and necrosis of small vessels, were seen in dogs treated with lovastatin at a dose of 180 mg/kg/day, a dose which produced plasma drug levels (Cmax) which were about 30 times higher than the mean values in humans taking 80 mg/day.
Similar optic nerve and CNS vascular lesions have been observed with other drugs of this class.
Cataracts were seen in dogs treated for 11 and 28 weeks at 180 mg/kg/day and one year at 60 mg/kg/day.
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 21-month carcinogenic study in mice, there was a statistically significant increase in the incidence of hepatocellular carcinomas and adenomas in both males and females at 500 mg/kg/day. This dose produced a total plasma drug exposure 3 to 4 times that of humans given the highest recommended dose of lovastatin (drug exposure was measured as total HMG-CoA reductase inhibitory activity in extracted plasma). Tumor increases were not seen at 20 and 100 mg/kg/day, doses that produced drug exposures of 0.3 to 2 times that of humans at the 80 mg/day dose. A statistically significant increase in pulmonary adenomas was seen in female mice at approximately 4 times the human drug exposure. (Although mice were given 300 times the human dose [HD] on a mg/kg body weight basis, plasma levels of total inhibitory activity were only 4 times higher in mice than in humans given 80 mg of lovastatin.)
There was an increase in incidence of papilloma in the nonglandular mucosa of the stomach of mice beginning at exposures of 1 to 2 times that of humans. The glandular mucosa was not affected. The human stomach contains only glandular mucosa.
In a 24-month carcinogenicity study in rats, there was a positive dose-response relationship for hepatocellular carcinogenicity in males at drug exposures between 2 to 7 times that of human exposure at 80 mg/day (doses in rats were 5, 30 and 180 mg/kg/day).
An increased incidence of thyroid neoplasms in rats appears to be a response that has been seen with other HMG-CoA reductase inhibitors.
A chemically similar drug in this class was administered to mice for 72 weeks at 25, 100, and 400 mg/kg body weight, which resulted in mean serum drug levels approximately 3, 15, and 33 times higher than the mean human serum drug concentration (as total inhibitory activity) after a 40 mg oral dose. Liver carcinomas were significantly increased in high dose females and mid and high dose males, with a maximum incidence of 90 % in males. The incidence of adenomas of the liver was significantly increased in mid and high dose females. Drug treatment also significantly increased the incidence of lung adenomas in mid and high dose males and females. Adenomas of the Harderian gland (a gland of the eye of rodents) were significantly higher in high dose mice than in controls.
No evidence of mutagenicity was observed in a microbial mutagen test using mutant strains of Salmonella typhimurium with or without rat or mouse liver metabolic activation. In addition, no evidence of damage to genetic material was noted in an in vitro alkaline elution assay using rat or mouse hepatocytes, a V-79 mammalian cell forward mutation study, an in vitro chromosome aberration study in CHO cells, or an in vivo chromosomal aberration assay in mouse bone marrow.
Drug-related testicular atrophy, decreased spermatogenesis, spermatocytic degeneration and giant cell formation were seen in dogs starting at 20 mg/kg/day. Similar findings were seen with another drug in this class. No drug-related effects on fertility were found in studies with lovastatin in rats. However, in studies with a similar drug in this class, there was decreased fertility in male rats treated for 34 weeks at 25 mg/kg body weight, although this effect was not observed in a subsequent fertility study when this same dose was administered for 11 weeks (the entire cycle of spermatogenesis, including epididymal maturation). In rats treated with this same reductase inhibitor at 180 mg/kg/day, seminiferous tubule degeneration (necrosis and loss of spermatogenic epithelium) was observed. No microscopic changes were observed in the testes from rats of either study. The clinical significance of these findings is unclear.
PREGNANCY
Pregnancy Category X
See
CONTRAINDICATIONS
.
Safety in pregnant women has not been established.
Lovastatin has been shown to produce skeletal malformations in offspring of pregnant mice and rats dosed during gestation at 80 mg/kg/day (affected mouse fetuses/total: 8/307 compared to 4/289 in the control group; affected rat fetuses/total: 6/324 compared to 2/308 in the control group). Female rats dosed before mating through gestation at 80 mg/kg/day also had fetuses with skeletal malformations (affected fetuses/total: 1/152 compared to 0/171 in the control group). The 80 mg/kg/day dose in mice is 7 times the human dose based on body surface area and in rats results in 5 times the human exposure based on AUC. In pregnant rats given doses of 2, 20, or 200 mg/kg/day and treated through lactation, the following effects were observed: neonatal mortality (4.1%, 3.5%, and 46%, respectively, compared to 0.6% in the control group), decreased pup body weights throughout lactation (up to 5%, 8%, and 38%, respectively, below control), supernumerary ribs in dead pups (affected fetuses/total:0/7, 1/17, and 11/79, respectively, compared to 0/5 in the control group), delays in ossification in dead pups (affected fetuses/total: 0/7, 0/17, and 1/79, respectively, compared to 0/5 in the control group) and delays in pup development (delays in the appearance of an auditory startle response at 200 mg/kg/day and free-fall righting reflexes at 20 and 200 mg/kg/day)..
Direct dosing of neonatal rats by subcutaneous injection with 10 mg/kg/day of the open hydroxyacid form of lovastatin resulted in delayed passive avoidance learning in female rats (mean of 8.3 trials to criterion, compared to 7.3 and 6.4 in untreated and vehicle-treated controls; no effects on retention one week later) at exposures 4 times the human systemic exposure at 80 mg/day based on AUC. No effect was seen in male rats. No evidence of malformations was observed when pregnant rabbits were given 5 mg/kg/day (doses equivalent to a human dose of 80 mg/day based on body surface area) or a maternally toxic dose of 15 mg/kg/day (3 times the human dose of 80 mg/day based on body surface area). .
Rare clinical reports of congenital anomalies following intrauterine exposure to HMG-CoA reductase inhibitors have been received. However, in an analysis
3
of greater than 200 prospectively followed pregnancies exposed during the first trimester to lovastatin or another closely related HMG-CoA reductase inhibitor, the incidence of congenital anomalies was comparable to that seen in the general population. This number of pregnancies was sufficient to exclude a 3-fold or greater increase in congenital anomalies over the background incidence.
Maternal treatment with lovastatin may reduce the fetal levels of mevalonate, which is a precursor of cholesterol biosynthesis. Atherosclerosis is a chronic process, and ordinarily discontinuation of lipid-lowering drugs during pregnancy should have little impact on the long-term risk associated with primary hypercholesterolemia For these reasons, lovastatin should not be used in women who are pregnant, or can become pregnant (see
CONTRAINDICATIONS
). Lovastatin should be administered to women of child-bearing potential only when such patients are highly unlikely to conceive and have been informed of the potential hazards. Treatment should be immediately discontinued as soon as pregnancy is recognized.
3
Manson, J.M., Freyssinges, C., Ducrocq, M.B., Stephenson, W.P., Postmarketing Surveillance of Lovastatin and Simavastin Exposure During Pregnancy. Reproductive Toxicology. 10(6):439446, 1996.
NURSING MOTHERS
It is not known whether lovastatin is excreted in human milk. Because a small amount of another drug in this class is excreted in human breast milk and because of the potential for serious adverse reactions in nursing infants, women taking lovastatin should not nurse their infants (see
CONTRAINDICATIONS
).
PEDIATRIC USE
Safety and effectiveness in patients 10 to 17 years of age with heFH have been evaluated in controlled clinical trials of 48 weeks duration in adolescent boys and controlled clinical trials of 24 weeks duration in girls who were at least one year postmenarche. Patients treated with lovastatin had an adverse experience profile generally similar to that of patients treated with placebo. Doses greater than 40 mg have not been studied in this population. In these limited controlled studies, there was no detectable effect on growth or sexual maturation in the adolescent boys or on menstrual cycle length in girls. See
CLINICAL PHARMACOLOGY: Clinical Studies in Adolescent Patients ADVERSE REACTIONS: Adolescent Patients DOSAGE AND ADMINISTRATION, Adolescent Patients (10 to 17 years of age) with Heterozygous Familial Hypercholesterolemia. Adolescent females should be counseled on appropriate contraceptive methods while on lovastatin therapy (see CONTRAINDICATIONSand PRECAUTIONS: Pregnancy). Lovastatin has not been studied in prepubertal patients or patients younger than 10 years of age.
GERIATRIC USE
65 years of age. Lipid lowering efficacy with lovastatin was at least as great in elderly patients compared with younger patients, and there were no overall differences in safety over the 20 to 80 mg/day dosage range (see
CLINICAL PHARMACOLOGY
).
ADVERSE REACTIONS
Phase III Clinical Studies
In Phase III controlled clinical studies involving 613 patients treated with lovastatin, the adverse experience profile was similar to that shown below for the 8,245 patient EXCEL study (see Expanded Clinical Evaluation of Lovastatin (EXCEL) Study).
Persistent increases of serum transaminases have been noted (see
WARNINGS: Liver Dysfunction
). About 11% of patients had elevations of CK levels of at least twice the normal value on one or more occasions. The corresponding values for the control agent cholestyramine was 9 %. This was attributable to the noncardiac fraction of CK. Large increases in CK have sometimes been reported (see
WARNINGS: Myopathy/Rhabdomyolysis
).
Expanded Clinical Evaluation of Lovastatin (EXCEL) Study
Lovastatin was compared to placebo in 8,245 patients with hypercholesterolemia (total-C 240 to 300 mg/dL [6.2 to 7.8 mmol/L]) in the randomized, double-blind, parallel, 48-week EXCEL study. Clinical adverse experiences reported as possibly, probably or definitely drug-related in1% in any treatment group are shown in the table below. For no event was the incidence on drug and placebo statistically different.
PlaceboLovastatinLovastatinLovastatinLovastatin20mg q.p.m.40mg q.p.m.20mg b.i.d.40mg b.i.d.(N=1663)(N=1642)(N=1645)(N=1646)(N=1649)%%%%%
Body As a WholeAsthenia1.41.71.41.51.2GastrointestinalAbdominal pain1.6222.22.5Constipation1.923.23.23.5Diarrhea2.32.62.42.22.6Dyspepsia1.91.31.311.6Flatulence4.23.74.33.94.5Nausea2.51.92.52.22.2MusculoskeletalMuscle cramps0.50.60.81.11Myalgia1.72.61.82.23Nervous System/ PsychiatricDizziness0.70.71.20.50.5Headache2.72.62.82.13.2SkinRash0.70.811.21.3Special SensesBlurred vision0.81.10.90.91.2
In the EXCEL study (see
CLINICAL PHARMACOLOGY: Clinical Studies
), 4.6% of the patients treated up to 48 weeks were discontinued due to clinical or laboratory adverse experiences which were rated by the investigator as possibly, probably or definitely related to therapy with lovastatin. The value for the placebo group was 2.5%.
Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS)
In AFCAPS/TexCAPS (see
CLINICAL PHARMACOLOGY: Clinical Studies
) involving 6,605 participants treated with 20 to 40 mg/day of lovastatin (n = 3,304) or placebo (n = 3,301), the safety and tolerability profile of the group treated with lovastatin was comparable to that of the group treated with placebo during a median of 5.1 years of follow-up. The adverse experiences reported in AFCAPS/TexCAPS were similar to those reported in EXCEL (see
ADVERSE REACTIONS: Expanded Clinical Evaluation of Lovastatin (EXCEL) Study
).
Concomitant Therapy
In controlled clinical studies in which lovastatin was administered concomitantly with cholestyramine, no adverse reactions peculiar to this concomitant treatment were observed. The adverse reactions that occurred were limited to those reported previously with lovastatin or cholestyramine. Other lipid lowering agents were not administered concomitantly with lovastatin during controlled clinical studies. Preliminary data suggests that the addition of gemfibrozil to therapy with lovastatin is not associated with greater reduction in LDL-C than that achieved with lovastatin alone. In uncontrolled clinical studies, most of the patients who have developed myopathy were receiving concomitant therapy with cyclosporine, gemfibrozil or niacin (nicotinic acid). The combined use of lovastatin at doses exceeding 20 mg/day with cyclosporine gemfibrozil, other fibrates or lipid lowering doses (1 g/day) of niacin should be avoided (see
WARNINGS: Myopathy/Rhabdomyolysis
).
The following effects have been reported with drugs in this class. Not all the effects listed below have necessarily been associated with lovastatin therapy.
Skeletal: muscle cramps, myalgia, myopathy, rhabdomyolysis, arthralgias
Neurological: dysfunction of certain cranial nerves (including alteration of taste, impairment of extraocular movement, facial paresis), tremor, dizziness, vertigo, memory loss, paresthesia, peripheral neuropathy, peripheral nerve palsy, psychic disturbances, anxiety, insomnia, depression
Hypersensitivity Reactions: An apparent hypersensitivity syndrome has been reported rarely which has included one or more of the following features: anaphylaxis, angioedema, lupus erythematous-like syndrome, polymyalgia rheumatica, dermatomyositis, vasculitis, purpura, thrombocytopenia, leukopenia, hemolytic anemia, positive ANA, ESR increase, eosinophilia, arthritis, arthralgia, urticaria, asthenia, photosensitivity, fever, chills, flushing, malaise, dyspnea, toxic epidermal necrolysis, erythema multiforme, including Stevens-Johnson Syndrome
Gastrointestinal: pancreatitis, hepatitis, including chronic active hepatitis, cholestatic jaundice, fatty change in liver; and rarely, cirrhosis, fulminant hepatic necrosis, and hepatoma; anorexia, vomiting
Skin: alopecia, pruritus. A variety of skin changes (e.g., nodules, discoloration, dryness of skin/mucous membranes, changes to hair/nails) have been reported
Reproductive: gynecomastia, loss of libido, erectile dysfunction
Eye: progression of cataracts (lens opacities), ophthalmoplegia
Laboratory Abnormalities: elevated transaminases, alkaline phosphatase,
Adolescent Patients (ages 10 to 17 years)
In a 48-week controlled study in adolescent boys with heFH (n = 132) and a 24-week controlled study in girls who were at least one year postmenarche with heFH (n = 54), the safety and tolerability profile of the groups treated with lovastatin (10 mg to 40 mg daily) was generally similar to that of the groups treated with placebo (see
CLINICAL PHARMACOLOGY: Clinical Studies in Adolescent Patients
and
PRECAUTIONS: Pediatric Use
).
OVERDOSAGE
After oral administration of lovastatin to mice, the median lethal dose observed was > 15 g/m2.
Five healthy human volunteers have received up to 200 mg of lovastatin as a single dose without clinically significant adverse experiences. A few cases of accidental overdosage have been reported; no patients had any specific symptoms, and all patients recovered without sequelae. The maximum dose taken was 5 g to 6 g.
Until further experience is obtained, no specific treatment of overdosage with lovastatin can be recommended.
The dialyzability of lovastatin and its metabolites in man is not known at present.
DOSAGE & ADMINISTRATION
The patient should be placed on a standard cholesterol lowering diet before receiving lovastatin and should continue on this diet during treatment with lovastatin (see
NCEP Treatment Guidelines
for details on dietary therapy). Lovastatin should be given with meals.
Adult Patients
NCEP Guidelines
and
CLINICAL PHARMACOLOGY
). Patients requiring reductions in LDL-C of 20% or more to achieve their goal (see
INDICATIONS AND USAGE
) should be started on 20 mg/day of lovastatin. A starting dose of 10 mg may be considered for patients requiring smaller reductions. Adjustments should be made at intervals of 4 weeks or more.
Cholesterol levels should be monitored periodically and consideration should be given to reducing the dosage of lovastatin if cholesterol levels fall significantly below the targeted range.
Dosage in Patients taking Cyclosporine or Danazol
In patients taking cyclosporine or danazol concomitantly with lovastatin (see
WARNINGS: Myopathy/Rhabdomyolysis
), therapy should begin with 10 mg of lovastatin and should not exceed 20 mg/day.
Dosage in Patients taking Amiodarone or Verapamil
In patients taking amiodarone or verapamil concomitantly with lovastatin, the dose should not exceed 40 mg/day (see
WARNINGS: Myopathy/Rhabdomyolysis
and
PRECAUTIONS: Drug Interactions: Other Drug Interactions
).
Adolescent Patients (10 to 17 years of age) with Heterozygous Familial Hypercholesterolemia
NCEP Pediatric Panel Guidelines4
CLINICAL PHARMACOLOGY
and
INDICATIONS AND USAGE
). Patients requiring reductions in LDL-C of 20% or more to achieve their goal should be started on 20 mg/day of lovastatin. A starting dose of 10 mg may be considered for patients requiring smaller reductions. Adjustments should be made at intervals of 4 weeks or more.
4
National Cholesterol Education Program (NCEP): Highlights of the Report of the Expert Panel on Blood Cholesterol Levels in Children and Adolescents. Pediatrics. 89(3):495501. 1992.
Concomitant Lipid Lowering Therapy
Lovastatin is effective alone or when used concomitantly with bile-acid sequestrants. If lovastatin is used in combination with gemfibrozil, other fibrates or lipid lowering doses (1g/day) of niacin, the dose of lovastatin should not exceed 20 mg/day (see
WARNINGS: Myopathy/Rhabdomyolysis
and
PRECAUTIONS: Drug Interactions
).
Dosage in Patients with Renal Insufficiency
In patients with severe renal insufficiency (creatinine clearance < 30 mL/min), dosage increases above 20 mg/day should be carefully considered and, if deemed necessary, implemented cautiously (see
CLINICAL PHARMACOLOGY
and
WARNINGS: Myopathy/Rhabdomyolysis
).
HOW SUPPLIED
Lovastatin Tablets, USP are available containing 10 mg, 20 mg or 40 mg of lovastatin, USP.
The 10 mg tablets are white to off-white, round, unscored tablets debossed with M over L19 on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-6510-91
bottles of 60 tablets
The 20 mg tablets are yellow, round, unscored tablets debossed with M over L20 on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-6520-91
bottles of 60 tablets
NDC 0378-6520-05
bottles of 500 tablets
The 40 mg tablets are pink, round, unscored tablets debossed with M over L21 on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-6540-91
bottles of 60 tablets
NDC 0378-6540-05
bottles of 500 tablets
Store at 20to 25(68to 77[See USP for Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505
REVISED JANUARY 2008
LOV:R8
| LOVASTATIN
lovastatin tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - REMEDYREPACK INC. (829572556) |
 DRUG: Lovastatin
DRUG: Lovastatin