ASEPSO GENTLE ANTIBACTERIAL- chloroxylenol soap
CODAA Switzerland / Euro Sensations
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
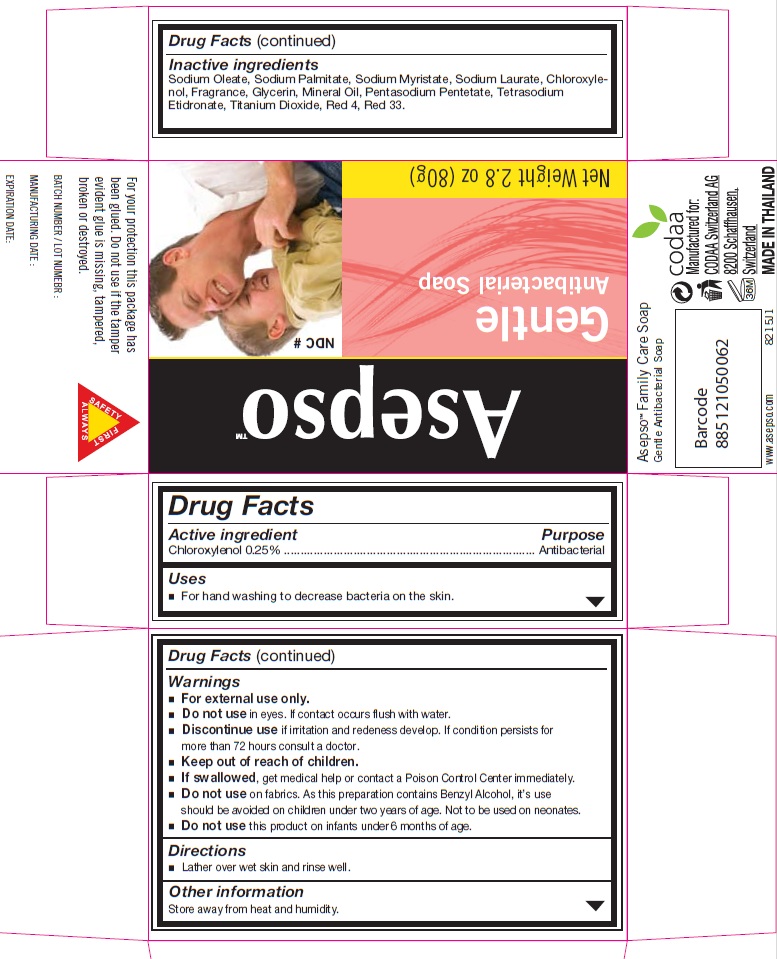
Asepso Gentle Antibacterial Soap
Warnings
For external use only.
Do not use in eyes. If contact occurs flush with water.
Discontinue use if irritation and redness develop. If condition persists for more than 72 hours consult a doctor.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Do not use on fabrics. As this preparation contains Benzyl Alcohol, it's use should be avoided on children under two years of age. Not to be used on neonates.
Do not use this product on infants under 6 months of age.
Inactive ingredients
Sodium Oleate, Sodium Palmitate, Sodium Mrystate, Sodium Laurate ,
Chloroxylenol, Fragrance, Glycerin, Mineral Oil, Pentasodium Pentetate, Tetrasodium Etidronate,
Titanium Dioxide, Red 4 , Red 33
For your protection this package has been glued. Do not use if the tamper
evident glue is missing, tampered, broken or destroyed.
BATCH NUMBER LOT NUMBER
MANUFACTURING DATE
EXPIRATION DATE
| ASEPSO GENTLE ANTIBACTERIAL
chloroxylenol soap |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - CODAA Switzerland / Euro Sensations (483467697) |