MENVEO- meningococcal (groups a, c, y and w-135) oligosaccharide diphtheria crm197 conjugate vaccine
Novartis Vaccines and Diagnostics s.r.l.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use MENVEO safely and effectively. See full prescribing information for MENVEO.
MENVEO® [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM197 Conjugate Vaccine] Solution for intramuscular injection Initial U.S. Approval: 2010 RECENT MAJOR CHANGESINDICATIONS AND USAGEMENVEO is a vaccine indicated for active immunization to prevent invasive meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135. MENVEO is approved for use in persons 2 months through 55 years of age. MENVEO does not prevent N. meningitidis serogroup B infections. (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSSolution for intramuscular injection supplied as a lyophilized Men A conjugate vaccine component to be reconstituted with the accompanying Men CYW-135 liquid conjugate vaccine component. A single dose, after reconstitution is 0.5 mL (3) CONTRAINDICATIONSSevere allergic reaction (e.g., anaphylaxis) after a previous dose of MENVEO, any component of this vaccine, or any other CRM197, diphtheria toxoid or meningococcal-containing vaccine is a contraindication to administration of MENVEO. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Novartis Vaccines at 1-877-683-4732 or VAERS at 1-800-822-7967 or http://vaers.hhs.gov. DRUG INTERACTIONSDo not mix MENVEO or any of its components with any other vaccine or diluent in the same syringe or vial. (7.1) USE IN SPECIFIC POPULATIONSPregnancy: Safety and effectiveness have not been established in pregnant women. Pregnancy registry available at +1 877-413-4759. (8.1) See 17 for PATIENT COUNSELING INFORMATION. Revised: 10/2016 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
MENVEO is a vaccine indicated for active immunization to prevent invasive meningococcal disease caused by Neisseria meningitidis serogroups A, C, Y and W-135. MENVEO is approved for use in persons 2 months through 55 years of age.
MENVEO does not prevent N. meningitidis serogroup B infections.
2 DOSAGE AND ADMINISTRATION
For intramuscular injection only.
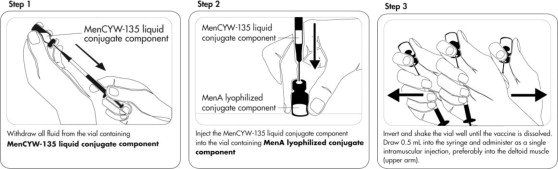
2.1 Reconstitution Instructions
MENVEO is supplied in two vials that must be combined prior to administration. MENVEO must be prepared for administration by reconstituting the MenA lyophilized conjugate vaccine component with the MenCYW-135 liquid conjugate vaccine component. Using a graduated syringe, withdraw the entire contents of the vial of MenCYW-135 liquid conjugate component and inject into the MenA lyophilized conjugate component vial. Invert the vial and shake well until the vaccine is dissolved and then withdraw 0.5mL of reconstituted product.
Please note that it is normal for a small amount of liquid to remain in the vial following withdrawal of the dose.
Following reconstitution, the vaccine is a clear, colorless solution, free from visible foreign particles. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If any of these conditions exist, MENVEO should not be administered.
The reconstituted vaccine should be used immediately, but may be held at or below 77°F (25°C) for up to 8 hours.
2.2 Administration
Each MENVEO dose should be administered as a single 0.5 mL intramuscular injection, preferably into the anterolateral aspect of the thigh in infants or into the deltoid muscle (upper arm) in toddlers, adolescents and adults. Do not administer MENVEO intravenously, subcutaneously or intradermally.
2.3 Dosing Schedule
The dosing schedule for individuals initiating vaccination is as follows:
Infants 2 Months of Age
MENVEO is to be administered as a four-dose series at 2, 4, 6, and 12 months of age.
Children 7 Months through 23 Months of Age
MENVEO is to be administered as a two-dose series with the second dose administered in the second year of life and at least three months after the first dose.
Children 2 Years through 10 Years of Age
MENVEO is to be administered as a single dose. For children 2 years through 5 years of age at continued high risk of meningococcal disease, a second dose may be administered 2 months after the first dose.
Adolescents and Adults 11 Years through 55 Years of Age
MENVEO is to be administered as a single dose.
3 DOSAGE FORMS AND STRENGTHS
MENVEO is a solution for intramuscular injection supplied as a lyophilized MenA conjugate vaccine component to be reconstituted with the accompanying MenCYW-135 liquid conjugate vaccine component. A single dose, after reconstitution, is 0.5 mL. [See Dosage and Administration (2), How Supplied/Storage and Handling (16)].
4 CONTRAINDICATIONS
Severe allergic reaction (e.g., anaphylaxis) after a previous dose of MENVEO, any component of this vaccine, or any other CRM197, diphtheria toxoid or meningococcal-containing vaccine is a contraindication to administration of MENVEO. [See Description (11)].
5 WARNINGS AND PRECAUTIONS
5.1 Management of Acute Allergic Reactions
Appropriate medical treatment must be available should an acute allergic reaction, including an anaphylactic reaction, occur following administration of MENVEO.
5.2 Syncope
Syncope, sometimes resulting in falling injury associated with seizure-like movements has been reported following vaccination with MENVEO. Vaccinees should be observed for at least 15 minutes after vaccine administration to prevent and manage syncopal reactions.
5.3 Altered Immunocompetence
Safety and effectiveness of MENVEO have not been evaluated in immunocompromised persons. If MENVEO is administered to immunocompromised persons, including those receiving immunosuppressive therapy, the expected immune response may not be obtained.
5.4 Guillain-Barré Syndrome
Guillain-Barré syndrome (GBS) has been reported in temporal relationship following administration of another U.S.-licensed meningococcal quadrivalent polysaccharide conjugate vaccine. The decision to administer MENVEO to subjects with a known history of Guillain-Barré Syndrome should take into account the potential benefits and risks.
5.5 Apnea in Premature Infants
Apnea following intramuscular vaccination has been observed in some infants born prematurely. The decision about when to administer an intramuscular vaccine, including MENVEO, to an infant born prematurely should be based on consideration of the individual infant's medical status, and the potential benefits and possible risks of vaccination.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
Children 2 Months Through 23 Months of Age
The safety of MENVEO in infants vaccinated at 2, 4, 6 and 12 months of age was evaluated in three randomized multicenter clinical studies 1-3 conducted in the U.S., Australia, Canada, Taiwan and several countries of Latin America in which 8735 infants received at least one dose of MENVEO and routine infant vaccines (diphtheria toxoid, acellular pertussis, tetanus toxoid, inactivated polio types 1, 2 and 3, hepatitis B, Haemophilus influenzae type b (Hib) antigens; pentavalent rotavirus, and 7-valent pneumococcal conjugate). With dose 4 of MENVEO, toddlers received concomitantly the following vaccines: 7-valent pneumococcal conjugate, measles, mumps, rubella and varicella, and inactivated hepatitis A. A total of 2864 infants in these studies received the routine infant/toddler vaccines only. The infants who received MENVEO were Caucasian (33%), Hispanic (44%), African American (8%), Asian (8%) and other racial/ethnic groups (7%); 51% were male, with a mean age of 65.1 days (SD 7.5 days) at the time of first vaccination.
Safety data for administration of two doses of MENVEO in children between 6 through 23 months of age are available from three randomized studies 1, 4, 5 conducted in the U.S., Latin America, and Canada, of which one U.S. study specifically addressed the safety of MENVEO administered concomitantly with measles, mumps, rubella and varicella vaccine (MMRV). The 1985 older infants and toddlers who received two doses of MENVEO were Caucasian (49%), Hispanic (32%), African American (11%), and other racial/ethnic groups (8%), 51% male, with a mean age of 10.1 months (SD 2.0 months).
Children 2 Years Through 10 Years of Age
The safety of MENVEO in children 2 years through 10 years of age was evaluated in four clinical trials 6-9 conducted in North America (66%), Latin America (28%) and Europe (6%) in which 3181 subjects received MENVEO and 2116 subjects received comparator vaccines (either Meningococcal Polysaccharide Vaccine, Groups A, C, Y and W-135 Combined - Menomune®, Sanofi Pasteur [N=861], or Meningococcal (Groups A, C, Y and W-135) Polysaccharide Diphtheria Toxoid Conjugate Vaccine - Menactra®, Sanofi Pasteur [N=1255]). The subjects 2 years through 10 years of age who received MENVEO were Caucasian (69%), Hispanic (13%), African American (7%), and other racial/ethnic groups (6%), 51% male, with a mean age of 5.2 years. The safety of a second dose of MENVEO administered 2 months following a first dose was studied in 351 children 2 years through 5 years of age.
Adolescents and Adults
The safety of MENVEO in individuals 11 through 55 years of age was evaluated in five randomized controlled clinical trials 10-14 in which 6185 participants received MENVEO alone (5286 participants), MENVEO concomitant with other vaccine(s) (899 participants), or a U.S. licensed comparator vaccine (1966 participants). In the concomitant trials 11, 14 MENVEO was given with vaccines containing: tetanus toxoid, diphtheria toxoid and pertussis (Tdap), or Tdap with human papillomavirus (HPV). The comparator vaccine was either Menomune (209 participants) or Menactra (1757 participants). The trials were conducted in North America (46%), Latin America (41%) and Europe (13%). In two of the studies, subjects received concomitant vaccination with Tdap, or with Tdap plus HPV. Overall, subjects were Caucasian (50%), followed by Hispanic (40%), African American (7%), and other racial/ethnic groups (3%). Among MENVEO recipients, 61%, 17% and 22% were in the 11 through 18 year, 19 through 34 year and 35 through 55 year age groups, respectively, with a mean age of 23.5 years (SD 12.9 years). Among Menactra recipients, 31%, 32% and 37% were in the 11 through 18 year, 19 through 34 year and 35 through 55 year age groups, respectively, with a mean age of 29.2 years (SD 13.4 years). Among Menomune recipients, 100% were in the 11 through 18 year age group, and the mean age was 14.2 years (SD 1.8 years).
In most trials, solicited local and systemic adverse reactions were monitored daily for 7 days following each (one or more) vaccination and recorded on a diary card. Participants were monitored for unsolicited adverse events which included adverse events requiring a physician visit or Emergency Department visit (i.e. medically-attended) or which led to a subject's withdrawal from the study. Among children, adolescents and adults aged 2 years to 55 years, medically significant adverse events and serious adverse events (SAE) were monitored for 6 months after vaccination. Across the studies of infants and toddlers aged 2 months through 23 months, either all medically-attended or all medically-significant adverse events were collected in the period between the infant dose(s) and the toddler doses and during the 6 month period after the toddler dose.
Solicited Adverse Reactions
The reported frequencies of solicited local and systemic adverse reactions from US infants in the largest multinational MENVEO safety study 2 are presented in Table 1. Among the US participants in the MENVEO with routine vaccines group, 51% were female; 64% were Caucasian, 12% were African-American, 15% were Hispanic, 2% were Asian, and 7% were of other racial/ethnic groups.
In infants initiating vaccination at 2 months of age and receiving the four-dose series, common solicited adverse reactions (≥ 10%) were tenderness (24-41%) and erythema at injection site (11-15%), irritability (42-57%), sleepiness (29-50%), persistent crying (21-41%), change in eating habits (17-23%), vomiting (5-11%) and diarrhea (8-16%). The rates of solicited adverse reactions reported for subjects aged 2 months and above receiving MENVEO with routine vaccines at 2, 4, 6 and 12 months of age were comparable to rates among subjects who only received routine vaccines.
| Clinicaltrials.gov Identifier NCT00806195 2 | ||||||||
| N= number of subjects who completed the diary card for a given symptom at the specified vaccination. | ||||||||
| a As Treated Safety Sub-population = US children who received at least one dose of study vaccine and whose diary cards were completed per protocol and returned to the site. | ||||||||
| b Routine infant/toddler vaccines include DTaP-IPV-Hib and PCV7 at doses 1,2,3 and PCV7, MMRV and Hepatitis A vaccines at dose 4. HBV and rotavirus vaccines were allowed according to ACIP recommendations. | ||||||||
| c Local reactogenicity of MENVEO and PCV7 was assessed. | ||||||||
| d Tenderness, severe = cried when injected limb moved. | ||||||||
| e Irritability, severe = unable to console. | ||||||||
| f Sleepiness, severe = sleeps most of the time, hard to arouse. | ||||||||
| g Change in eating habits, severe = missed > 2 feeds. | ||||||||
| h Vomiting, severe = little/no intake for more prolonged time. | ||||||||
| i Diarrhea, severe = ≥ 6 liquid stools, no solid consistency. | ||||||||
| j Rash was assessed only as present or not present, without a grading for severity | ||||||||
| k Axillary temperature. | ||||||||
|
Dose 1 |
Dose 2 |
Dose 3 |
Dose 4 |
|||||
|
MENVEO with
|
Routine Vaccines b
|
MENVEOwith
|
Routine Vaccines b
|
MENVEOwith
|
Routine Vaccines b
|
MENVEOwith
|
Routine Vaccines b
|
|
|
Local Adverse Reactions c |
N= |
N= |
N= |
N= |
N= |
N= |
N= |
N= |
|
Tenderness, any |
41 |
45 |
31 |
36 |
24 |
32 |
29 |
39 |
|
Erythema, any |
11 |
14 |
12 |
21 |
14 |
23 |
15 |
25 |
|
Induration, any |
8 |
16 |
9 |
17 |
8 |
19 |
8 |
21 |
|
Systemic Adverse Reactions |
N= |
N= |
N= |
N= |
N= |
N= |
N= |
N= |
|
Irritability, any |
57 |
59 |
48 |
46 |
42 |
38 |
43 |
42 |
|
Sleepiness, any |
50 |
50 |
37 |
36 |
30 |
30 |
29 |
27 |
|
Persistent crying, any |
41 |
38 |
28 |
24 |
22 |
17 |
21 |
18 |
|
Change in eating habits, any |
23 |
24 |
18 |
17 |
17 |
13 |
19 |
16 |
|
Vomiting, any |
11 |
9 |
7 |
6 |
6 |
4 |
5 |
4 |
|
Diarrhea, any |
16 |
11 |
11 |
8 |
8 |
6 |
13 |
9 |
|
Rash j |
3 |
3 |
3 |
4 |
3 |
3 |
4 |
3 |
|
Fever ≥38.°C k |
3 |
2 |
4 |
6 |
7 |
6 |
9 |
7 |
|
Fever 38.0-38.9°C |
3 |
2 |
4 |
5 |
7 |
6 |
6 |
5 |
|
Fever 39.0-39.9°C |
0 |
0 |
1 |
1 |
<1 |
0 |
2 |
2 |
|
Fever ≥40.0°C |
0 |
<1 |
0 |
<1 |
0 |
0 |
<1 |
0 |
The safety of a second dose of MENVEO administered at 12 months of age concomitantly with MMRV was investigated in a randomized, controlled, multicenter study 5 conducted in the U.S. The rates of solicited adverse reactions reported were comparable between the concomitantly administered group (MENVEO with MMRV) and the group which received MMRV alone, or MENVEO alone. The frequency and severity of solicited local and systemic reactions occurring within 7 days following vaccination at 12 months of age, are shown in Table 2. In subjects who received both MENVEO and MMRV at 12 months of age local reactions at both injection sites were evaluated separately. Body temperature measurements were collected for 28 days following the 12 months of age visit, when MMRV was administered to the vaccinees. Common solicited adverse reactions (≥ 10%) among children initiating vaccination at 7 months through 23 months of age and receiving the two-dose series were tenderness (10-16%) and erythema at injection site (12-15%), irritability (27-40%), sleepiness (17-29%), persistent crying (12-21%), change in eating habits (12-20%) and diarrhea (10-16%). An examination of the fever profile during this period showed that MENVEO administered with MMRV did not increase the frequency or intensity of fever above that observed for the MMRV-only group.
| Clinicaltrials.gov Identifier NCT00626327 5 | ||||||||||||||
| N= number of subjects who completed the diary card for a given symptom at the specified vaccination. | ||||||||||||||
| a As Treated Safety Sub-population = US children who received at least one dose of study vaccine and whose diary cards were completed per protocol and returned to the site.. | ||||||||||||||
| b Tenderness, severe = cried when injected limb moved. | ||||||||||||||
| c Irritability, severe = unable to console. | ||||||||||||||
| d Sleepiness, severe = sleeps most of the time, hard to arouse. | ||||||||||||||
| e Change in eating habits, severe = missed > 2 feeds. | ||||||||||||||
| f Vomiting, severe = little/no intake for more prolonged time. | ||||||||||||||
| g Diarrhea, severe = ≥ 6 liquid stools, no solid consistency. | ||||||||||||||
| h Rash was assessed only as present or not present, without a grading for severity | ||||||||||||||
| i Axillary temperature. | ||||||||||||||
|
MENVEO Group |
MENVEO + MMRV Group |
MMRV Group |
||||||||||||
|
MENVEO
|
MENVEO
|
MENVEO
|
MENVEO with MMRV
|
MMRV
|
||||||||||
|
Local Adverse Reactions– MENVEO site |
N=460-462 |
N=381-384 |
N=430-434 |
N= 386-387 | ||||||||||
|
Tenderness, any |
11 |
10 |
11 |
16 |
N/A |
|||||||||
|
Erythema, any |
15 |
13 |
13 |
12 |
N/A |
|||||||||
|
Induration, any |
8 |
8 |
7 |
8 |
N/A |
|||||||||
|
Local Adverse Reactions– MMRV site |
N=382-383 |
N=518-520 |
||||||||||||
|
Tenderness, any |
N/A |
N/A |
N/A |
16 |
19 |
|||||||||
|
Erythema, any |
N/A |
N/A |
N/A |
15 |
14 |
|||||||||
|
Induration, any |
N/A |
N/A |
N/A |
13 |
8 |
|||||||||
|
Systemic Adverse Reactions |
N=461-463 |
N=385-386 |
N=430-434 |
N=387-389 |
N=522-524 |
|||||||||
|
Irritability, any |
40 |
27 |
37 |
37 |
44 |
|||||||||
|
Sleepiness, any |
26 |
17 |
29 |
26 |
32 |
|||||||||
|
Persistent crying, any |
21 |
12 |
20 |
19 |
20 |
|||||||||
|
Change in eating habits, any |
17 |
12 |
17 |
20 |
20 |
|||||||||
|
Vomiting, any |
9 |
6 |
9 |
6 |
6 |
|||||||||
|
Diarrhea, any |
16 |
10 |
15 |
15 |
20 |
|||||||||
|
Rash h |
3 |
5 |
6 |
6 |
8 |
|||||||||
|
Fever ≥38.0°C i |
5 |
5 |
6 |
9 |
7 |
|||||||||
|
Fever 38.0-38.9°C |
3 |
3 |
5 |
7 |
7 |
|||||||||
|
Fever 39.0-39.9°C |
2 |
2 |
1 |
1 |
1 |
|||||||||
|
Fever ≥40.0°C |
<1 |
1 |
<1 |
<1 |
0 |
|||||||||
In clinical trials of children 2 years through 10 years of age 6-9, the most frequently occurring adverse reactions (≥ 10%) among all subjects who received MENVEO were injection site pain (31%), erythema (23%), irritability (18%), induration (16%), sleepiness (14%), malaise (12%), and headache (11%). Among subjects 11 years through 55 years of age, the most frequently occurring adverse reactions (≥ 10%) among all subjects who received MENVEO were pain at the injection site (41%), headache (30%), myalgia (18%), malaise (16%) and nausea (10%).
The rates of solicited adverse reactions reported for subjects 2 years through 5 years and 6 years through 10 years of age who received a single dose of MENVEO or Menactra in a randomized controlled, multicenter study 9 conducted in the U.S. and Canada are shown in Table 3. Following a second dose of MENVEO administered to children 2 years through 5 years of age, the most common solicited adverse reactions (≥ 10%) were pain at injection site (28%), erythema (22%), irritability (16%), induration (13%), and sleepiness (12%). The solicited adverse events from a separate randomized, controlled, multicenter study conducted in the U.S. in adolescents and adults 12 are provided in Tables 4 and 5, respectively. In neither study were concomitant vaccines administered with the study vaccines.
| Clinicaltrials.gov Identifier NCT00616421 9 | |||||||||||||||||||||
| a Moderate: Some limitation in normal daily activity, Severe: Unable to perform normal daily activity. | |||||||||||||||||||||
| b Moderate: ≥50-100mm, Severe: ≥ 100mm. | |||||||||||||||||||||
| c Rash was assessed only as present or not present, without a grading for severity. | |||||||||||||||||||||
| d Fever grading: Any: ≥ 38°C, Moderate: 39-39.9°C, Severe: ≥ 40°C. Parents reported the use of antipyretic medication to treat or prevent symptoms in 11% and 13% of subjects 2 through 5 years of age, 9% and 10% of subjects 6 through 10 years of age for MENVEO and Menactra, respectively. | |||||||||||||||||||||
| e Different systemic reactions were solicited in different age groups. | |||||||||||||||||||||
|
Participants 2 through 5 Years of Age |
|||||||||||||||||||||
|
MENVEO
|
Menactra
|
||||||||||||||||||||
|
Any |
Moderate |
Severe |
Any |
Moderate |
Severe |
||||||||||||||||
|
| |||||||||||||||||||||
|
Injection site pain a |
33 |
6 |
1 |
35 |
8 |
0.4 |
|||||||||||||||
|
Erythema b |
27 |
5 |
1 |
25 |
3 |
0.3 |
|||||||||||||||
|
Induration b |
18 |
2 |
0.4 |
18 |
2 |
0.3 |
|||||||||||||||
|
| |||||||||||||||||||||
|
Irritability a |
21 |
6 |
1 |
22 |
7 |
1 |
|||||||||||||||
|
Sleepiness a |
16 |
3 |
1 |
18 |
5 |
1 |
|||||||||||||||
|
Change in eating a |
9 |
2 |
1 |
10 |
2 |
0.3 |
|||||||||||||||
|
Diarrhea a |
7 |
1 |
0.1 |
8 |
1 |
0 |
|||||||||||||||
|
Headache a |
5 |
1 |
0 |
6 |
1 |
0.3 |
|||||||||||||||
|
Rash c |
4 |
- |
- |
5 |
- |
- |
|||||||||||||||
|
Arthralgia a |
3 |
1 |
0.1 |
4 |
1 |
0 |
|||||||||||||||
|
Vomiting a |
3 |
1 |
0.1 |
3 |
1 |
0 |
|||||||||||||||
|
Fever d |
2 |
0.4 |
0 |
2 |
0.3 |
0 |
|||||||||||||||
|
|
|||||||||||||||||||||
|
MENVEO
|
Menactra
|
||||||||||||||||||||
|
Any |
Moderate |
Severe |
Any |
Moderate |
Severe |
||||||||||||||||
|
| |||||||||||||||||||||
|
Injection site paina |
39 |
8 |
1 |
45 |
10 |
2 |
|||||||||||||||
|
Erythemab |
28 |
5 |
1 |
22 |
2 |
0.2 |
|||||||||||||||
|
Indurationb |
17 |
2 |
0.3 |
13 |
2 |
0 |
|||||||||||||||
|
| |||||||||||||||||||||
|
Headachea |
18 |
3 |
1 |
13 |
2 |
1 |
|||||||||||||||
|
Malaisea |
14 |
3 |
1 |
11 |
3 |
1 |
|||||||||||||||
|
Myalgiaa |
10 |
2 |
1 |
10 |
2 |
1 |
|||||||||||||||
|
Nauseaa |
8 |
2 |
1 |
6 |
2 |
0.4 |
|||||||||||||||
|
Arthralgiaa |
6 |
1 |
0 |
4 |
1 |
0.4 |
|||||||||||||||
|
Chillsa |
5 |
1 |
0 |
5 |
1 |
0.4 |
|||||||||||||||
|
Rashc |
5 |
- |
- |
3 |
- |
- |
|||||||||||||||
|
Feverd |
2 |
1 |
0 |
2 |
0 |
0.4 |
|||||||||||||||
| Clinicaltrials.gov Identifier NCT00450437 12 | ||||||||||||||||||||||||||||
| aModerate: Some limitation in normal daily activity, Severe: Unable to perform normal daily activity. | ||||||||||||||||||||||||||||
| b Moderate: ≥50-100mm, Severe: ≥ 100mm. c Rash was assessed only as present or not present, without a grading for severity. |
||||||||||||||||||||||||||||
| d Fever grading: Any: ≥ 38°C, Moderate: 39-39.9°C, Severe: ≥ 40°C. | ||||||||||||||||||||||||||||
|
MENVEO
|
Menactra
|
|||||||||||||||||||||||||||
|
Reaction |
Any |
Moderate |
Severe |
Any |
Moderate |
Severe |
||||||||||||||||||||||
|
Local | ||||||||||||||||||||||||||||
|
Injection site pain a |
44 |
9 |
1 |
53 |
11 |
1 |
||||||||||||||||||||||
|
Erythema b |
15 |
2 |
0.4 |
16 |
1 |
0 |
||||||||||||||||||||||
|
Induration b |
12 |
2 |
0.2 |
11 |
1 |
0 |
||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||
|
Headache a |
29 |
8 |
2 |
28 |
7 |
1 |
||||||||||||||||||||||
|
Myalgia a |
19 |
4 |
1 |
18 |
5 |
0.4 |
||||||||||||||||||||||
|
Nausea a |
12 |
3 |
1 |
9 |
2 |
1 |
||||||||||||||||||||||
|
Malaise a |
11 |
3 |
1 |
12 |
5 |
1 |
||||||||||||||||||||||
|
Chills a |
8 |
2 |
1 |
7 |
1 |
0.2 |
||||||||||||||||||||||
|
Arthralgia a |
8 |
2 |
0.4 |
6 |
1 |
0 |
||||||||||||||||||||||
|
Rash c |
3 |
- |
- |
3 |
- |
- |
||||||||||||||||||||||
|
Fever d |
1 |
0.4 |
0 |
1 |
0 |
0 |
||||||||||||||||||||||
| Clinicaltrials.gov Identifier NCT00450437 12 | |||||||||||||||||||||||||||||||||||
| a Moderate: Some limitation in normal daily activity, Severe: Unable to perform normal daily activity. | |||||||||||||||||||||||||||||||||||
| b Moderate: ≥50-100mm, Severe: ≥100mm. c Rash was assessed only as present or not present, without a grading for severity. |
|||||||||||||||||||||||||||||||||||
| d Fever grading: Any: ≥ 38°C, Moderate: 39-39.9°C, Severe: ≥ 40°C. | |||||||||||||||||||||||||||||||||||
|
MENVEO
|
Menactra
|
||||||||||||||||||||||||||||||||||
|
Reaction |
Any |
Moderate |
Severe |
Any |
Moderate |
Severe |
|||||||||||||||||||||||||||||
|
Local | |||||||||||||||||||||||||||||||||||
|
Injection site pain a |
38 |
7 |
0.3 |
41 |
6 |
0 |
|||||||||||||||||||||||||||||
|
Erythema b |
16 |
2 |
1 |
12 |
1 |
0 |
|||||||||||||||||||||||||||||
|
Induration b |
13 |
1 |
0.4 |
9 |
0.3 |
0 |
|||||||||||||||||||||||||||||
|
Systemic | |||||||||||||||||||||||||||||||||||
|
Headache a |
25 |
7 |
2 |
25 |
7 |
1 |
|||||||||||||||||||||||||||||
|
Myalgia a |
14 |
4 |
0.5 |
15 |
3 |
1 |
|||||||||||||||||||||||||||||
|
Malaise a |
10 |
3 |
1 |
10 |
2 |
1 |
|||||||||||||||||||||||||||||
|
Nausea a |
7 |
2 |
0.4 |
5 |
1 |
0.3 |
|||||||||||||||||||||||||||||
|
Arthralgia a |
6 |
2 |
0.4 |
6 |
1 |
1 |
|||||||||||||||||||||||||||||
|
Chills a |
4 |
1 |
0.1 |
4 |
1 |
0 |
|||||||||||||||||||||||||||||
|
Rash c |
2 |
- |
- |
1 |
- |
- |
|||||||||||||||||||||||||||||
|
Fever d |
1 |
0.3 |
0 |
1 |
0.3 |
0 |
|||||||||||||||||||||||||||||
Solicited Adverse Reactions following Concomitant Vaccine Administration
The safety of 4-dose MENVEO series administered concomitantly with U.S.-licensed routine infant and toddler vaccines was evaluated in one pivotal trial 2. The safety of a 2-dose series of MENVEO initiated at 7 – 9 months of age, with the second dose administered concomitantly with U.S.-licensed MMR and V vaccine at 12 months of age, was evaluated in one pivotal trial 5. Rates of solicited adverse reactions which occurred 7 days following vaccination are shown in Tables 1 and 2, respectively. There was no significant increase in the rates of solicited systemic or local reactions observed in recipients of routine childhood vaccines when concomitantly vaccinated with MENVEO. [See Concomitant Administration with Other Vaccines (7.1)]
The safety of MENVEO administered concomitantly with Tdap and HPV was evaluated in a single center study 14 conducted in Costa Rica. Solicited local and systemic adverse reactions were reported as noted above. In this study, subjects 11 through 18 years of age received MENVEO concomitantly with Tdap and HPV (N=540), or MENVEO followed one month later by Tdap and then one month later by HPV (N=541), or Tdap followed one month later by MENVEO and then one month later by HPV (N=539). Some solicited systemic adverse reactions were more frequently reported in the group that received MENVEO, Tdap and HPV concomitantly, (headache 40%, malaise 25%, myalgia 27%, and arthralgia 17%) compared to the group that first received MENVEO alone (headache 36%, malaise 20%, myalgia 19%, and arthralgia 11%). Among subjects administered MENVEO alone (one month prior to Tdap), 36% reported headache, 20% malaise, and 16% myalgia. Among subjects administered MENVEO one month after Tdap, 27% reported headache, 18% malaise, and 16% myalgia.
Serious Adverse Events in All Safety Studies
Serious adverse events in subjects receiving a four-dose series of MENVEO at 2, 4, 6 and 12 months were evaluated in three randomized multicenter clinical studies 1-3. In the two controlled studies2, 3, the proportions of infants randomized to receive the four-dose MENVEO series concomitantly with routine vaccinations and infants who received routine vaccinations alone that reported serious adverse events during different study periods were, respectively: a) 2.7% and 2.2%, during the infant series; b) 2.5% and 2.5%, between the infant series and the toddler dose; c) 0.3% and 0.3%, in the one month following the toddler dose; and d) 1.6% and 2.2%, during the 6 months follow up period after the last dose. In the third study 1, which was controlled up to the toddler dose, the proportions of infants randomized to dosing regimens that included receiving four doses of MENVEO concomitantly with routine vaccinations at 2, 4, 6, and 12 months and infants who received routine vaccinations alone that reported serious adverse events during different study periods were, respectively: a) 3.5% and 3.6%, during the infant series; and b) 2.8% and 3.3%, between the infant series and the toddler dose; and c) 0.5% and 0.7%, in the one month following the toddler dose. In the same study, 1.9% of infants randomized to receive the four-dose MENVEO series concomitantly with routine vaccinations reported serious adverse events during the 6 month follow up period after the toddler dose. The most common serious adverse events reported in these three studies were wheezing, pneumonia, gastroenteritis and convulsions, and most occurred at highest frequency after the infant series.
In a study of older infants 5 randomized to receive the two-dose MENVEO series concomitantly with MMRV at 12 months of age, the rates of serious adverse events during the study, including the 6 month follow-up period after the last dose, were 3.6% and 3.8%, for the MENVEO with MMRV and MENVEO-only groups, respectively. Infants receiving MMRV alone, who had a shorter period of study participation as they were enrolled at 12 months of age, had a lower rate of serious adverse events (1.5%). Among 1597 study subjects, included in the safety population, the most commonly reported serious adverse events in all study arms combined were dehydration (0.4%) and gastroenteritis (0.3%). Across the submitted studies of individuals 2 through 23 months of age, within 28 days of vaccination, two deaths were reported in the MENVEO treatment groups (one case of sudden death and one case of sepsis), while no deaths were reported in the control group. None of the deaths was assessed as related to vaccination. Among subjects with symptom onset within 42 days of vaccination (days 12, 25, 29), 3/12049 [0.02%, 95% CI: (0.01% , 0.07%)] MENVEO recipients and 0/2877 [0%, 95% CI: (0%, 0.13%)] control recipients were diagnosed with Kawasaki Disease. One case of acute disseminated encephalomyelitis with symptom onset 29 days post-dose 4 was observed in a participant given MENVEO co-administered with routine US childhood vaccines at 12 months of age (including MMR and varicella vaccines).
The information regarding serious adverse events in subjects 2 years through 10 years of age was derived from 3 randomized, controlled clinical trials 7-9. Safety follow-up ranged from 6 months through 12 months and included 2883 subjects administered MENVEO. Serious adverse events reported during the safety follow-up periods occurred in 21/2883 (0.7%) of MENVEO subjects, in 7/1255 (0.6%) of Menactra subjects, and 2/861 (0.2%) of Menomune subjects. In the subjects receiving either one or two doses of MENVEO, there were 6 subjects with pneumonia, 3 subjects with appendicitis and 2 subjects with dehydration; all other events were reported to occur in one subject. Among 1255 subjects administered a single dose of Menactra and 861 subjects administered Menomune, there were no events reported to occur in more than one subject. The serious adverse events occurring within the first 30 days after receipt of each vaccine were as follows: MENVEO (6/2883 [0.2%]) – appendicitis, pneumonia, staphylococcal infection, dehydration, febrile convulsion, and tonic convulsion; Menactra (1/1255 [0.1%]) – inguinal hernia; Menomune (2/861 [0.2%]) – abdominal pain, lobar pneumonia. In a supportive study 6, 298 subjects received one or two doses of MENVEO and 22 (7%) had serious adverse events over a 13 month follow-up period including 13 subjects with varicella and 2 subjects with laryngitis. All other events were reported to occur in one subject. During the 30 days post vaccination in this study, one limb injury and one case of varicella were reported.
The information regarding serious adverse events in subjects 11 years through 55 years of age was derived from 5 randomized, controlled clinical trials 10-14. Serious adverse events reported within 6 months of vaccination occurred in 40/6185 (0.6%) of MENVEO subjects, 13/1757 (0.7%) of Menactra subjects, and 5/209 (2.4%) of Menomune subjects. During the 6 months following immunization, serious adverse events reported by more than one subject were as follows: MENVEO - appendicitis (3 subjects), road traffic accident (3 subjects), and suicide attempt (5 subjects); Menactra - intervertebral disc protrusion (2 subjects); Menomune - none. Serious adverse events that occurred within 30 days of vaccination were reported by 7 of 6185 (0.1%) subjects in the MENVEO group, 4 of 1757 (0.2%) subjects in the Menactra group, and by none of 209 subjects in the Menomune group. The events that occurred during the first 30 days post immunization with MENVEO were: vitello-intestinal duct remnant; Cushing's syndrome; viral hepatitis; pelvic inflammatory disease; intentional multiple drug overdose; simple partial seizure; and suicidal depression. The events that occurred during the first 30 days post immunization with Menactra were: herpes zoster; fall; intervertebral disc protrusion; and angioedema.
6.2 Postmarketing Experience
In addition to reports in clinical trials, worldwide voluntary reports of adverse events received for MENVEO in persons 11 through 55 years of age since market introduction of this vaccine are listed below. This list includes serious events or events which have plausible causal connection to MENVEO. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to the vaccine.
Ear and Labyrinth Disorders: Hearing impaired, ear pain, vertigo, vestibular disorder.
Eye Disorders: Eyelid ptosis.
General Disorders and Administration Site Conditions: Injection site pruritus, pain, erythema, inflammation and swelling, fatigue, malaise, pyrexia.
Immune System Disorders: Hypersensitivity.
Infections and Infestations: Vaccination site cellulitis.
Injury, Poisoning and Procedural Complications: Fall, head injury.
Investigation: Alanine aminotransferase increased, body temperature increased.
Musculoskeletal and Connective Tissue Disorders: Arthralgia, bone pain.
Nervous System Disorders: Dizziness, syncope, tonic convulsion, headache, facial paresis, balance disorder.
Respiratory, Thoracic and Mediastinal Disorders: Oropharyngeal pain.
Skin and Subcutaneous Tissue Disorders: Skin exfoliation.
Postmarketing Observational Safety Study
In a postmarketing observational safety study conducted in a United States health maintenance organization, data from electronic health records of 48899 persons 11 through 21 years of age were used to evaluate pre-specified events of interest following vaccination with Menveo. Using a self-controlled case series method, Bell’s palsy showed a statistically significant increased risk in the period 1 to 84 days post-vaccination compared to the control period, with an overall adjusted relative incidence of 2.9 (95% CI: 1.1-7.5). Among the 8 reported cases of Bell’s palsy, 6 cases occurred in persons who received Menveo concomitantly with one or more of the following vaccines: Tdap, HPV, and Influenza vaccine. All reported Bell’s palsy cases resolved.
7 DRUG INTERACTIONS
7.1 Concomitant Administration with Other Vaccines
Do not mix MENVEO or any of its components with any other vaccine or diluent in the same syringe or vial.
In two clinical trials of infants initiating vaccination at 2 months of age 1, 3, MENVEO was given concomitantly at 2, 4 and 6 months with routine infant vaccines: diphtheria toxoid, acellular pertussis, tetanus toxoid, inactivated polio types 1, 2 and 3, hepatitis B, Haemophilus influenzae type b (Hib) antigens; pentavalent rotavirus, and 7-valent pneumococcal conjugate vaccine. For dose 4 given at 12 months of age, MENVEO was given concomitantly with the following vaccines: 7-valent pneumococcal conjugate, MMRV or MMR+V, and inactivated hepatitis A. In a clinical trial of older infants (≥ 7 months of age) and toddlers 5, MENVEO was administered concomitantly with MMRV or MMR+V vaccine(s) at 12 months of age. No immune interference was observed for the concomitantly administered vaccines, including most pneumococcal vaccine serotypes (post-dose 3); no immune interference was observed post-dose 4 for any pneumococcal vaccine serotypes1, 3.[See Immunogenicity of Concomitantly Administered Vaccines (14.5)]
For children 2 years through 10 years of age, no data are available to evaluate safety and immunogenicity of other childhood vaccines when administered concomitantly with MENVEO.
In a clinical trial in adolescents 14, MENVEO was given concomitantly with the following: Tdap and HPV, no interference was observed in meningococcal immune responses when compared to MENVEO given alone. Lower geometric mean antibody concentrations (GMCs) for antibodies to the pertussis antigens filamentous hemagglutinin (FHA) and pertactin were observed when MENVEO was administered concomitantly with Tdap and HPV as compared with Tdap alone. [See Immunogenicity of Concomitantly Administered Vaccines, (14.5)]
7.2 Immunosuppressive Treatments
Immunosuppressive therapies, such as irradiation, antimetabolite medications, alkylating agents, cytotoxic drugs, and corticosteroids (when used in greater than physiologic doses) may reduce the immune response to MENVEO [See Altered Immunocompetence (5.3)]. The immunogenicity of MENVEO has not been evaluated in persons receiving such therapies.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category B
Reproduction studies have been performed in female rabbits at a dose of approximately 20 times the human dose (on a mg/kg basis) and have revealed no evidence of impaired fertility or harm to the fetus due to MENVEO. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, MENVEO should be given to a pregnant woman only if clearly needed.
Nonclinical Studies
The effect of MENVEO on embryo-fetal and post-natal development was evaluated in pregnant rabbits. Animals were administered MENVEO 3 times prior to gestation, during the period of organogenesis (gestation day 7) and later in pregnancy (gestation day 20), 0.5 mL/rabbit/occasion (approximately 20-fold excess relative to the projected human dose on a body weight basis) by intramuscular injections. There were no adverse effects attributable to the vaccine on mating, female fertility, pregnancy, or embryo-fetal development. There were no vaccine related fetal malformations or other evidence of teratogenesis noted in this study.
Clinical Studies
To date, no clinical trials have been specifically designed to evaluate the use of MENVEO in pregnant or lactating women.
Among the 5065 women in the adolescent and adult populations enrolled in the studies, 43 women were found to be pregnant during the 6-month follow-up period after vaccination. Of these, 37 pregnancies occurred among 3952 MENVEO recipients (7 spontaneous abortions, no congenital anomalies). Six pregnancies occurred among 1113 Menactra recipients (no spontaneous abortions, one congenital anomaly (hydrocephalus)).
Among the seven subjects with adverse pregnancy outcomes who had received MENVEO, the estimated dates of conception were 5 days prior to vaccination (1 subject), 6 to 17 weeks post vaccination (5 subjects), and 6 months post vaccination (1 subject). In the subject who had received Menactra the estimated date of conception was approximately 15 weeks post immunization.
Safety and effectiveness of MENVEO have not been established in pregnant women.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when MENVEO is administered to a nursing woman. No studies have been conducted to assess the impact of MENVEO on milk production, its presence in breast milk or its effects on the breast-fed child.
11 DESCRIPTION
MENVEO [Meningococcal (Groups A, C, Y and W-135) Oligosaccharide Diphtheria CRM197 Conjugate Vaccine] is a sterile liquid vaccine administered by intramuscular injection that contains N. meningitidis serogroup A, C, Y and W-135 oligosaccharides conjugated individually to Corynebacterium diphtheriae CRM197 protein. The polysaccharides are produced by bacterial fermentation of N. meningitidis (serogroups A, C, Y or W-135). N. meningitidis strains A, C, Y and W-135 are each cultured and grown on Franz Complete medium and treated with formaldehyde. MenA, MenW-135 and MenY polysaccharides are purified by several extraction and precipitation steps. MenC polysaccharide is purified by a combination of chromatography and precipitation steps.
The protein carrier (CRM197) is produced by bacterial fermentation and is purified by a series of chromatography and ultrafiltration steps. C. diphtheriae is cultured and grown on CY medium containing yeast extracts and amino acids.
The oligosaccharides are prepared for conjugation from purified polysaccharides by hydrolysis, sizing, and reductive amination. After activation, each oligosaccharide is covalently linked to the CRM197 protein. The resulting glycoconjugates are purified to yield the four drug substances, which compose the final vaccine. The vaccine contains no preservative or adjuvant. Each dose of vaccine contains 10 μg MenA oligosaccharide, 5 μg of each of MenC, MenY and MenW-135 oligosaccharides and 32.7 to 64.1 μg CRM197 protein. Residual formaldehyde per dose is estimated to be not more than 0.30 μg.
The vials in which the vaccine components are contained are composed of Type I glass, USP. The container closures (synthetic rubber stoppers) do not contain latex.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Neisseria meningitidis is a gram-negative diplococcus that causes life-threatening invasive disease such as meningitis and sepsis. Globally, 5 serogroups, A, B, C, Y and W-135 cause almost all invasive meningococcal infections. The presence of serum bactericidal antibodies protects against invasive meningococcal disease 16. Vaccination with MENVEO leads to the production of bactericidal antibodies directed against the capsular polysaccharides of serogroups A, C, Y and W-135.
14 CLINICAL STUDIES
For all age groups, effectiveness has been inferred from the measurement of serogroup-specific anticapsular antibodies with bactericidal activity using pooled human serum that lacked bactericidal activity as the source of exogenous complement (hSBA). In the absence of a licensed comparator vaccine for use in infants, the pre-specified endpoint for effectiveness of MENVEO in U.S. infants receiving a four- dose series at 2, 4, 6 and 12 months of age was the proportion of subjects achieving an hSBA ≥ 1:8, with the lower limit of the 2-sided 95% CI for the point estimate being ≥ 80% of vaccinees for serogroup A, and ≥ 85% of vaccinees for serogroups C, W-135 and Y one month following the final dose.
The effectiveness of MENVEO in subjects 2 years through 55 years of age was assessed by comparing the hSBA responses to immunization with MENVEO to those following immunization with the licensed meningococcal quadrivalent conjugate vaccine Menactra.
The primary effectiveness endpoint was hSBA seroresponse to each serogroup 28 days after vaccination. Seroresponse was defined as: a) post vaccination hSBA ≥1:8 for subjects with a baseline hSBA <1:4; or, b) at least 4-fold higher than baseline titers for subjects with a pre-vaccination hSBA ≥1:4. Secondary endpoints included the proportion of subjects with post vaccination hSBA ≥1:8 and the hSBA Geometric Mean Titer (GMT) for each serogroup. In a separate group of children 2 years through 5 years of age randomized to receive two doses of MENVEO administered two months apart, seroresponse rate, proportion with post-vaccination hSBA ≥1:8 and GMT were determined for each serogroup.
14.1 Immunogenicity in Infants
The effectiveness of MENVEO in infants was assessed in a randomized, controlled, multicenter study 3. Among the MENVEO subjects who were included in the per protocol analysis, the mean age at enrollment was 65 days; 51% were male; 67% were Caucasian, 6% were African American, 15% were Hispanic, 2% were Asian, and 9% were noted as other racial/ethnic groups. The pre-defined criteria for immunogenicity were met for all four serogroups A, C, W-135 and Y at one month following completion of a four-dose series at 2, 4, 6 and 12 months of age (Table 6).
The percentage of subjects with hSBA ≥1:8 at 7 months was 94% to 98% for serogroups C, W-135, and Y and 76% for serogroup A.
| Serogroup | Post 3rd dose | Post
4th dose |
|
|---|---|---|---|
| Clinicaltrials.gov Identifier NCT01000311 3 | |||
| %≥1:8 = proportions of subjects with hSBA ≥ 1:8 against a given serogroup; CI = confidence interval; GMT = geometric mean antibody titer; N = number of infants eligible for inclusion in the Per-Protocol Immunogenicity population for whom serological results were available for the post-dose 3 and post-dose 4 evaluations. | |||
| Serum Bactericidal Assay with exogenous human complement source (hSBA). | |||
| a Prespecified criteria for adequacy of immune response were met (LL of the 95% CI > 80% for serogroup A and > 85% for serogroups C, W, and Y). | |||
|
A |
N=202 |
N=168 |
|
|
% ≥1:8 |
76 |
89 |
|
|
GMT |
21 |
54 |
|
|
C |
N=199 |
N=156 |
|
|
% ≥1:8 |
94 |
95 |
|
|
GMT |
74 |
135 |
|
|
W-135 |
N=194 |
N=153 |
|
|
% ≥1:8 |
98 |
97 |
|
|
GMT |
79 |
215 |
|
|
Y |
N=188 |
N=153 |
|
|
% ≥1:8 |
94 |
96 |
|
|
GMT |
51 |
185 |
|
The effectiveness of two doses of MENVEO given at 7-9 months and 12 months of age was assessed in a randomized, multicenter, controlled clinical trial 5 conducted in the U.S. This study also investigated the concomitant administration of MENVEO and MMRV. The per protocol population for assessing the response to two doses of MENVEO consisted of 386 subjects. Among subjects who completed the per protocol analysis, their mean age at enrollment was 8.5 months (SD 0.8 months); they were 50% male; 61% were Caucasian, 15% were Hispanic, 10% were African American, 4% were Asian, and 10% were noted as other racial/ethnic groups.
Among the per protocol population, after MENVEO administered at 7-9 and at 12 months, the proportions of subjects with hSBA ≥ 1:8 for serogroups A, C, W-135, and Y were respectively: 88% (84-91), 100% (98-100), 98% (96-100), 96% (93-99).
14.2 Immunogenicity in Children
Effectiveness in subjects 2 years through 10 years of age was evaluated in a randomized, multicenter, active controlled clinical study 9 comparing hSBA responses following one dose of MENVEO or Menactra. The study was conducted in the U.S. and Canada and was stratified by age (2 years through 5 years and 6 years through 10 years). The per protocol population evaluated after a single dose of vaccine consisted of 1170 subjects who received MENVEO and 1161 who received Menactra (see Table 7) and included serological results for 89 % to 95% of subjects, depending on serogroup and age group. Demographics for the 616 and 619 subjects 2 through 5 years of age for MENVEO and Menactra were as follows: mean age 3.6 years (SD 1.1) vs. 3.6 years (SD 1.1); 51% vs. 52% male; 62% vs. 62% Caucasian, 14% vs. 13% Hispanic, 12% vs. 13% African American, 6% vs. 4% Asian, and 7% vs. 8% other racial/ethnic groups. Demographics were for 554 and 542 per protocol subjects 6 through 10 years of age for MENVEO and Menactra were as follows: mean age 7.9 years (SD 1.4) vs. 8.1 years (SD 1.4); 52% vs. 56% male; 66% vs. 66% Caucasian, 14% vs. 14% African American, 7% vs. 7% Hispanic, 5% vs. 6% Asian, and 8% vs. 8% other racial/ethnic groups. In a separate group of children 2 years through 5 years of age randomized to receive two doses of MENVEO administered two months apart, the per protocol population evaluated after two doses of MENVEO consisted of 297 subjects and included serologic results for 96-99% of subjects, depending on serogroup.
In study participants 2 years through 5 years and 6 years through 10 years of age, non-inferiority of MENVEO to Menactra for the proportion of subjects with a seroresponse was demonstrated for serogroups C, W-135 and Y, but not for serogroup A (Table 7).
| Clinicaltrials.gov Identifier NCT00616421 9 | ||||||
| a Serum Bactericidal Assay with exogenous human complement source (hSBA). | ||||||
| b Seroresponse was defined as: subjects with a pre-vaccination hSBA <1:4, a post vaccination titer of ≥ 1:8 and among subjects with a pre-vaccination hSBA ≥ 1:4, a post vaccination titer at least 4-fold higher than baseline. | ||||||
| c Non-inferiority criterion not met (the lower limit of the two-sided 95% CI ≤ -10% for vaccine group differences). | ||||||
| d Non-inferiority criterion met (the lower limit of the two-sided 95% CI >-10 % for vaccine group differences [MENVEO minus Menactra]). | ||||||
|
2-5 Years |
6-10 Years |
|||||
|
Endpoint by serogroup |
MENVEO
|
Menactra
|
Percent Difference (MENVEO – Menactra) or GMT ratio (MENVEO/ Menactra)
|
MENVEO
|
Menactra
|
Percent Difference (MENVEO – Menactra) or GMT ratio (MENVEO/ Menactra)
|
|
A |
N=606 |
N=611 |
N=551 |
N=541 | ||
|
% |
72 |
77 |
-5 |
77 |
83 |
-6 |
|
% ≥1:8 |
72 |
78 |
-6 |
77 |
83 |
-6 |
|
GMT |
26 |
25 |
1.04 |
35 |
35 |
1.01 |
|
C |
N=607 |
N=615 |
N=554 |
N=539 | ||
|
% |
60 |
56 |
4 |
63 |
57 |
6 |
|
% ≥1:8 |
68 |
64 |
4 |
77 |
74 |
3 |
|
GMT |
18 |
13 |
1.33 |
36 |
27 |
1.36 |
|
W-135 |
N=594 |
N=605 |
N=542 |
N=533 | ||
|
% Seroresponse b |
72 |
58 |
14 |
57 |
44 |
13 |
|
% ≥1:8 |
90 |
75 |
15 |
91 |
84 |
7 |
|
GMT |
43 |
21 |
2.02 |
61 |
35 |
1.72 |
|
Y |
N=593 |
N=600 |
N=545 |
N=539 | ||
|
% |
66 |
45 |
21 |
58 |
39 |
19 |
|
% ≥1:8 |
76 |
57 |
19 |
79 |
63 |
16 |
|
GMT |
24 |
10 |
2.36 |
34 |
14 |
2.41 |
In the 297 per protocol subjects 2 years through 5 years of age observed at 1 month after the second dose of MENVEO, the proportions of subjects with seroresponse (95% CI) were: 91% (87-94), 98% (95-99), 89% (85-92), and 95% (91-97) for serogroups A, C, W-135 and Y, respectively. The proportion of subjects with hSBA ≥1:8 (95% CI) were 91% (88-94), 99% (97-100), 99% (98-100), and 98% (95-99) for serogroups A, C, W-135 and Y, respectively. The hSBA GMTs (95% CI) for this group were 64 (51-81), 144 (118-177), 132 (111-157), and 102 (82-126) for serogroups A, C, W-135 and Y, respectively.
14.3 Immunogenicity in Adolescents
Effectiveness in subjects 11 through 55 years of age was evaluated in a randomized, multicenter, active controlled clinical study 12 comparing the hSBA responses following one dose of MENVEO or Menactra. The study was conducted in the U.S. and stratified by age (11 through 18 years of age and 19 through 55 years of age). This study enrolled 3539 participants, who were randomized to receive a dose of MENVEO (N=2663) or Menactra (N=876). Among subjects who completed the per-protocol evaluation for immunogenicity (N=3393, MENVEO= 2549, Menactra=844), demographics for MENVEO and Menactra subjects respectively were as follows: mean age 23.9 (SD 13.6) vs. 23.7 (SD 13.7), 42% vs. 42% male, 79% vs. 78% Caucasian, 8% vs. 9% African American, 7% vs. 7% Hispanic, 3% vs. 3% Asian, 2% vs. 3% other racial/ethnic groups. Immunogenicity for each serogroup was assessed in a subset of study participants (see Tables 8 and 10).
In study participants 11 through 18 years of age, non-inferiority of MENVEO to Menactra was demonstrated for all four serogroups for the proportion of subjects with a seroresponse (Table 8).
| Clinicaltrials.gov Identifier NCT00450437 12 | ||||
| a Serum Bactericidal Assay with exogenous human complement source (hSBA). | ||||
| b Seroresponse was defined as: a) post vaccination hSBA ≥1:8 for subjects with a pre-vaccination hSBA <1:4; or, b) at least 4-fold higher than baseline titers for subjects with a pre-vaccination hSBA ≥1:4. | ||||
| c Non-inferiority criterion for the primary endpoint met (the lower limit of the two-sided 95% CI >-10 % for vaccine group differences [MENVEO minus Menactra]). | ||||
|
Bactericidal Antibody Response† |
Comparison of MENVEO and
|
|||
|
Endpoint by Serogroup |
MENVEO
|
Menactra
|
MENVEO / Menactra
|
MENVEO minus Menactra
|
|
A |
N=1075 |
N=359 | ||
|
% Seroresponse b |
75 |
66 |
8 |
|
|
% ≥ 1:8 |
75 |
67 |
- |
8 |
|
GMT |
29 |
18 |
1.63 |
- |
|
C |
N=1396 |
N=460 | ||
|
% Seroresponse b |
76 |
73 |
2 |
|
|
% ≥ 1:8 |
85 |
85 |
- |
0 |
|
GMT |
50 |
41 |
1.22 |
- |
|
W-135 |
N=1024 |
N=288 | ||
|
% Seroresponse b |
75 |
63 |
12 |
|
|
% ≥ 1:8 |
96 |
88 |
- |
8 |
|
GMT |
87 |
44 |
2.00 |
- |
|
Y |
N=1036 |
N=294 | ||
|
% Seroresponse b |
68 |
41 |
27 |
|
|
% ≥ 1:8 |
88 |
69 |
- |
19 |
|
GMT |
51 |
18 |
2.82 |
- |
A subset of adolescents, who had received either MENVEO (n=275) or Menactra (n=179), was enrolled in a follow-up observational study along with separately enrolled age-matched naive subjects (n=97) to assess the persistence of antibody responses through 21 months following vaccination. Data are presented in Table 9.
| Clinicaltrials.gov Identifier NCT00856297 15 | |||||||||||
|
% hSBA ≥1:8
|
hSBA GMTs
|
||||||||||
|
Serogroup |
MENVEO |
Menactra |
Naive |
MENVEO |
Menactra |
Naive |
|||||
|
A |
N=275 |
N=179 |
N=97 |
N=275 |
N=179 |
N=97 |
|||||
|
36 |
23 |
5 |
5.29 |
3.5 |
2.36 |
||||||
|
C |
N=275 |
N=179 |
N=97 |
N=275 |
N=179 |
N=97 |
|||||
|
62 |
59 |
42 |
10 |
8.96 |
5.95 |
||||||
|
W-135 |
N=273 |
N=176 |
N=97 |
N=273 |
N=176 |
N=97 |
|||||
|
84 |
74 |
51 |
18 |
14 |
7.80 |
||||||
|
Y |
N=275 |
N=179 |
N=97 |
N=275 |
N=179 |
N=97 |
|||||
|
67 |
54 |
40 |
12 |
7.85 |
5.14 |
||||||
14.4 Immunogenicity in Adults
The study in subjects 11 through 55 years of age was a randomized, multicenter, active controlled clinical trial 12 conducted in the U.S. and stratified by age (11 through 18 years of age and 19 through 55 years of age) as described above [See Immunogenicity in Adolescents (14.3)]
In study participants 19 through 55 years of age, non-inferiority of MENVEO to Menactra was demonstrated for all four serogroups for the proportion of subjects with a seroresponse (Table 10).
| Clinicaltrials.gov Identifier NCT00450437 12 | ||||
| a Serum Bactericidal Assay with exogenous human complement source (hSBA). | ||||
| b Seroresponse was defined as: a) post vaccination hSBA ≥1:8 for subjects with a pre-vaccination hSBA <1:4; or, b) at least 4-fold higher than baseline titers for subjects with a pre-vaccination hSBA ≥1:4. | ||||
| c Non-inferiority criterion for the primary endpoint met (the lower limit of the two-sided 95% CI >-10 % for vaccine group differences [MENVEO minus Menactra]). | ||||
|
Bactericidal Antibody Response a |
Comparison of MENVEO and
|
|||
|
Endpoint by Serogroup |
MENVEO
|
Menactra
|
MENVEO / Menactra
|
MENVEO minus Menactra
|
|
A |
N=963 |
N=321 | ||
|
% Seroresponse b |
67 |
68 |
-1 |
|
|
% ≥ 1:8 |
69 |
71 |
- |
-2 |
|
GMT |
31 |
30 |
1.06 |
- |
|
C |
N=902 |
N=300 | ||
|
% Seroresponse b |
68 |
60 |
8 |
|
|
% ≥ 1:8 |
80 |
74 |
- |
6 |
|
GMT |
50 |
34 |
1.50 |
- |
|
W-135 |
N=484 |
N=292 | ||
|
% Seroresponse b |
50 |
41 |
9 |
|
|
% ≥ 1:8 |
94 |
90 |
- |
4 |
|
GMT |
111 |
69 |
1.61 |
- |
|
Y |
N=503 |
N=306 | ||
|
% Seroresponse b |
56 |
40 |
16 |
|
|
% ≥ 1:8 |
79 |
70 |
- |
9 |
|
GMT |
44 |
21 |
2.10 |
- |
14.5 Immunogenicity of Concomitantly Administered Vaccines
In US infants 1, 3 who received MENVEO concomitantly with DTaP-IPV-Hib and PCV7 at 2, 4, and 6 months of age and HBV administered according to ACIP recommendations, there was no evidence for reduced antibody response to pertussis antigens (GMC to pertussis toxin, filamentous hemagglutinin, fimbriae, and pertactin), diphtheria toxoid (antibody levels ≥0.1 IU/mL), tetanus toxoid (antibody levels ≥0.1 IU/mL), poliovirus types 1, 2, and 3 (neutralizing antibody levels ≥1:8 to each virus), Haemophilus influenzae type b (anti-PRP antibody ≥0.15μg/mL), hepatitis B (anti-hepatitis B surface antigen ≥10 mIU/mL), or most serotypes of PCV7 (antibody levels ≥0.35 mcg/mL) relative to the response in infants administered DTaP-IPV-Hib, PCV7 and HBV. The immune responses to DTaP-IPV-Hib, PCV7 and HBV were evaluated one month following dose 3 1, 3. No interference was observed for pertussis based on GMC ratios, or for the other concomitantly administered vaccines, with the exception of pneumococcal serotype 6B1,.3 and 23F3, for which interference was suggested post-dose 3. No interference was observed post-dose 4 for these serotypes1, 3.
There was no evidence for interference in the immune response to MMR and varicella vaccines (among initially seronegative children) in terms of percentages of children with anti-measles antibodies ≥255 mIU/mL, anti-mumps ≥10 ELISA antibody units, anti-rubella ≥10 IU/mL, and anti-varicella ≥5 gp ELISA units/mL, administered at 12 months of age 5 concomitantly with MENVEO relative to these vaccines administered alone. The immune responses to MMR and varicella vaccines were evaluated 6 weeks post-vaccination.
For children 2 years through 10 years of age, no data are available for evaluating safety and immunogenicity of other childhood vaccines when administered concomitantly with MENVEO.
For individuals 11 through 18 years of age, the effect of concomitant administration of MENVEO with Tdap and HPV was evaluated in a study 14 conducted in Costa Rica (see also section 6.1 for the safety results from this trial). Subjects were randomized to receive one of the following regimens at the start of the trial: MENVEO plus Tdap plus HPV (N=540); MENVEO alone (N=541); Tdap alone (N=539). Subjects were healthy adolescents 11 through 18 years of age (mean age between groups was 13.8 to 13.9 years). For MENVEO antigens, the proportion (95% CI) of subjects achieving an hSBA seroresponse among those who received MENVEO plus Tdap plus HPV vs. MENVEO alone, respectively, were: serogroup A 80% (76, 84) vs. 82% (78, 85); serogroup C 83% (80, 87) vs. 84% (80, 87); serogroup W-135 77% (73, 80) vs. 81% (77, 84); serogroup Y 83% (79, 86) vs. 82% (79, 86). Among subjects who received Tdap plus MENVEO plus HPV, compared with Tdap alone, the proportions (95% CI) of subjects who achieved an anti-tetanus or anti-diphtheria toxoids levels ≥1.0 IU/mL in the two groups respectively were 100% (99, 100) vs. 98% (96, 99) and 100% (99, 100) vs. 100% (99, 100). For pertussis antigens, among subjects who received Tdap plus MENVEO plus HPV, compared with Tdap alone, the responses respectively for anti-pertussis toxin GMCs (95% CI) were 51 (47, 55) vs. 63 (58, 69) ELISA Units (EU)/mL, for anti-filamentous hemagglutinin were 342 (310, 376) vs. 511 (464, 563) EU/mL, and for anti-pertactin were 819 (727, 923) vs. 1197 (1061, 1350) EU/mL. Because there are no established serological correlates of protection for pertussis, the clinical implications of the lower pertussis antigen responses are unknown.
15 REFERENCES
All NCT numbers are as noted in the National Library of Medicine clinical trial database (see clinicaltrial.gov)
- 1.
- NCT00474526 (V59P14)
- 2.
- NCT00806195 (V59P23)
- 3.
- NCT01000311 (V59_33)
- 4.
- NCT00310856 (V59P9)
- 5.
- NCT00626327 (V59P21)
- 6.
- NCT00310817 (V59P7)
- 7.
- NCT00262028 (V59P8)
- 8.
- NCT00329849 (V59P10)
- 9.
- NCT00616421 (V59P20)
- 10.
- NCT01018732 (V59P6)
- 11.
- NCT00329901 (V59P11)
- 12.
- NCT00450437 (V59P13)
- 13.
- NCT00474487 (V59P17)
- 14.
- NCT00518180 (V59P18)
- 15.
- NCT00856297 (V59P13E1)
- 16.
- Goldschneider I, Gotschlich EC, Artenstein MS. Human immunity to the meningococcus. I. The role of humoral antibodies. J Exp Med (1969);129:1307-1326.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
MENVEO product presentation is listed in Table 11 below. The container closures (synthetic rubber stoppers) do not contain latex.
|
Presentation |
Carton NDC Number |
Components |
|
Five doses (10 vials) per package |
46028-208-01 |
|
|
16.2 Storage and Handling
Do not freeze. Frozen/previously frozen product should not be used.
Store refrigerated, away from the freezer compartment, at 36°F to 46°F (2°C to 8°C).
Protect from light. Vaccine must be maintained at 36°F to 46°F during transport.
Do not use after the expiration date. The reconstituted vaccine should be used immediately, but may be held at or below 77°F (25°C) for up to 8 hours.
17 PATIENT COUNSELING INFORMATION
Vaccine Information Statements are required by the National Childhood Vaccine Injury Act of 1986 to be given prior to immunization to the patient, parent, or guardian. These materials are available free of charge at the Centers for Disease Control and Prevention (CDC) website (www.cdc.gov/vaccines.).
Inform patients, parents or guardians about:
- •
- Potential benefits and risks of immunization with MENVEO.
- •
- The importance of completing the immunization series.
- •
- Potential for adverse reactions that have been temporally associated with administration of MENVEO or other vaccines containing similar components.
- •
- Reporting any adverse reactions to their healthcare provider.
- •
- The Novartis Vaccines and Diagnostics, Inc. pregnancy registry, as appropriate.
Manufactured by:
Novartis Vaccines and Diagnostics S.r.l.,
Bellaria-Rosia 53018, Sovicille (SI), Italy.
An affiliate of:
Novartis Vaccines and Diagnostics, Inc.
350 Massachusetts Avenue,
Cambridge, MA 02139-4182, USA
1-877-683-4732
MENVEO is a registered trademark of Novartis AG.
PRINICIPAL DISPLAY PANEL
NDC 46028-208-01
MENINGOCOCCAL (GROUPS A, C, Y AND W-135)
OLIGOSACCHARIDE
DIPHTHERIA CRM197 CONJUGATE VACCINE
MENVEO®
Powder and solvent for injection
Rx Only
NOVARTIS VACCINES
Manufactured by
Novartis Vaccines and Diagnostics S.r.l. Bellaria-Rosia 53018 Sovicille (SI), Italy
Distributed by
Novartis Vaccines and Diagnostics Inc., 350 Massachusetts Avenue, Cambridge, MA 02139
US License No. 1751
MCK008(0113A)
| MENVEO
meningococcal (groups a, c, y and w-135) oligosaccharide diphtheria crm197 conjugate vaccine kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Novartis Vaccines and Diagnostics s.r.l. (442017901) |