Label: COUGH DROP- menthol lozenge
NORTH COUGH DROP- menthol lozenge
-
NDC Code(s):
0498-0119-00,
0498-0119-05,
0498-0119-07,
0498-1119-00, view more0498-1119-05, 0498-1119-10, 0498-1120-00, 0498-1120-05, 0498-1120-10
- Packager: Honeywell Safety Products USA, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
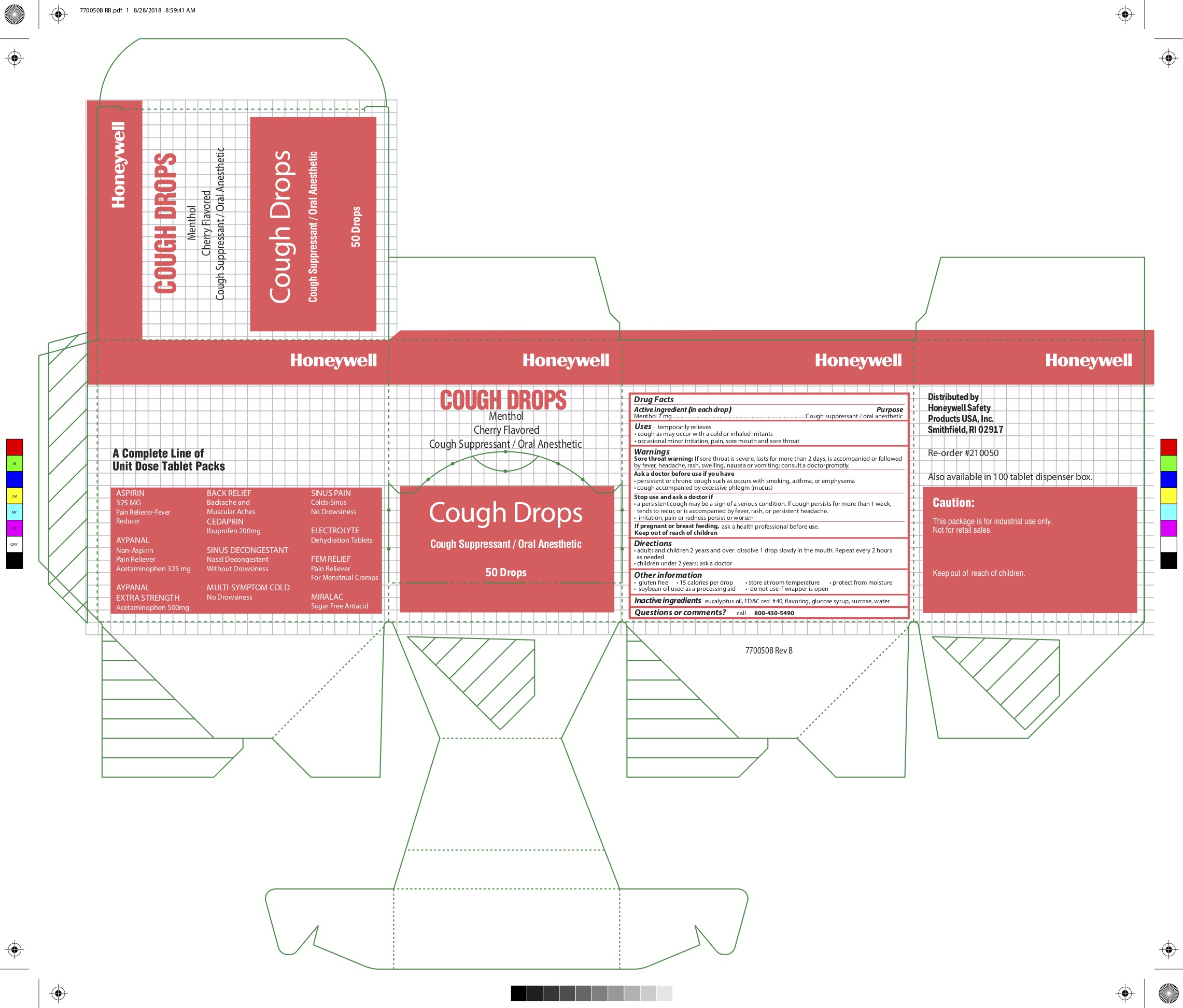
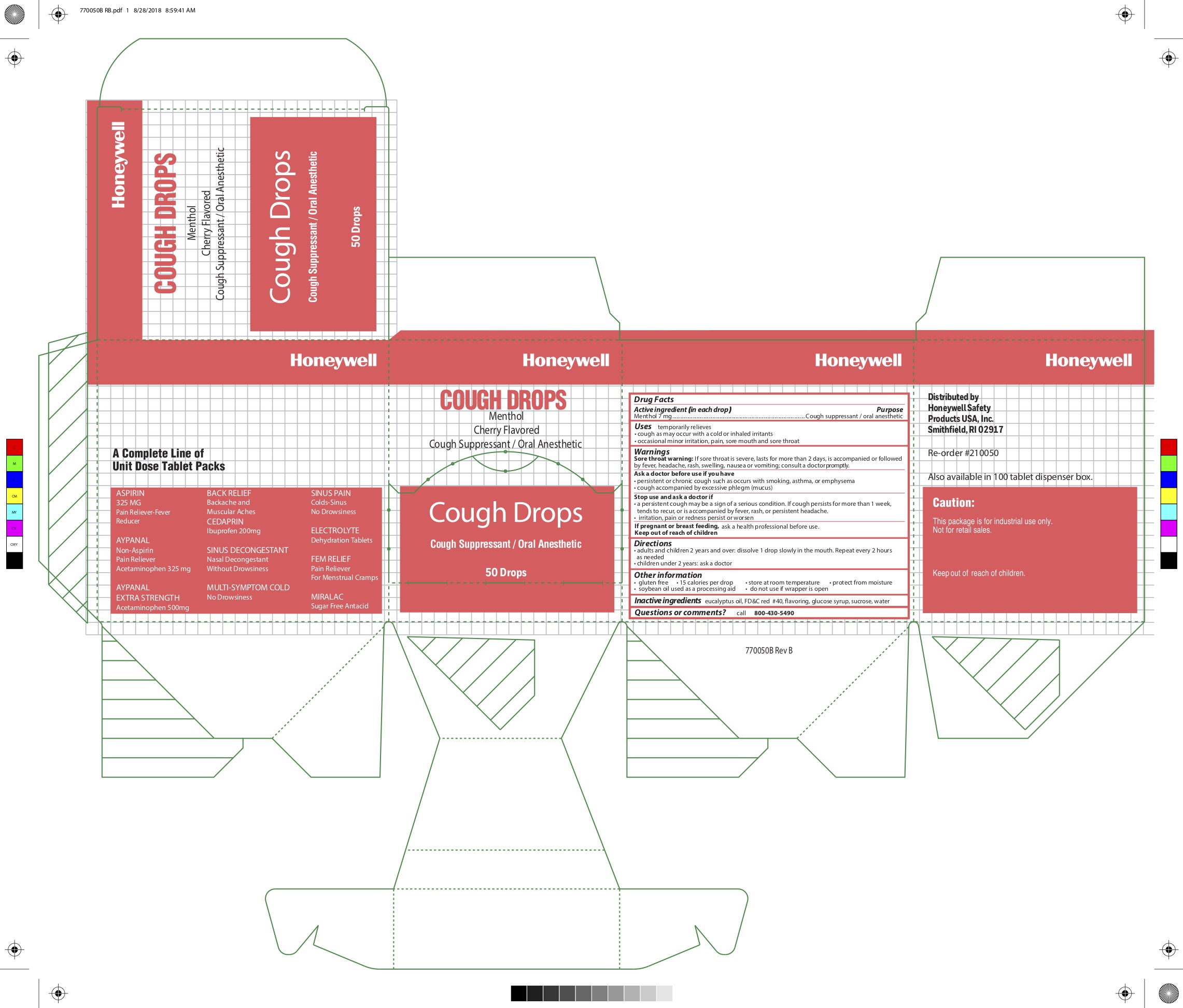
- Active ingredient (in each drop)
- Purpose
- Uses
-
Warnings
Sore throat warning: if sore throat is severe, lasts for more than 2 days, is accompanied or followed by a fever, headache, rash, swelling, nausea or vomiting, consult a doctor promptly
Ask a doctor before use if you have:
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
- cough accompanied by excessive phlegm (mucus)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
- Cough drops original
- Honeywell Cough Drop Label
-
INGREDIENTS AND APPEARANCE
COUGH DROP
menthol lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-1120 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 7 mg Inactive Ingredients Ingredient Name Strength CORN SYRUP (UNII: 9G5L16BK6N) EUCALYPTUS OIL (UNII: 2R04ONI662) FD&C RED NO. 40 (UNII: WZB9127XOA) SUCROSE (UNII: C151H8M554) WATER (UNII: 059QF0KO0R) Product Characteristics Color red (RED) Score no score Shape OVAL Size 22mm Flavor CHERRY (CHERRY) Imprint Code B Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-1120-00 1 in 1 POUCH; Type 0: Not a Combination Product 09/18/2018 2 NDC:0498-1120-05 50 in 1 BOX; Type 0: Not a Combination Product 09/18/2018 3 NDC:0498-1120-10 100 in 1 BOX; Type 0: Not a Combination Product 09/18/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 NORTH COUGH DROP
menthol lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-0119 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 7 mg Inactive Ingredients Ingredient Name Strength CORN SYRUP (UNII: 9G5L16BK6N) EUCALYPTUS OIL (UNII: 2R04ONI662) FD&C RED NO. 40 (UNII: WZB9127XOA) SUCROSE (UNII: C151H8M554) WATER (UNII: 059QF0KO0R) Product Characteristics Color red (RED) Score no score Shape OVAL Size 22mm Flavor CHERRY (CHERRY) Imprint Code B Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0119-00 1 in 1 POUCH; Type 0: Not a Combination Product 12/18/2017 12/31/2017 2 NDC:0498-0119-05 50 in 1 BOX, UNIT-DOSE; Type 0: Not a Combination Product 12/18/2017 12/31/2017 3 NDC:0498-0119-07 100 in 1 BOX, UNIT-DOSE; Type 0: Not a Combination Product 08/24/2013 12/31/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/24/2013 12/31/2017 NORTH COUGH DROP
menthol lozengeProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-1119 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 7 mg Inactive Ingredients Ingredient Name Strength CORN SYRUP (UNII: 9G5L16BK6N) EUCALYPTUS OIL (UNII: 2R04ONI662) FD&C RED NO. 40 (UNII: WZB9127XOA) SUCROSE (UNII: C151H8M554) WATER (UNII: 059QF0KO0R) Product Characteristics Color red (RED) Score no score Shape OVAL Size 22mm Flavor CHERRY (CHERRY) Imprint Code B Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-1119-00 1 in 1 POUCH; Type 0: Not a Combination Product 01/02/2017 09/18/2018 2 NDC:0498-1119-05 50 in 1 BOX; Type 0: Not a Combination Product 01/02/2017 09/18/2018 3 NDC:0498-1119-10 100 in 1 BOX; Type 0: Not a Combination Product 01/02/2017 09/18/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/24/2013 09/18/2018 Labeler - Honeywell Safety Products USA, Inc (118768815) Registrant - Honeywell Safety Products USA, Inc (118768815)