WOMENS LAXATIVE- bisacodyl tablet, coated
SUPERVALU INC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Equaline 44-326 Delisted
Uses
- for gentle, overnight relief of occasional constipation (irregularity)
- this product generally produces bowel movement in 6 to 12 hours
Warnings
Ask a doctor before use if you have
- abdominal pain
- nausea
- vomiting
- sudden change in bowel movements that persists over a period of 2 weeks
Directions
- do not chew or crush tablets
- do not take within 1 hour after taking an antacid or milk
|
adults and children |
oral dosage is 1 to 3 tablets in |
|
children 6 to under |
oral dosage is 1 tablet in a |
|
children under |
ask a doctor |
Other information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- protect from excessive moisture
- see end flap for expiration date and lot number
Inactive ingredients
acacia, ammonium hydroxide, black iron oxide, calcium carbonate, carnauba wax, colloidal silicon dioxide, corn starch, D&C red #27 aluminum lake, FD&C blue #2 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, lactose, magnesium stearate, methylparahydroxy benzoate, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, povidone, propylene glycol, propylparahydroxy benzoate, shellac glaze, simethicone, sodium alginate, sodium benzoate, sodium bicarbonate, stearic acid, sucrose, talc, titanium dioxide, triacetin, triethyl citrate
Principal display panel
EQUALINE®
women’s laxative
tablets
bisacodyl 5mg
(stimulant laxative)
compare to
Correctol®
active ingredient*
NDC 41163-326-52
• gentle, dependable overnight relief
• enteric coated
90 tablets
actual size
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
DOES NOT CONTAIN GLUTEN
*This product is not manufactured or distributed by
MSD Consumer Care, Inc., owner of the registered
trademark Correctol®.
50844 ORG041232652
DISTRIBUTED BY SUPERVALU INC.
EDEN PRAIRIE, MN 55344 USA
SUPERVALU QUALITY Guaranteed
We’re committed to your
satisfaction and guarantee
the quality of this product.
Contact us at 1-877-932-7948, or
www.supervalu-ourownbrands.com.
Please have package available.
Equaline 44-326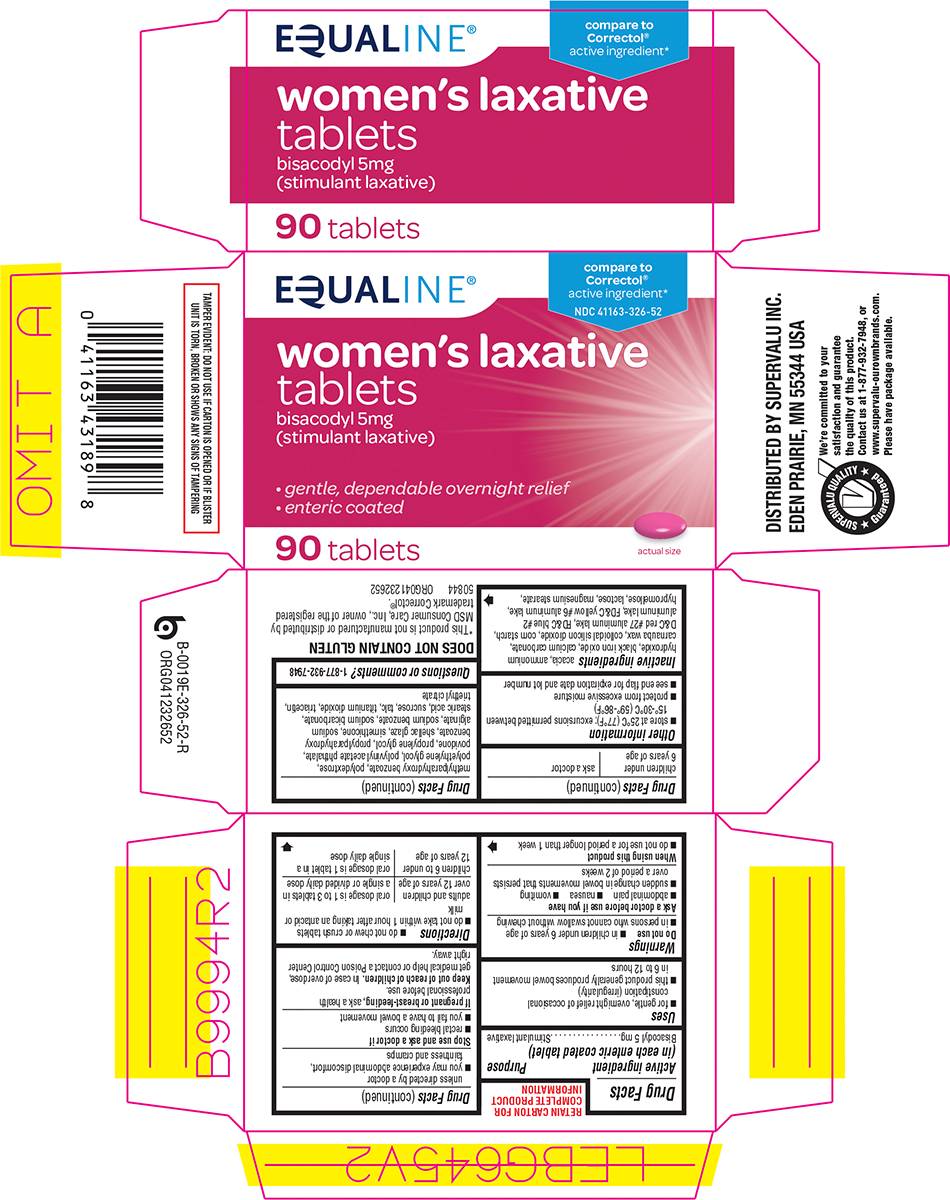
| WOMENS LAXATIVE
bisacodyl tablet, coated |
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - SUPERVALU INC. (006961411) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | PACK(41163-326) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | MANUFACTURE(41163-326) | |