MECLIZINE HYDROCHLORIDE- meclizine hydrochloride tablet
Bryant Ranch Prepack
----------
Rx Only
DESCRIPTION
Meclizine hydrochloride, an oral antiemetic, is a white, slightly yellowish, crystalline powder which has a slight odor and is tasteless. It has the following structural formula:
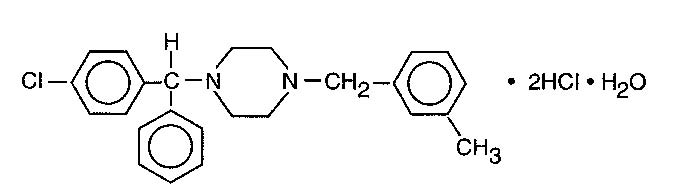
C25H27CIN2•2HCI•H2O M.W. 481.89
The chemical name is 1-(p-chloro-alpha-phenylbenzyl)-4-(m-methyl-benzyl) - piperazine dihydrochloride monohydrate.
Meclizine Hydrochloride Tablets are available in 12.5 mg, and *25 mg strengths for oral administration.
*Contains FD&C Yellow #5 (see PRECAUTIONS).
Each tablet contains the following inactive ingredients: colloidal silicon dioxide, lactose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, starch, and stearic acid. In addition, the 12.5 mg tablet contains FD&C Blue #1; and the 25 mg tablet contains D&C Yellow #10 and FD&C Yellow #5.
CLINICAL PHARMACOLOGY
Meclizine hydrochloride is an antihistamine which shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
Pharmacokinetics
The available pharmacokinetic information for meclizine following oral administration has been summarized from published literature.
Absorption
Meclizine is absorbed after oral administration with maximum plasma concentrations reaching at a median Tmax value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Distribution
Drug distribution characteristics for meclizine in humans in unknown.
Metabolism
The metabolic fate of meclizine in humans in unknown. In an in vitro metabolic study using human hepatic microsome and recombinant CYP enzyme, CYP2D6 was found to be the dominant enzyme for metabolism of meclizine.
The genetic polymorphism of CYP2D6 that results in extensive-, poor-,intermediate- and ultrarapid metabolizer phenotypes could contribute to large inter-individual variability in meclizine exposure.
Elimination
Meclizine has a plasma elimination half-life of about 5 to 6 hours in humans.
INDICATIONS AND USAGE
INDICATIONS
Based on a review of this drug by the National Academy of Sciences – National Research Council and/or other information, FDA has classified the indications as follows:
Effective: Management of nausea and vomiting, and dizziness associated with motion sickness.
Possibly Effective: Management of vertigo associated with diseases affecting the vestibular system.
Final classification of the less than effective indications required further investigation.
CONTRAINDICATIONS
Meclizine hydrochloride is contraindicated in individuals who have shown a previous hypersensitivity to it.
WARNINGS
Since drowsiness may, on occasion, occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery.
Patients should avoid alcoholic beverages while taking the drug. Due to its potential anticholinergic action, this drug should be used with caution in patients with asthma, glaucoma, or enlargement of the prostate gland.
Usage in Children
Clinical studies establishing safety and effectiveness in children have not been done; therefore, usage is not recommended in children under 12 years of age.
Usage in Pregnancy
Pregnancy Category B. Reproduction studies in rats have shown cleft palates at 25 to 50 times the human dose. Epidemiological studies in pregnant women, however, do not indicate that meclizine increases the risk of abnormalities when administered during pregnancy. Despite the animal findings, it would appear that the possibility of fetal harm is remote. Nevertheless, meclizine, or any other medication, should be used during pregnancy only if clearly necessary.
PRECAUTIONS
The Meclizine Hydrochloride Tablets, 25 mg contain FD&C Yellow #5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible individuals. Although the overall incidence of FD&C Yellow #5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when meclizine is administered to a nursing woman.
Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetic of meclizine has not been evaluated. As meclizine undergoes metabolism, hepatic impairment may result in increased systemic exposure of the drug. Treatment with meclizine should be administered with caution in patients with hepatic impairment.
Renal Impairment
The effect of renal impairment on the pharmacokinetics of meclizine has not been evaluated. Due to a potential for drug/metabolite accumulation, meclizine should be administered with caution in patients with renal impairment and in the elderly as renal function generally declines with age.
Drug Interactions
There may be increased CNS depression when meclizine is administered concurrently with other CNS depressants, including alcohol, tranquilizers, and sedatives. (see WARNINGS).
Based on in-vitro evaluation, meclizine is metabolized by CYP2D6. Therefore there is a possibility for a drug interaction between meclizine and CYP2D6 inhibitors.
ADVERSE REACTIONS
Anaphylactoid reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision have been reported.
DOSAGE AND ADMINISTRATION
Vertigo
For the control of vertigo associated with diseases affecting the vestibular system, the recommended dose is 25 to 100mg daily, in divided dosage, depending upon clinical response.
Motion Sickness: The initial dose of 25 to 50 mg meclizine hydrochloride, should be taken one hour prior to embarkation for protection against motion sickness. Thereafter, the dose may be repeated every 24 hours for the duration of the journey.
| MECLIZINE HYDROCHLORIDE
meclizine hydrochloride tablet |
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Bryant Ranch Prepack (171714327) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bryant Ranch Prepack | 171714327 | REPACK(63629-1266) , RELABEL(63629-1266) | |
