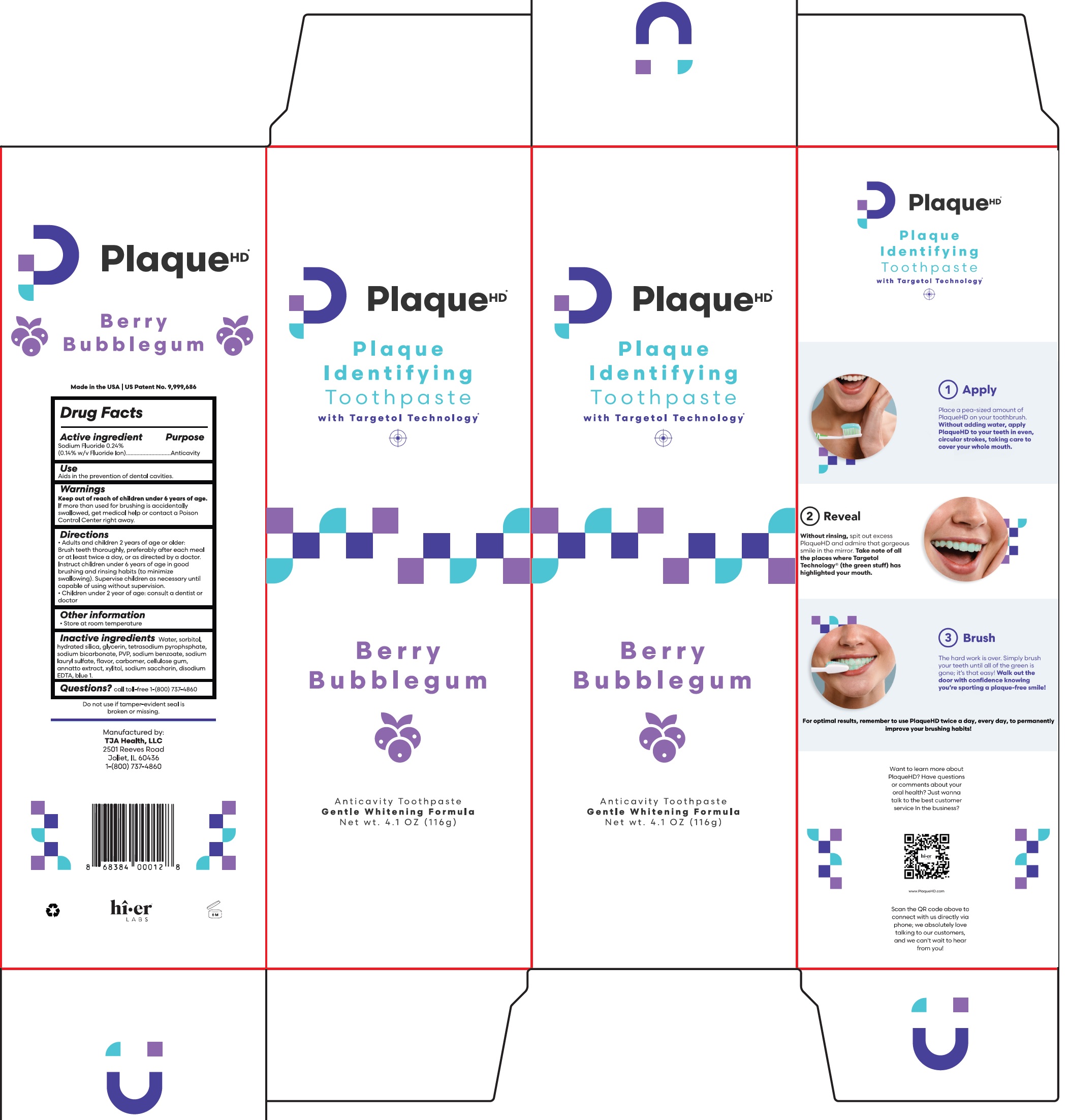
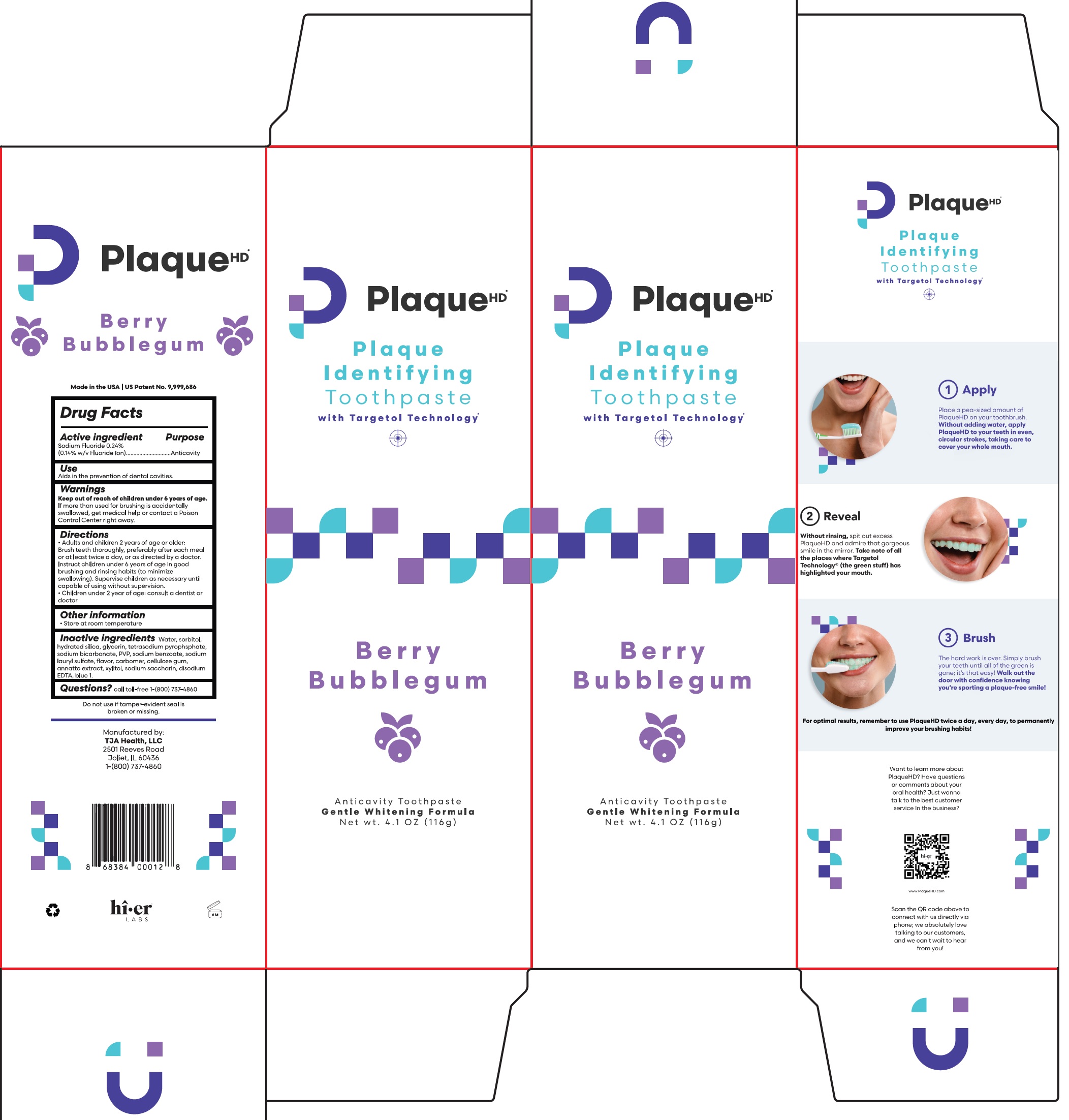
Label: PLAQUE HD ANTICAVITY BERRY BUBBLE GUM FLAVOR PROFESSIONAL PLAQUE-IDENTIFYING- sodium fluoride paste, dentifrice
- NDC Code(s): 57660-001-01, 57660-001-02
- Packager: TJA Health LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Use
- Warnings
-
Directions
- Adults and children 2 years of age or older: Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a doctor. Instruct children under 6 years of age in good brushing and rinsing habits (to minimize swallowing). Supervise children as necessary until capable of using without supervision.
- Children under 2 year of age: Consult a dentist or doctor
- Other information
- Inactive ingredients
- Questions?
- Package Labeling (57660-001-01)
- Representative Labeling (57660-001-02)
-
INGREDIENTS AND APPEARANCE
PLAQUE HD ANTICAVITY BERRY BUBBLE GUM FLAVOR PROFESSIONAL PLAQUE-IDENTIFYING
sodium fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57660-001 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.14 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) HYDRATED SILICA (UNII: Y6O7T4G8P9) GLYCERIN (UNII: PDC6A3C0OX) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) SODIUM BICARBONATE (UNII: 8MDF5V39QO) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM LAURYL SULFATE (UNII: 368GB5141J) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) ANNATTO (UNII: 6PQP1V1B6O) XYLITOL (UNII: VCQ006KQ1E) SACCHARIN SODIUM (UNII: SB8ZUX40TY) EDETATE DISODIUM (UNII: 7FLD91C86K) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Product Characteristics Color Score Shape Size Flavor BERRY, BUBBLE GUM (Berry Bubble Gum Flavor) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57660-001-01 1 in 1 PACKAGE 03/19/2014 1 116 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:57660-001-02 1 in 1 PACKAGE 03/19/2014 2 116 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 03/19/2014 Labeler - TJA Health LLC (078799634) Registrant - TJA Health LLC (078799634) Establishment Name Address ID/FEI Business Operations TJA Health LLC 078799634 manufacture(57660-001)