ALLEGRA- fexofenadine hydrochloride tablet, orally disintegrating
ALLEGRA- fexofenadine hydrochloride tablet, film coated
ALLEGRA- fexofenadine hydrochloride suspension
sanofi-aventis U.S. LLC
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ALLEGRA safely and effectively. See full prescribing information for ALLEGRA.
ALLEGRA (fexofenadine hydrochloride) tablet, orally disintegrating for oral use ALLEGRA (fexofenadine hydrochloride) tablet, film coated for oral use ALLEGRA (fexofenadine hydrochloride) suspension for oral use Initial U.S. Approval: 1996 INDICATIONS AND USAGEDOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSCONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions (≥ 2%) in subjects age 12 years and older were headache, back pain, dizziness, stomach discomfort, and pain in extremity. In subjects aged 6 to 11 years, cough, upper respiratory tract infection, pyrexia and otitis media were more frequently reported. In subjects aged 6 months to 5 years, vomiting, diarrhea, somnolence/fatigue and rhinorrhea were more frequently reported. (6.1) Other adverse reactions have been reported. (6) To report SUSPECTED ADVERSE REACTIONS, contact sanofi-aventis at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION Revised: 3/2012 |
|||||||||||||||||||||||||
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 ALLEGRA tablets
Seasonal Allergic Rhinitis and Chronic Idiopathic Urticaria
Adults and Children 12 Years and Older
The recommended dose of ALLEGRA tablets is 60 mg twice daily or 180 mg once daily with water. A dose of 60 mg once daily is recommended as the starting dose in patients with decreased renal function [see Clinical Pharmacology (12.3)].
Children 6 to 11 Years.
The recommended dose of ALLEGRA tablets is 30 mg twice daily with water. A dose of 30 mg once daily is recommended as the starting dose in pediatric patients with decreased renal function [see Clinical Pharmacology (12.3)].
2.2 ALLEGRA ODT
Seasonal Allergic Rhinitis and Chronic Idiopathic Urticaria
Children 6 to 11 Years:
ALLEGRA ODT is intended for use only in children 6 to 11 years of age. The recommended dose of ALLEGRA ODT is 30 mg twice daily. A dose of 30 mg once daily is recommended as the starting dose in pediatric patients with decreased renal function [see Clinical Pharmacology (12.3)].
ALLEGRA ODT is designed to disintegrate on the tongue, followed by swallowing with or without water. ALLEGRA ODT should be taken on an empty stomach. ALLEGRA ODT is not intended to be chewed.
ALLEGRA ODT should not be removed from the original blister package until the time of administration.
2.3 ALLEGRA oral suspension
Seasonal Allergic Rhinitis
Children 2 to 11 Years:
The recommended dose of ALLEGRA oral suspension is 30 mg twice daily. A dose of 30 mg (5 mL) once daily is recommended as the starting dose in pediatric patients with decreased renal function [see Clinical Pharmacology (12.3)].
Shake bottle well, before each use.
Chronic Idiopathic Urticaria
Children 6 Months to 11 Years:
The recommended dose of ALLEGRA oral suspension is 30 mg (5 mL) twice daily for patients 2 to 11 years of age and 15 mg (2.5 mL) twice daily for patients 6 months to less than 2 years of age. For pediatric patients with decreased renal function, the recommended starting doses of ALLEGRA oral suspension are 30 mg (5 mL) once daily for patients 2 to 11 years of age and 15 mg (2.5 mL) once daily for patients 6 months to less than 2 years of age [see Clinical Pharmacology (12.3)].
Shake bottle well, before each use.
3 DOSAGE FORMS AND STRENGTHS
ALLEGRA tablets are available in 30 mg, 60 mg, and 180 mg strengths. ALLEGRA tablets are coated with a peach colored film coating. Tablets have the following unique shape and identifiers: 30 mg tablets are round, bi-convex and have 03 on one side and a scripted "e" on the other; 60 mg tablets are oval, bi-convex and have 06 on one side and a scripted "e" on the other; and 180 mg tablets are oblong, bi-convex and have 018 on one side and a scripted "e" on the other.
ALLEGRA ODT is available as a 30 mg orally disintegrating tablet and is white, flat-faced, ½-inch round shaped with beveled edges and debossed with a scripted "e" on one side and "311AV" on the other side.
ALLEGRA oral suspension is available as 30 mg/ 5 mL (6 mg/mL).
4 CONTRAINDICATIONS
ALLEGRA tablets, ALLEGRA ODT and ALLEGRA oral suspension are contraindicated in patients with known hypersensitivity to fexofenadine and any of the ingredients of ALLEGRA. Rare cases of hypersensitivity reactions with manifestations such as angioedema, chest tightness, dyspnea, flushing and systemic anaphylaxis have been reported.
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described below reflect exposure to fexofenadine hydrochloride in 5083 patients in trials for allergic rhinitis and chronic idiopathic urticaria. In these trials, 3010 patients 12 years of age and older with seasonal allergic rhinitis were exposed to fexofenadine hydrochloride at doses of 20 to 240 mg twice daily or 120 to 180 mg once daily. A total of 646 patients 6 to 11 years of age with seasonal allergic rhinitis were exposed to fexofenadine hydrochloride at doses of 15 to 60 mg twice daily. The duration of treatment in these trials was 2 weeks. A total of 534 patients 6 months to 5 years of age with allergic rhinitis were exposed to fexofenadine hydrochloride at doses of 15 to 30 mg twice daily. The duration of treatment in these trials ranged from 1 day to 2 weeks. There were 893 patients 12 years of age and older with chronic idiopathic urticaria exposed to fexofenadine hydrochloride at doses of 20 to 240 mg twice daily or 180 mg once daily. The duration of treatment in these trials was 4 weeks.
Seasonal Allergic Rhinitis
Adults and Adolescents: In placebo-controlled seasonal allergic rhinitis clinical trials in subjects 12 years of age and older, 2439 subjects received fexofenadine hydrochloride capsules at doses of 20 mg to 240 mg twice daily. All adverse reactions that were reported by greater than 1% of subjects who received the recommended daily dose of fexofenadine hydrochloride (60 mg capsules twice daily) are listed in Table 1.
In another placebo-controlled clinical study in the United States, 571 subjects aged 12 years and older received fexofenadine hydrochloride tablets at doses of 120 or 180 mg once daily. Table 1 also lists adverse reactions that were reported by greater than 2% of subjects treated with fexofenadine hydrochloride tablets at doses of 180 mg once daily.
The incidence of adverse reactions, including somnolence/fatigue, was not dose-related and was similar across subgroups defined by age, gender, and race.
| Twice-daily dosing with fexofenadine capsules at rates of greater than 1% | ||
| Adverse reaction | Fexofenadine 60 mg
Twice Daily | Placebo
Twice Daily |
| (n=680) | (n=674) | |
| Frequency | Frequency | |
| Dysmenorrhea | 1.5% | 0.3% |
| Once-daily dosing with fexofenadine hydrochloride tablets at rates of greater than 2% | ||
| Adverse reaction | Fexofenadine 180 mg
Once Daily | Placebo
|
| (n=283) | (n=293) | |
| Frequency | Frequency | |
| Headache | 10.3% | 7.2% |
| Back Pain | 2.5% | 1.4% |
The frequency and magnitude of laboratory abnormalities were similar in fexofenadine hydrochloride- and placebo-treated subjects.
Pediatrics: Table 2 lists adverse reactions in subjects aged 6 years to 11 years of age which were reported by greater than 2% of subjects treated with fexofenadine hydrochloride tablets at a dose of 30 mg twice daily in placebo-controlled seasonal allergic rhinitis studies in the United States and Canada.
| Adverse reaction | Fexofenadine 30 mg
Twice Daily (n=209) | Placebo
(n=229) |
|---|---|---|
| Frequency | Frequency | |
| Cough | 3.8% | 1.3% |
| Upper Respiratory Tract Infection | 2.9% | 0.9% |
| Pyrexia | 2.4% | 0.9% |
| Otitis Media | 2.4% | 0.0% |
Table 3 lists adverse reactions in subjects 6 months to 5 years of age which were reported by greater than 2% of subjects treated with fexofenadine hydrochloride in 3 open single- and multiple-dose pharmacokinetic studies and 3 placebo-controlled safety studies with fexofenadine hydrochloride capsule content (484 subjects) and suspension (50 subjects) at doses of 15 mg (108 subjects) and 30 mg (426 subjects) given twice a day.
| Adverse reaction | Fexofenadine 15 mgTwice Daily | Fexofenadine 30 mgTwice Daily | Fexofenadine Total
Twice Daily | Placebo |
|---|---|---|---|---|
| (n=108) | (n=426) | (n=534) | (n=430) | |
| Frequency | Frequency | Frequency | Frequency | |
| Vomiting | 12.0% | 4.2% | 5.8% | 8.6% |
| Diarrhea | 3.7% | 2.8% | 3.0% | 2.6% |
| Somnolence/Fatigue | 2.8% | 0.9% | 1.3% | 0.2% |
| Rhinorrhea | 0.9% | 2.1% | 1.9% | 0.9% |
Chronic Idiopathic Urticaria
Adverse reactions reported by subjects 12 years of age and older in placebo-controlled chronic idiopathic urticaria studies were similar to those reported in placebo-controlled seasonal allergic rhinitis studies.
In placebo-controlled chronic idiopathic urticaria clinical trials, 726 subjects 12 years of age and older received fexofenadine hydrochloride tablets at doses of 20 to 240 mg twice daily. Table 4 lists adverse reactions in subjects aged 12 years and older which were reported by greater than 2% of subjects treated with fexofenadine hydrochloride 60 mg tablets twice daily in controlled clinical studies in the United States and Canada.
In a placebo-controlled clinical study in the United States, 167 subjects aged 12 years and older received fexofenadine hydrochloride 180 mg tablets. Table 4 also lists adverse reactions that were reported by greater than 2% of subjects treated with fexofenadine hydrochloride tablets at doses of 180 mg once daily.
| Twice-daily dosing with fexofenadine hydrochloride in studies in the United States and Canada at rates of greater than 2% | ||
| Adverse reaction | Fexofenadine 60 mg Twice Daily (n=191) | Placebo (n=183) |
| Frequency | Frequency | |
| Dizziness | 2.1% | 1.1% |
| Back Pain | 2.1% | 1.1% |
| Stomach discomfort | 2.1% | 0.6% |
| Pan in extremity | 2.1% | 0.0% |
| Once-daily dosing with fexofenadine hydrochloride in a study in the United States at rates of greater than 2% | ||
| Adverse reaction | Fexofenadine 180 mg
Once Daily (n=167) | Placebo(n=92) |
| Frequency | Frequency | |
| Headache | 4.8% | 3.3% |
The safety of fexofenadine hydrochloride in the treatment of chronic idiopathic urticaria in pediatric patients 6 months to 11 years of age is based on the safety profile of fexofenadine hydrochloride in adults and pediatric patients at doses equal to or higher than the recommended dose [see Use in Specific Populations (8.4)]
6.2 Postmarketing Experience
In addition to the adverse reactions reported during clinical studies and listed above, the following adverse events have been identified during post-approval use of ALLEGRA. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Events that have been reported rarely during postmarketing experience include: insomnia, nervousness, sleep disorders or paroniria, and hypersensitivity reactions (including anaphylaxis, urticaria, angioedema, chest tightness, dyspnea, flushing, pruritus, and rash).
7 DRUG INTERACTIONS
7.1 Antacids
Fexofenadine hydrochloride should not be taken closely in time with aluminum and magnesium containing antacids. In healthy adult subjects, administration of 120 mg of fexofenadine hydrochloride (2 x 60 mg capsule) within 15 minutes of an aluminum and magnesium containing antacid (Maalox®) decreased fexofenadine AUC by 41% and Cmax by 43%.
7.2 Erythromycin and Ketoconazole
Fexofenadine has been shown to exhibit minimal (ca. 5%) metabolism. However, co–administration of fexofenadine hydrochloride with either ketoconazole or erythromycin led to increased plasma concentrations of fexofenadine in healthy adult subjects. Fexofenadine had no effect on the pharmacokinetics of either erythromycin or ketoconazole. In 2 separate studies in healthy adult subjects, fexofenadine hydrochloride 120 mg twice daily (240 mg total daily dose) was co-administered with either erythromycin 500 mg every 8 hours or ketoconazole 400 mg once daily under steady-state conditions to healthy adult subjects (n=24, each study). No differences in adverse events or QTc interval were observed when subjects were administered fexofenadine hydrochloride alone or in combination with either erythromycin or ketoconazole. The findings of these studies are summarized in the following table
| Concomitant Drug | CmaxSS
(Peak plasma concentration) | AUCss(0–12h)
(Extent of systemic exposure) |
|---|---|---|
| Erythromycin (500 mg every 8 hrs) | +82% | +109% |
| Ketoconazole (400 mg once daily) | +135% | +164% |
The changes in plasma levels were within the range of plasma levels achieved in adequate and well-controlled clinical trials.
The mechanism of these interactions has been evaluated in in vitro, in situ, and in vivo animal models. These studies indicate that ketoconazole or erythromycin co-administration enhances fexofenadine gastrointestinal absorption. This observed increase in the bioavailability of fexofenadine may be due to transport-related effects, such as p-glycoprotein. In vivo animal studies also suggest that in addition to enhancing absorption, ketoconazole decreases fexofenadine gastrointestinal secretion, while erythromycin may also decrease biliary excretion.
7.3 Fruit Juices
Fruit juices such as grapefruit, orange and apple may reduce the bioavailability and exposure of fexofenadine. This is based on the results from 3 clinical studies using histamine induced skin wheals and flares coupled with population pharmacokinetic analysis. The size of wheal and flare were significantly larger when fexofenadine hydrochloride was administered with either grapefruit or orange juices compared to water. Based on the literature reports, the same effects may be extrapolated to other fruit juices such as apple juice. The clinical significance of these observations is unknown. In addition, based on the population pharmacokinetics analysis of the combined data from grapefruit and orange juices studies with the data from a bioequivalence study, the bioavailability of fexofenadine was reduced by 36%. Therefore, to maximize the effects of fexofenadine, it is recommended that ALLEGRA tablets should be taken with water [see Clinical Pharmacology (12.3) and Dosage and Administration (2.2)].
ALLEGRA ODT can be taken with or without water.(see Clinical Pharmacology (12.3) and Dosage and Administration (2.1)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C. There was no evidence of teratogenicity in rats or rabbits at oral doses of terfenadine up to 300 mg/kg (which led to fexofenadine exposures that were approximately 4 and 30 times, respectively, the exposure at the maximum recommended human daily oral dose of 180 mg of fexofenadine hydrochloride based on comparison of AUCs).
In mice, no adverse effects and no teratogenic effects during gestation were observed with fexofenadine hydrochloride at oral doses up to 3730 mg/kg (which led to fexofenadine exposures that were approximately 15 times the exposure at the maximum recommended human daily oral dose of 180 mg of fexofenadine hydrochloride based on comparison of AUCs).
There are no adequate and well controlled studies in pregnant women. Fexofenadine hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects: Dose-related decreases in pup weight gain and survival were observed in rats exposed to an oral dose of 150 mg/kg of terfenadine (which led to fexofenadine exposures that were approximately 3 times the exposure at the maximum recommended human daily oral dose of 180 mg of fexofenadine hydrochloride based on comparison of AUCs).
8.3 Nursing Mothers
It is not known if fexofenadine is excreted in human milk. There are no adequate and well-controlled studies in women during lactation. Because many drugs are excreted in human milk, caution should be exercised when fexofenadine hydrochloride is administered to a nursing woman.
8.4 Pediatric Use
The recommended doses of fexofenadine hydrochloride in pediatric patients 6 months to 11 years of age are based on cross-study comparison of the pharmacokinetics of fexofenadine in adults and pediatric subjects and on the safety profile of fexofenadine hydrochloride in both adult and pediatric subjects at doses equal to or higher than the recommended doses.The safety and effectiveness of fexofenadine hydrochloride in pediatric patients under 6 months of age have not been established.
The safety of fexofenadine hydrochloride is based on the administration of ALLEGRA tablets at a dose of 30 mg twice daily demonstrated in 438 pediatric subjects 6 years to 11 years of age in 2 placebo-controlled 2-week seasonal allergic rhinitis trials. The safety of fexofenadine hydrochloride at doses of 15mg and 30 mg given once and twice a day has been demonstrated in 969 pediatric subjects (6 months to 5 years of age) with allergic rhinitis in 3 pharmacokinetic studies and 3 safety studies. The safety of fexofenadine hydrochloride for the treatment of chronic idiopathic urticaria in subjects 6 months to 11 years of age is based on cross-study comparison of the pharmacokinetics of ALLEGRA in adult and pediatric subjects and on the safety profile of fexofenadine in both adult and pediatric subjects at doses equal to or higher than the recommended dose.
The effectiveness of fexofenadine hydrochloride for the treatment of seasonal allergic rhinitis in subjects 6 to 11 years of age was demonstrated in 1 trial (n=411) in which ALLEGRA tablets 30 mg twice daily significantly reduced total symptom scores compared to placebo, along with extrapolation of demonstrated efficacy in subjects aged 12 years and above, and the pharmacokinetic comparisons in adults and children. The effectiveness of fexofenadine hydrochloride 30 mg twice daily for the treatment of seasonal allergic rhinitis in patients 2 to 5 years of age is based on the pharmacokinetic comparisons in adult and pediatric subjects and an extrapolation of the demonstrated efficacy of fexofenadine hydrochloride in adult subjects with this condition and the likelihood that the disease course, pathophysiology, and the drug's effect are substantially similar in pediatric patients to those in adult patients. The effectiveness of fexofenadine hydrochloride for the treatment of chronic idiopathic urticaria in patients 6 months to 11 years of age is based on the pharmacokinetic comparisons in adults and children and an extrapolation of the demonstrated efficacy of ALLEGRA in adults with this condition and the likelihood that the disease course, pathophysiology and the drug's effect are substantially similar in children to that of adult patients. Administration of a 15 mg dose of fexofenadine hydrochloride to pediatric subjects 6 months to less than 2 years of age and a 30 mg dose to pediatric subjects 2 to 11 years of age produced exposures comparable to those seen with a dose of 60 mg administered to adults.
8.5 Geriatric Use
Clinical studies of ALLEGRA tablets and capsules did not include sufficient numbers of subjects aged 65 years and over to determine whether this population responds differently from younger subjects. Other reported clinical experience has not identified differences in responses between the geriatric and younger subjects. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Based on increases in bioavailability and half-life, a dose of 60 mg once daily is recommended as the starting dose in adult patients with decreased renal function (mild, moderate or severe renal impairment). For pediatric patients with decreased renal function (mild, moderate or severe renal impairment), the recommended starting dose of fexofenadine is 30 mg once daily for patients 2 to 11 years of age and 15 mg once daily for patients 6 months to less than 2 years of age. [See Dosage and Administration (2.2,2.3 ) and Clinical Pharmacology (12.3)]
10 OVERDOSAGE
Dizziness, drowsiness, and dry mouth have been reported with fexofenadine hydrochloride overdose. Single doses of fexofenadine hydrochloride up to 800 mg (6 healthy subjects at this dose level), and doses up to 690 mg twice daily for 1 month (3 healthy subjects at this dose level) or 240 mg once daily for 1 year (234 healthy subjects at this dose level) were administered without the development of clinically significant adverse events as compared to placebo.
In the event of overdose, consider standard measures to remove any unabsorbed drug. Symptomatic and supportive treatment is recommended. Following administration of terfenadine, hemodialysis did not effectively remove fexofenadine, the major active metabolite of terfenadine, from blood (up to 1.7% removed).
No deaths occurred at oral doses of fexofenadine hydrochloride up to 5000 mg/kg in mice (110 times the maximum recommended daily oral dose in adults and children based on mg/m2) and up to 5000 mg/kg in rats (230 times the maximum recommended daily oral dose in adults and 210 times the maximum recommended daily oral dose in children based on mg/m2). Additionally, no clinical signs of toxicity or gross pathological findings were observed. In dogs, no evidence of toxicity was observed at oral doses up to 2000 mg/kg (300 times the maximum recommended daily oral dose in adults and 280 times the maximum recommended daily oral dose in children based on mg/m2).
11 DESCRIPTION
Fexofenadine hydrochloride, the active ingredient of ALLEGRA tablets, ALLEGRA ODT and ALLEGRA oral suspension, is a histamine H1-receptor antagonist with the chemical name (±)-4-[1 hydroxy-4-[4-(hydroxydiphenylmethyl)-1-piperidinyl]-butyl]-α, α-dimethyl benzeneacetic acid hydrochloride. It has the following chemical structure
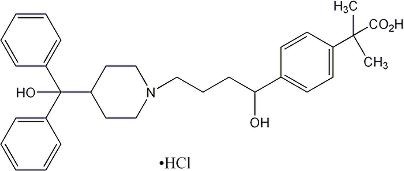
The molecular weight is 538.13 and the empirical formula is C32H39NO4•HCl.
Fexofenadine hydrochloride is a white to off-white crystalline powder. It is freely soluble in methanol and ethanol, slightly soluble in chloroform and water, and insoluble in hexane. Fexofenadine hydrochloride is a racemate and exists as a zwitterion in aqueous media at physiological pH.
ALLEGRA is formulated as a tablet for oral administration. Each tablet contains 30, 60, or 180 mg fexofenadine hydrochloride (depending on the dosage strength) and the following excipients: croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The aqueous tablet film coating is made from hypromellose, iron oxide blends, polyethylene glycol, povidone, silicone dioxide, and titanium dioxide.
ALLEGRA ODT is formulated for disintegration in the mouth immediately following administration. Each orally disintegrating tablet contains 30 mg fexofenadine hydrochloride and the following excipients: citric acid anhydrous, crospovidone, magnesium stearate, mannitol, methacrylate copolymer, microcrystalline cellulose, povidone K-30, sodium bicarbonate, sodium starch glycolate, aspartame, natural and artificial orange flavor, artificial cream flavor, and alcohol anhydrous; the alcohol is predominantly removed during the manufacturing process.
ALLEGRA oral suspension, a white uniform suspension, contains 6 mg fexofenadine hydrochloride per mL and the following excipients: propylene glycol, edetate disodium, propylparaben, butylparaben, xanthan gum, poloxamer 407, titanium dioxide, sodium phosphate monobasic monohydrate, sodium phosphate dibasic heptahydrate, artificial raspberry cream flavor, sucrose, xylitol and purified water.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Fexofenadine hydrochloride, the major active metabolite of terfenadine, is an antihistamine with selective H1-receptor antagonist activity. Both enantiomers of fexofenadine hydrochloride displayed approximately equipotent antihistaminic effects. Fexofenadine hydrochloride inhibited antigen-induced bronchospasm in sensitized guinea pigs and histamine release from peritoneal mast cells in rats. The clinical significance of these findings is unknown. In laboratory animals, no anticholinergic or alpha1-adrenergic blocking effects were observed. Moreover, no sedative or other central nervous system effects were observed. Radiolabeled tissue distribution studies in rats indicated that fexofenadine does not cross the blood-brain barrier.
12.2 Pharmacodynamics
Wheal and Flare. Human histamine skin wheal and flare studies in adults following single and twice daily doses of 20 and 40 mg fexofenadine hydrochloride demonstrated that the drug exhibits an antihistamine effect by 1 hour, achieves maximum effect at 2 to 3 hours, and an effect is still seen at 12 hours. There was no evidence of tolerance to these effects after 28 days of dosing. The clinical significance of these observations is unknown.
Histamine skin wheal and flare studies in 7 to 12 year old subjects showed that following a single dose of 30 or 60 mg, antihistamine effect was observed at 1 hour and reached a maximum by 3 hours. Greater than 49% inhibition of wheal area, and 74% inhibition of flare area were maintained for 8 hours following the 30 and 60 mg dose.
No statistically significant increase in mean QTc interval compared to placebo was observed in 714 adult subjects with seasonal allergic rhinitis given fexofenadine hydrochloride capsules in doses of 60 to 240 mg twice daily for 2 weeks. Pediatric subjects from 2 placebo-controlled trials (n=855) treated with up to 60 mg fexofenadine hydrochloride twice daily demonstrated no significant treatment- or dose-related increases in QTc. In addition, no statistically significant increase in mean QTc interval compared to placebo was observed in 40 healthy adult subjects given fexofenadine hydrochloride as an oral solution at doses up to 400 mg twice daily for 6 days, or in 230 healthy adult subjects given fexofenadine hydrochloride 240 mg once daily for 1 year. In subjects with chronic idiopathic urticaria, there were no clinically relevant differences for any ECG intervals, including QTc, between those treated with fexofenadine hydrochloride 180 mg once daily (n = 163) and those treated with placebo (n = 91) for 4 weeks.
12.3 Pharmacokinetics
The pharmacokinetics of fexofenadine hydrochloride in subjects with seasonal allergic rhinitis and subjects with chronic urticaria were similar to those in healthy subjects.
Absorption:
ALLEGRA tablets: Fexofenadine hydrochloride was absorbed following oral administration of a single dose of two 60 mg capsules to healthy male subjects with a mean time to maximum plasma concentration occurring at 2.6 hours post-dose. After administration of a single 60 mg capsule to healthy adult subjects, the mean maximum plasma concentration (Cmax) was 131 ng/mL. Following single dose oral administrations of either the 60 and 180 mg tablet to healthy adult male subjects, mean Cmax were 142 and 494 ng/mL, respectively. The tablet formulations are bioequivalent to the capsule when administered at equal doses. Fexofenadine hydrochloride pharmacokinetics are linear for oral doses up to a total daily dose of 240 mg (120 mg twice daily). The administration of the 60 mg capsule contents mixed with applesauce did not have a significant effect on the pharmacokinetics of fexofenadine in adults. Co-administration of 180 mg fexofenadine hydrochloride tablet with a high fat meal decreased the mean area under the curve (AUC) and (Cmax) of fexofenadine by 21 and 20% respectively.
ALLEGRA ODT: Fexofenadine hydrochloride was absorbed following single-dose oral administration of ALLEGRA ODT 30 mg to healthy adult subjects with a mean time to maximum plasma concentration occurring at approximately 2.0 hours post-dose. After single-dose administration of ALLEGRA 30 mg ODT to healthy adult subjects, the mean maximum plasma concentration (Cmax) was 88.0 ng/mL. ALLEGRA ODT 30 mg tablets are bioequivalent to the 30 mg ALLEGRA tablets. The administration of ALLEGRA ODT 30 mg with a high-fat meal decreased the AUC and Cmax by approximately 40% and 60% respectively and a 2-hour delay in the time to peak exposure (Tmax) was observed. ALLEGRA ODT should be taken on an empty stomach. The bioavailability of ALLEGRA ODT was comparable whether given with or without water [see Dosage and Administration (2.2)]
ALLEGRA oral suspension: A dose of 5 mL of ALLEGRA oral suspension containing 30 mg of fexofenadine hydrochloride is bioequivalent to a 30 mg dose of ALLEGRA tablets. Following oral administration of a 30 mg dose of ALLEGRA oral suspension to healthy adult subjects, the mean Cmax was 118 ng/mL and occurred at approximately 1 hour. The administration of 30 mg ALLEGRA oral suspension with a high fat meal decreased the AUC and the mean Cmax by approximately 30 and 47%, respectively in healthy adult subjects.
Distribution:
Fexofenadine hydrochloride is 60% to 70% bound to plasma proteins, primarily albumin and α1-acid glycoprotein.
Metabolism:
Approximately 5% of the total dose of fexofenadine hydrochloride was eliminated by hepatic metabolism.
Elimination:
The mean elimination half-life of fexofenadine was 14.4 hours following administration of 60 mg twice daily in healthy adult subjects.
Human mass balance studies documented a recovery of approximately 80% and 11% of the [14C] fexofenadine hydrochloride dose in the feces and urine, respectively. Because the absolute bioavailability of fexofenadine hydrochloride has not been established, it is unknown if the fecal component represents primarily unabsorbed drug or is the result of biliary excretion.
Special Populations:
Pharmacokinetics in renally and hepatically impaired subjects and geriatric subjects, obtained after a single dose of 80 mg fexofenadine hydrochloride, were compared to those from healthy subjects in a separate study of similar design.
Renally Impaired:
In subjects with mild to moderate (creatinine clearance 41–80 mL/min) and severe (creatinine clearance 11–40 mL/min) renal impairment, peak plasma concentrations of fexofenadine were 87% and 111% greater, respectively, and mean elimination half-lives were 59% and 72% longer, respectively, than observed in healthy subjects. Peak plasma concentrations in subjects on dialysis (creatinine clearance ≤10 mL/min) were 82% greater and half-life was 31% longer than observed in healthy subjects. Based on increases in bioavailability and half-life, a dose of 60 mg once daily is recommended as the starting dose in adult patients with decreased renal function. For pediatric patients with decreased renal function, the recommended starting dose of fexofenadine is 30 mg once daily for patients 2 to 11 years of age and 15 mg once daily for patients 6 months to less than 2 years of age.[see Dosage and Administration (2.2,2.3)].
Hepatically Impaired:
The pharmacokinetics of fexofenadine hydrochloride in subjects with hepatic impairment did not differ substantially from that observed in healthy subjects.
Geriatric Subjects:
In older subjects (≥65 years old), peak plasma levels of fexofenadine were 99% greater than those observed in younger subjects (<65 years old). Mean fexofenadine elimination half-lives were similar to those observed in younger subjects.
Pediatric Subjects:
A population pharmacokinetic analysis was performed with data from 77 pediatric subjects (6 months to 12 years of age) with allergic rhinitis and 136 adult subjects. The individual apparent oral clearance estimates of fexofenadine were on average 44% and 36% lower in pediatric subjects 6 to 12 years (n=14) and 2 to 5 years of age (n=21), respectively, compared to adult subjects.
Administration of a 15 mg dose of fexofenadine hydrochloride to pediatric subjects 6 months to less than 2 years of age and a 30 mg dose to pediatric subjects 2 to 11 years of age produced exposures comparable to those seen with a dose of 60 mg administered to adults.
13 NONCLINICAL TOXICOLOGY
13. 1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of fexofenadine was assessed using terfenadine studies with adequate fexofenadine exposure (based on plasma area-under-the-concentration vs. time [AUC] values). No evidence of carcinogenicity was observed in an 18-month study in mice and in a 24-month study in rats at oral doses up to 150 mg/kg of terfenadine (which led to fexofenadine exposures that were approximately 3 and 5 times the exposure at the maximum recommended daily oral dose of fexofenadine hydrochloride in adults [180 mg] and children [60 mg] respectively).
In in vitro (Bacterial Reverse Mutation, CHO/HGPRT Forward Mutation, and Rat Lymphocyte Chromosomal Aberration assays) and in vivo (Mouse Bone Marrow Micronucleus assay) tests, fexofenadine hydrochloride revealed no evidence of mutagenicity.
In rat fertility studies, dose-related reductions in implants and increases in postimplantation losses were observed at an oral dose of 150 mg/kg of terfenadine (which led to fexofenadine exposures that were approximately 3 times the exposure at the maximum recommended human daily oral dose of 180 mg of fexofenadine hydrochloride based on comparison of AUCs). In mice, fexofenadine hydrochloride produced no effect on male or female fertility at average oral doses up to 4438 mg/kg (which led to fexofenadine exposures that were approximately 13 times the exposure at the maximum recommended human daily oral dose of 180 mg of fexofenadine hydrochloride based on comparison of AUCs).
13.2 Animal Toxicology and/or Pharmacology
In dogs (30 mg/kg/orally twice daily for 5 days) and rabbits (10 mg/kg, intravenously over 1 hour), fexofenadine hydrochloride did not prolong QTc. In dogs, the plasma fexofenadine concentration was approximately 9 times the therapeutic plasma concentrations in adults receiving the maximum recommended human daily oral dose of 180 mg. In rabbits, the plasma fexofenadine concentration was approximately 20 times the therapeutic plasma concentration in adults receiving the maximum recommended human daily oral dose of 180 mg. No effect was observed on calcium channel current, delayed K+ channel current, or action potential duration in guinea pig myocytes, or on the delayed rectifier K+ channel cloned from human heart at concentrations up to 1 x 10-5 M of fexofenadine.
14 CLINICAL STUDIES
14.1 Seasonal Allergic Rhinitis
Adults: In three 2-week, multicenter, randomized, double-blind, placebo-controlled trials in subjects 12 to 68 years of age with seasonal allergic rhinitis (n=1634), fexofenadine hydrochloride 60 mg twice daily significantly reduced total symptom scores (the sum of the individual scores for sneezing, rhinorrhea, itchy nose/palate/throat, itchy/watery/red eyes) compared to placebo. Statistically significant reductions in symptom scores were observed following the first 60 mg dose, with the effect maintained throughout the 12-hour interval. In these studies, there was no additional reduction in total symptom scores with higher doses of fexofenadine hydrochloride up to 240 mg twice daily.
In one 2-week, multicenter, randomized, double-blind clinical trial in subjects 12 to 65 years of age with seasonal allergic rhinitis (n=863), fexofenadine hydrochloride 180 mg once daily significantly reduced total symptom scores (the sum of the individual scores for sneezing, rhinorrhea, itchy nose/palate/throat, itchy/watery/red eyes) compared to placebo. Although the number of subjects in some of the subgroups was small, there were no significant differences in the effect of fexofenadine hydrochloride across subgroups of subjects defined by gender, age, and race. Onset of action for reduction in total symptom scores, excluding nasal congestion, was observed at 60 minutes compared to placebo following a single 60 mg fexofenadine hydrochloride dose administered to subjects with seasonal allergic rhinitis who were exposed to ragweed pollen in an environmental exposure unit. In 1 clinical trial conducted with ALLEGRA 60 mg capsules, and in 1 clinical trial conducted with ALLEGRA-D 12 Hour extended release tablets, onset of action was seen within 1 to 3 hours.
Pediatrics: Two 2-week, multicenter, randomized, placebo-controlled, double-blind trials in 877 pediatric subjects 6 to 11 years of age with seasonal allergic rhinitis were conducted at doses of 15, 30, and 60 mg (tablets) twice daily. In 1 of these 2 studies, conducted in 411 pediatric subjects, all 3 doses of fexofenadine hydrochloride significantly reduced total symptom scores (the sum of the individual scores for sneezing, rhinorrhea, itchy nose/palate/throat, itchy/watery/red eyes) compared to placebo, however, a dose-response relationship was not seen. The 60 mg twice daily dose did not provide any additional benefit over the 30 mg twice daily dose in pediatric subjects 6 to 11 years of age.
Administration of a 30 mg dose to pediatric subjects 2 to 11 years of age produced exposures comparable to those seen with a dose of 60 mg administered to adults.[See Clinical Pharmacology (12.3).]
14.2 Chronic Idiopathic Urticaria
Two 4-week, multicenter, randomized, double-blind, placebo-controlled clinical trials compared four different doses of fexofenadine hydrochloride tablet (20, 60, 120, and 240 mg twice daily) to placebo in subjects aged 12 to 70 years with chronic idiopathic urticaria (n=726). Efficacy was demonstrated by a significant reduction in mean pruritus scores (MPS), mean number of wheals (MNW), and mean total symptom scores (MTSS, the sum of the MPS and MNW score). Although all 4 doses were significantly superior to placebo, symptom reduction was greater and efficacy was maintained over the entire 4-week treatment period with fexofenadine hydrochloride doses of ≥60 mg twice daily. However, no additional benefit of the 120 or 240 mg fexofenadine hydrochloride twice daily dose was seen over the 60 mg twice daily dose in reducing symptom scores. There were no significant differences in the effect of fexofenadine hydrochloride across subgroups of subjects defined by gender, age, weight, and race.
In one 4-week, multicenter, randomized, double-blind, placebo-controlled clinical trial in subjects 12 years of age and older with chronic idiopathic urticaria (n=259), fexofenadine hydrochloride 180 mg once daily significantly reduced the mean number of wheals (MNW), the mean pruritus score (MPS), and the mean total symptom score (MTSS, the sum of the MPS and MNW scores). Similar reductions were observed for mean number of wheals and mean pruritus score at the end of the 24-hour dosing interval. Symptom reduction was greater with fexofenadine hydrochloride180 mg than with placebo. Improvement was demonstrated within 1 day of treatment with fexofenadine hydrochloride 180 mg and was maintained over the entire 4-week treatment period. There were no significant differences in the effect of fexofenadine hydrochloride across subgroups of subjects defined by gender, age, and race.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 ALLEGRA tablets
ALLEGRA 30 mg tablets are available in: high-density polyethylene (HDPE) bottles of 100 (NDC 0088-1106-47) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal and HDPE bottles of 500 (NDC 0088-1106-55) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal.
ALLEGRA 60 mg tablets are available in: HDPE bottles of 100 (NDC 0088-1107-47) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal; HDPE bottles of 500 (NDC 0088-1107-55) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal; and aluminum foil-backed clear blister packs of 100 (NDC 0088-1107-49).
ALLEGRA 180 mg tablets are available in: HDPE bottles of 100 (NDC 0088-1109-47) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal and HDPE bottles of 500 (NDC 0088-1109-55) with a polypropylene screw cap containing a pulp/wax liner with heat-sealed foil inner seal.
ALLEGRA tablets are coated with a peach colored film coating. Tablets have the following unique shape and identifiers: 30 mg tablets are round, bi-convex and have 03 on one side and a scripted "e" on the other; 60 mg tablets are oval, bi-convex and have 06 on one side and a scripted "e" on the other; and 180 mg tablets are oblong, bi-convex and have 018 on one side and a scripted "e" on the other.
Store ALLEGRA tablets at controlled room temperature 20–25°C (68–77°F). (See USP Controlled Room Temperature). Foil-backed blister packs containing ALLEGRA tablets should be protected from excessive moisture.
16.2 ALLEGRA ODT
ALLEGRA ODT 30 mg orally disintegrating tablets are available in aluminum-foil blister packs of 60 (NDC 0088-1113-30).
Each ALLEGRA ODT is white, flat-faced, ½-inch round shaped with beveled edges and debossed with a scripted "e" on one side and "311AV" on the other side.
Store ALLEGRA ODT at controlled room temperature 20–25°C (68–77°F). (See USP Controlled Room Temperature). Foil-backed blister packs containing ALLEGRA ODT should be protected from moisture. ALLEGRA ODT should not be removed from the original blister package until the time of administration.
16.3 ALLEGRA oral suspension
ALLEGRA oral suspension (fexofenadine hydrochloride, 30 mg/5 mL (6 mg/mL)) is available in an amber PET bottle containing 300 mL (NDC 0088-1097-20) of suspension.
Store ALLEGRA oral suspension at controlled room temperature 20–25°C (68–77°F). (See USP Controlled Room Temperature.)
Shake bottle well, before each use.
17 PATIENT COUNSELING INFORMATION
Provide the following information to patients and parents/caregivers of pediatric patients taking ALLEGRA tablets, ALLEGRA ODT or ALLEGRA oral suspension:
-
ALLEGRA tablets, ALLEGRA ODT or ALLEGRA oral suspension are prescribed for the relief of symptoms of seasonal allergic rhinitis or for the relief of symptoms of chronic idiopathic urticaria (hives). Instruct patients to take ALLEGRA only as prescribed. Do not exceed the recommended dose.If any untoward effects occur while taking ALLEGRA, discontinue use and consult a doctor.
-
Patients who are hypersensitive to any of the ingredients should not use these products.
-
Patients who are pregnant or nursing should use these products only if the potential benefit justifies the potential risk to the fetus or nursing infant.
-
Advise patients and parents/caregivers of pediatric patients to store the medication in a tightly closed container in a cool, dry place, away from small children.
-
Advise patients and parents/caregivers not to take ALLEGRA with fruit juices.
For ALLEGRA tablets: Advise patients to take the ALLEGRA tablets with water.
For ALLEGRA ODT: Advise patients to take their dose on an empty stomach. Allow ALLEGRA ODT to disintegrate on the tongue before swallowing, with or without water. ALLEGRA ODT is not intended to be chewed. Store ALLEGRA ODT in its original blister package. Do not remove ALLEGRA ODT from the original blister package until the time of administration.
Phenylketonurics: ALLEGRA ODT contains phenylalanine, a component of aspartame. Each 30-mg ALLEGRA ODT contains 5.3 mg phenylalanine. ALLEGRA products other than ALLEGRA ODT do not contain phenylalanine.
For ALLEGRA oral suspension: Advise patients and parents/caregivers of pediatric patients to shake the ALLEGRA oral suspension bottle well, before each use.
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
ALLEGRA ODT manufactured for:
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
©2007 sanofi-aventis U.S. LLC
PRINCIPAL DISPLAY PANEL - 10 Blister Carton
NDC 0088-1113-30
Rx ONLY
allegra®
fexofenadine HCl
ODT Orally Disintegrating Tablets, 30 mg
Ten 6 Tablet Blisters
sanofi aventis

PRINCIPAL DISPLAY PANEL - 30 mg Bottle Label
NDC 0088-1106-47
allegra®
fexofenadine hydrochloride
Tablets
30mg
100 Tablets
sanofi aventis

PRINCIPAL DISPLAY PANEL - 60 mg Bottle Label
NDC 0088-1107-47
allegra®
fexofenadine hydrochloride
Tablets
60mg
100 Tablets
sanofi aventis

PRINCIPAL DISPLAY PANEL - 180 mg Bottle Label
NDC 0088-1109-47
allegra®
fexofenadine
hydrochloride
Tablets
180mg
100 Tablets
sanofi aventis

PRINCIPAL DISPLAY PANEL - 300 mL Bottle Label
NDC 0088-1097-20
allegra®
fexofenadine HCl
Oral Suspension, 30mg/5mL (6mg/mL)
One 300mL Bottle
sanofi aventis
SHAKE WELL BEFORE USING
Rx ONLY
Each mL of ALLEGRA® Oral Suspension contains 6mg
of fexofenadine hydrochloride
Dosage and Administration: See package insert.
WARNING: Keep out of reach of children.
Pharmacist: Dispense in light-resistant, tight
container with child-resistant closure.
Store at controlled room temperature 20–25°C
(68–77°F). [See USP Controlled Room
Temperature].
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
©2008
50087397

| ALLEGRA
fexofenadine hydrochloride tablet, orally disintegrating |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ALLEGRA
fexofenadine hydrochloride tablet, film coated |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| ALLEGRA
fexofenadine hydrochloride tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| ALLEGRA
fexofenadine hydrochloride tablet, film coated |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| ALLEGRA
fexofenadine hydrochloride suspension |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - sanofi-aventis U.S. LLC (783243835) |