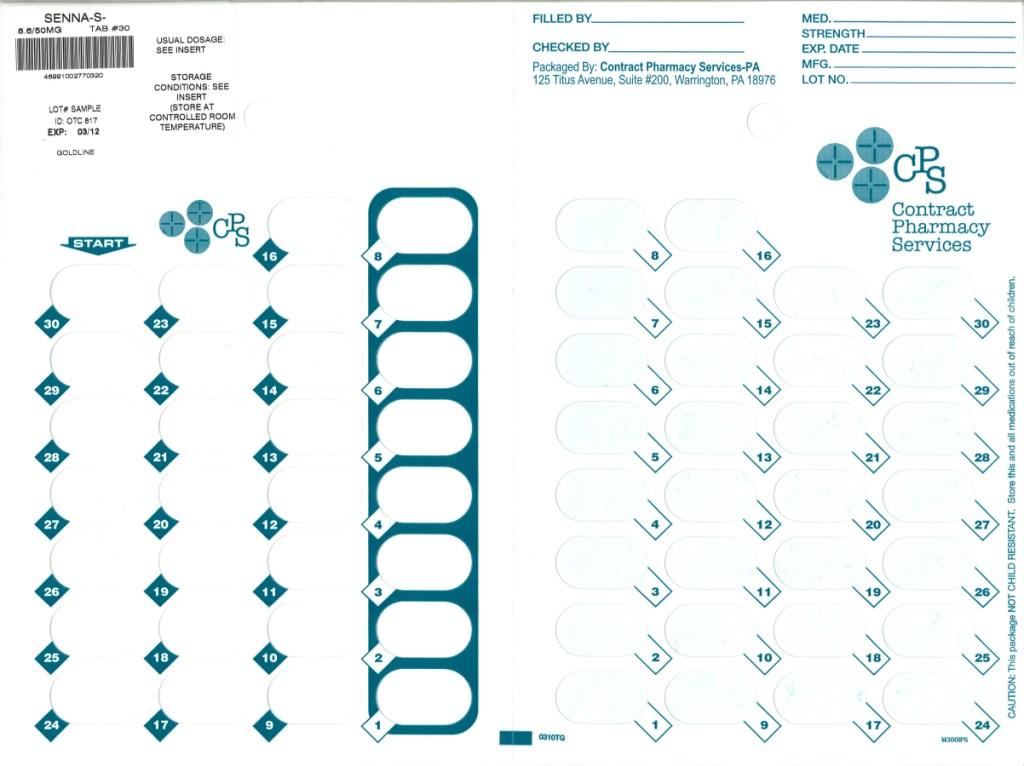
Label: SENNA-S- docusate sodium and sennosides tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 67046-670-30 - Packager: Contract Pharmacy Services-PA
- This is a repackaged label.
- Source NDC Code(s): 0182-1113
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 30, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS
- ACTIVE INGREDIENT (IN EACH TABLET)
- PURPOSE
- USES
-
WARNINGS
Do not use
- this product if you are presently taking mineral oil, unless directed by a doctor
- laxative products for more than 1 week unless directed by a doctor
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- a sudden change in bowel habits that persists over 2 weeks
-
DIRECTIONS
- take preferably at bedtime or as directed by a doctor
- if you do not have a comfortable bowel movement by the second day, increase dose by one tablet (do not exceed maximum dosage); or decrease dose until you are comfortable
-
age starting dose maximum dose adults and children 12 years and over 2 tablets once a day 4 tablets twice a day children 6 to under 12 years 1 tablet once a day 2 tablets twice a day children 2 to under 6 years 1/2 tablet once a day 1 tablet twice a day children under 2 years ask a doctor ask a doctor
- OTHER INFORMATION
-
INACTIVE INGREDIENTS
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate dihydrate, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, sodium benzoate, stearic acid, tapioca starch1, tartaric acid1, titanium dioxide
1may contain these ingredients
- QUESTIONS OR COMMENTS?
- Package Label
-
INGREDIENTS AND APPEARANCE
SENNA-S
docusate sodium and sennosides tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67046-670(NDC:0182-1113) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSE (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) SODIUM BENZOATE (UNII: OJ245FE5EU) STEARIC ACID (UNII: 4ELV7Z65AP) STARCH, TAPIOCA (UNII: 24SC3U704I) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ALUMINUM OXIDE (UNII: LMI26O6933) Product Characteristics Color YELLOW Score no score Shape ROUND Size 8mm Flavor Imprint Code 8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67046-670-30 30 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 08/17/2010 Labeler - Contract Pharmacy Services-PA (945429777)