GUAIFENESIN- guaifenesin tablet
AvKARE, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
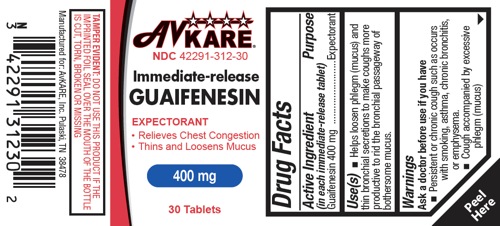
Drug Facts
Uses
- Helps loosen phlegm (mucus) and thin bronchial secretions to make coughs more productive to rid the bronchial passageway of bothersome mucus.
Ask a doctor before use if you have
-
Persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema.
- Cough accompanied by excessive phlegm (mucus)
Stop use and ask a doctor if
- cough lasts for more than 7 days, recurs, or is accompanied by fever, rash, or persistent headache. These could be signs of a serious condition.
Keep out of reach of children
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- adults and children 12 years of age and older, take 1 tablet every 4 hours with a full glass of water while symptoms persist.Do not exceed 6 doses in 24 hours.
- children under 12 years of age do not use.
Other information
- store at 25°C (77°F) excursions permitted between 15°-30°C (59°-86°F)
- keep in a dry place and do not expose to excessive heat
Inactive ingredients
Colloidal Silicon Dioxide, Magnesium Stearate, Maltodextrin, Microcrystalline Cellulose, Povidone, Silicon Dioxide, Sodium Starch Glycolate, Stearic Acid
Principal display panel
AvKARE
NDC 42291-312-30
Immediate-release
GUAIFENESIN
EXPECTORANT
• Relieves Chest Congestion
• Thins and Loosens Mucus
400 mg
30 Tablets
TAMPER EVIDENT: DO NOT USE THIS PRODUCT IF THE IMPRINTED FOIL SEAL OVER THE MOUTH OF THE BOTTLE IS CUT, TORN, BROKEN OR MISSING
Manufactured for: AvKARE, Inc. Pulaski, TN 38478
Lot:
Exp Date:
AV 05/12

| GUAIFENESIN
guaifenesin tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - AvKARE, Inc. (796560394) |