GAS RELIEF EXTRA STRENGTH- simethicone tablet, chewable
Rite Aid Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Rite Aid 44-608-bisected-Delisted
Directions
- chew or crush tablets completely before swallowing; do not swallow tablets whole
- adults: take 1 or 2 chewable tablets as needed after meals and at bedtime
- do not exceed 4 chewable tablets in 24 hours unless directed by a doctor
Other information
- each tablet contains: calcium 90 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- protect from moisture
- see end flap for expiration date and lot number
Inactive ingredients
D&C red #27 aluminum lake, D&C red #30 aluminum lake, dextrates hydrated, FD&C red #40 aluminum lake, flavor, silicon dioxide, sorbitol, starch, stearic acid, talc, tribasic calcium phosphate
Principal Display Panel
RITE
AID®
PHARMACY
*Compare to the active ingredient
in Gas-X® Extra Strength
EXTRA STRENGTH
gas relief
simethicone 125 mg
chewable tablets
antigas
cherry flavor
relieves:
gas pressure
bloating
discomfort
chew or crush tablets completely before swallowing.
ACTUAL SIZE
2 CHEWABLE TABLETS
125 mg EACH
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Novartis Consumer Health,
Inc., owner of the registered trademark Gas-X® Extra Strength.
50844 ORG011960824
DISTRIBUTED BY: RITE AID
30 HUNTER LANE, CAMP HILL, PA 17011
IF YOU'RE NOT SATISFIED,
WE'LL HAPPILY REFUND
YOUR MONEY.
44-608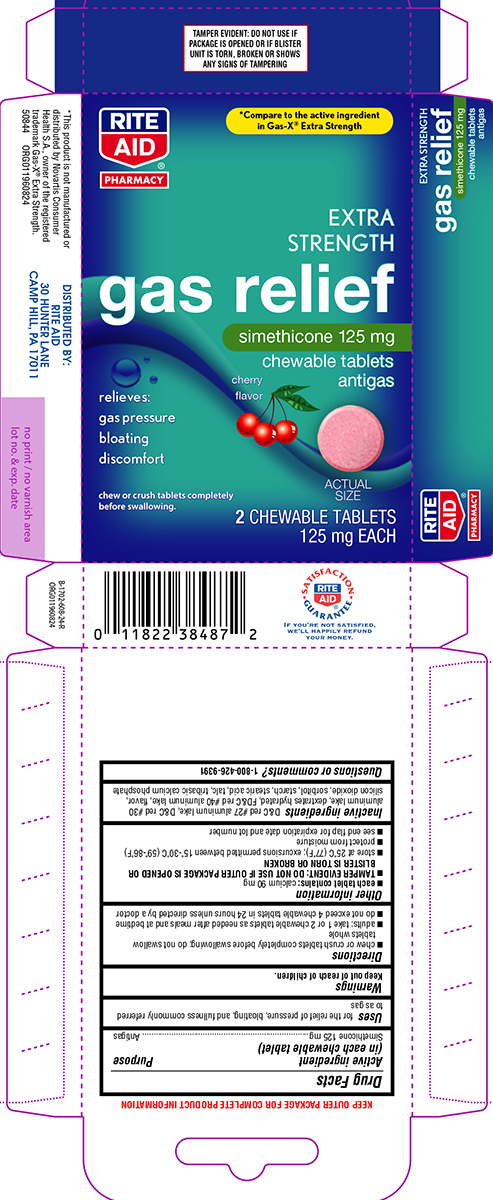
| GAS RELIEF
EXTRA STRENGTH
simethicone tablet, chewable |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Rite Aid Corporation (014578892) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(11822-0608) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(11822-0608) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | pack(11822-0608) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(11822-0608) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | pack(11822-0608) | |