AMBI ACNE SPOT TREATMENT- benzoyl peroxide cream
Valeant Pharmaceuticals North America LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
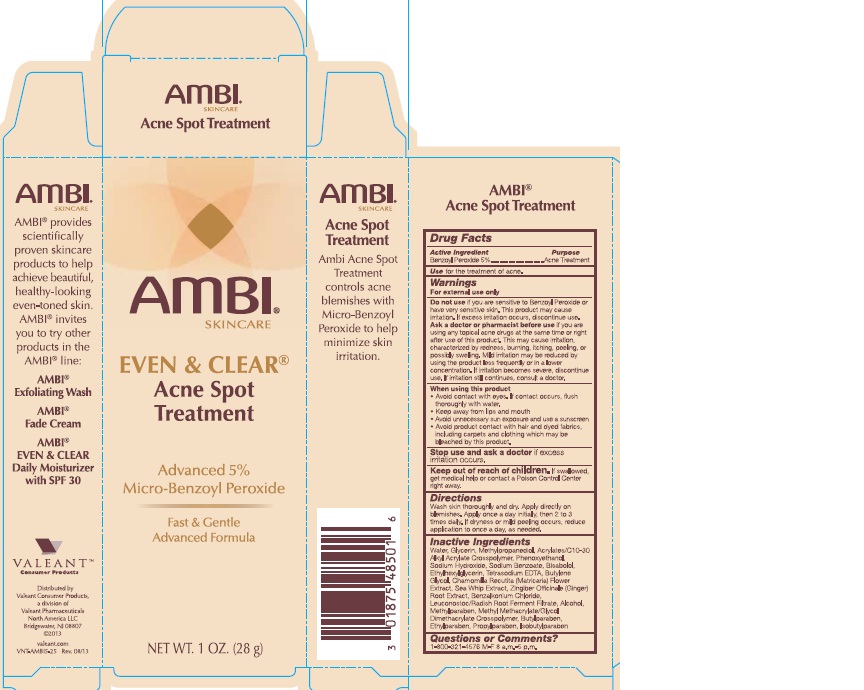
AMBI Even & Clear® Acne Spot Treatment
Warnings
For external use only
Do not use if you are sensitive to Benzoyl Peroxide or have very sensitive skin. This product may cause irritation. If excess irritation occurs, discontinue use.
Ask a doctor or pharmacist before use if you are using any topical acne drugs at the same time or right after use of this product. This may cause irritation, characterized by redness, burning, itching, peeling, or possibly swelling. Mild irritation may be reduced by using the product less frequently or in a lower concentration. If irritation becomes severe, discontinue use. If irritation still continues, consult a doctor.
When using this product
- •
- Avoid contact with eyes. If contact occurs, flush thoroughly with water.
- •
- Keep away from lips and mouth
- •
- Avoid unnecessary sun exposure and use a sunscreen
- •
- Avoid product contact with hair and dyed fabrics, including carpets and clothing which may be bleached by this product.
Directions
- •
- Wash skin thoroughly and dry.
- •
- Apply directly on blemishes.
- •
- Apply one a day intially, then 2 to 3 times daily. If dryness or mild peeling occurs, reduce application to once a day, as needed.
Inactive ingredients
Water, Glycerin, Methylpropanediol, Acryaltes/C10-30 Alkyl Acrylate Crosspolymer, Phenoxyethanol, Sodium Hydroxide, Sodium Benzoate, Bisabolol, Ethylhexylglycerin, Tetrasodium EDTA, Butylene Glycol, Chamomilla Recutita (Matricaria) Flower Extract, Sea Whip Extract, Zingiber Officinale (Ginger) Root Extract, Benzalkonium Chloride, Leuconostoc/Radish Root Ferment Filtrate, Alcohol, Methylparaben, Methyl Methacrylate/Glycol Dimethacrylate Crosspolymer, Butylparaben, Ethylparaben, Propylparaben, Isobutylparaben
| AMBI ACNE SPOT TREATMENT
benzoyl peroxide cream |
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Valeant Pharmaceuticals North America LLC (042230623) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Product Quest Mfg, LLC | 927768135 | MANUFACTURE(0187-5485) | |