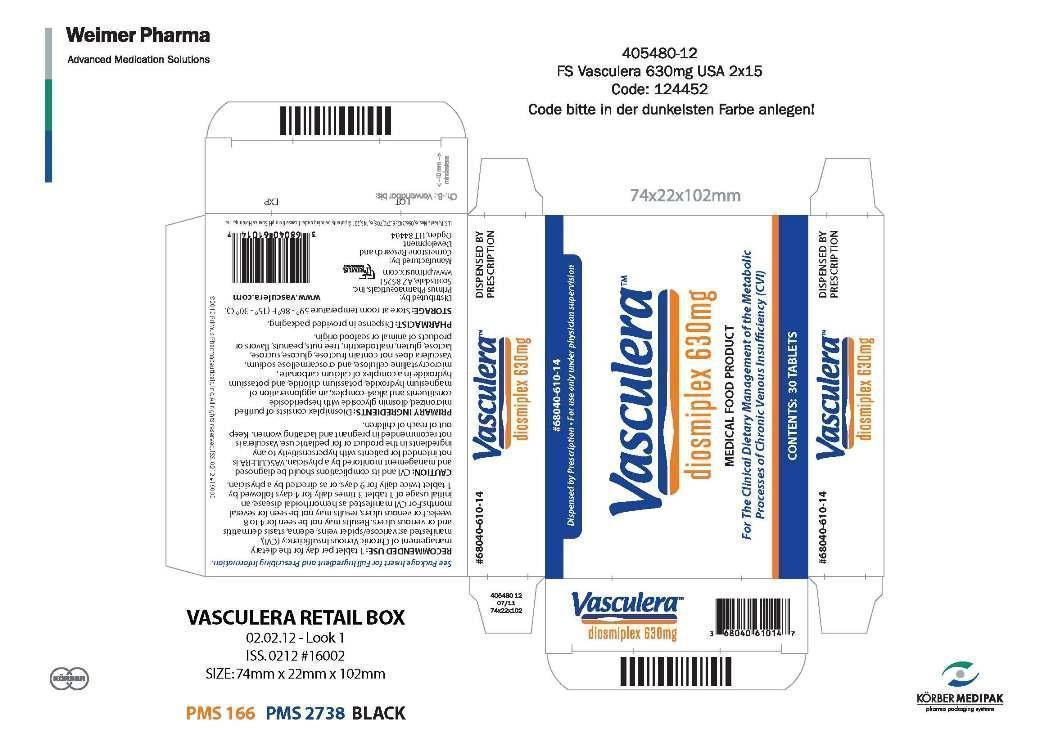
Label: VASCULERA- diosmiplex tablet
- NHRIC Code(s): 68040-610-14
- Packager: Primus Pharmaceuticals, Inc.
- Category: MEDICAL FOOD
- DEA Schedule: None
- Marketing Status: MEDICAL FOOD
Drug Label Information
Updated April 26, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
VASCULERA (diosmiplex) consists of a specially formulated proprietary blend of micronized, highly purified diosmin glycoside in combination with alkaline granules, alka4-complex. Diosmin glycoside manages venous inflammation, accumulation of polymorphonuclear leukocytes, platelets and other thrombotic components as well as edema, caused by a deterioration of venous vessel walls. Alka4-complex works by buffering stomach acid and managing blood pH to affect local metabolic acidosis in veins.
Diosmin Glycoside
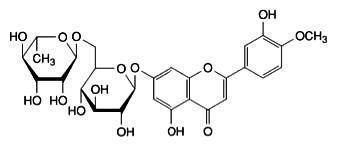
Each VASCULERA tablet contains 600 mg of diosmin glycoside (diosmin), a micronized, highly purified flavonoid fraction (from citrus) with hesperidoside constituents. In clinical trials, this level of intake has been shown to manage chronic venous insufficiency (CVI). Diosmin is chemically described as (7-[[6-O-(6-Deoxy-α-L-mannopyranosyl)-ß-D-glucopyranosyl]oxy]-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-1-benzopyran-4-one) with a molecular weight of 608.5. The empirical formula for diosmin is C28H32O15. The structural formula is:
Alka4-complex
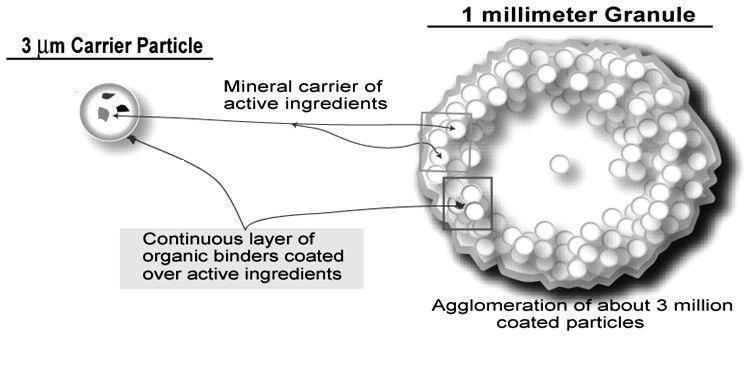
Each VASCULERA tablet contains 30 mg of a proprietary, structured, highly alkaline agglomeration of reactive magnesium hydroxide (Mg(OH)2, molecular weight 58.3), potassium chloride (KCl, molecular weight 74.6) and potassium hydroxide (KOH, molecular weight 56.1) held apart from each other in a matrix of calcium carbonate (CaCO3, molecular weight 100.1), microcrystalline cellulose and croscarmellose sodium. The agglomerated particle matrix has the following approximate characteristics:
Other Ingredients
VASCULERA contains the following other ingredients as fillers and excipients: microcrystalline cellulose, stearic acid, croscarmellose sodium, povidone, silicone dioxide, and water. Tablets do not contain fructose, glucose, sucrose, lactose, gluten, maltodextrin, tree nuts, peanuts, or flavors. VASCULERA is suitable for vegans.
Medical Food
The U.S. Congress defined “medical food” in the Orphan Drug Act and Amendments of 1988 as “a food which is formulated to be consumed or administered enterally under the supervision of a physician, and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” VASCULERA has been developed, manufactured and labeled in accordance with the statutory definition of a medical food. VASCULERA is to be used only under a physician’s supervision.
Generally Recognized As Safe
The ingredients in VASCULERA are Generally Recognized as Safe (GRAS). This is the statutory U.S. safety standard that the U.S. Food and Drug Administration (FDA) requires of all ingredients added to food products. The standard for an ingredient to achieve GRAS status requires technical demonstration of non-toxicity and safety, general recognition of safety through widespread usage and agreement of that safety by experts in the field. -
CHRONIC VENOUS INSUFFICIENCY (CVI)
Chronic venous insufficiency is caused by venous hypertension (VH). VH, in turn, aggravates these metabolic imbalances leading to a self perpetuating cycle of further metabolic changes, including venous acidosis. These changes promote further inflammation in vascular tissue leading to edema, skin damage and possible ulceration and deep vein thrombosis (DVT). Varicose veins and hemorrhoids are also part of the spectrum of CVI disorders. -
CLINICAL PHARMACOLOGY
Mechanism of Action
VASCULERA acts by restoring toward normal the metabolic aspects of CVI including modulation of venous tone and capillary resistance, management of lymphatic drainage, and inflammation in the microcirculation. Preclinical and clinical data suggest that the diosmin glycoside in VASCULERA manages venous tone by increasing smooth muscle contractibility. In addition, diosmin has been shown to decrease edema by increasing lymphatic contractility and drainage. Finally, diosmin manages the oxidative and inflammatory factors induced by VH. Alka4-complex has been shown in preclinical and clinical studies to act as an acid buffering agent in the gut and to counteract the effects of increased acid production systemically.
Diosmin Glycoside
The diosmin in VASCULERA has been shown in both cell and animal models to enhance venous tone by prolonging the post-synaptic response to norepinephrine (NE) and increasing the sensitivity of vascular smooth muscle to NE and calcium, even under conditions of acidosis. Diosmin inhibits the NE degrading enzyme catechol-O-methyltransferase (COMT). Diosmin has also been shown to manage lymphatic drainage and decrease edema by increasing lymphatic vessel contraction frequency and amplitude. Venous hypertension induces production of inflammatory factors such as prostaglandins and leukotrienes, metalloproteinaises, cytokines, adhesion molecules, and VEGF. Localization of inflammatory cells generates high levels of tissue destructive reactive oxygen species (ROS). Diosmin acts as a scavenger of ROS, inhibits 5-lipoxygenase and the production of prostaglandins E2 and thromboxane B2, which may help to protect endothelial cells from damage associated with inlammation and acute oxidative stress.
Alka4-complex
Clinical studies have shown that alka4-complex in VASCULERA can reduce acidosis under extreme exercise loads. The specially agglomerated matrix of alka4-complex has been shown to resist neutralization by acid solutions. This ability to survive the acid environment of the stomach and to be taken up in the intestine may account for its effect on blood pH during exercise. Alka4-complex may counter the local acidosis produced in CVI and work synergystically with diosmin to manage venous inflammtion. In addition, the alka4-complex has been shown to reduce the acid producing effects of certain foods by buffering the acidity in the gastrointestinal tract, while allowing a sufficient portion of the complex to survive in the stomach, and is absorbed in the small intestine thereby being available to manage mild acidosis.
-
METABOLISM
Diosmin glycoside
Diosmin is hydrolyzed to its aglycone form, diosmetin, by intestinal microorganisms and enzymes in the intestinal lumen prior to absorption. Once it reaches the liver, diosmetin is extensively glucuronidated before entering the systemic circulation. Diosmetin may also be reduced to its corresponding flavanone aglycone, hesperetin, by intestinal bacteria in second pass metabolism. No diosmin is detected in the serum in animals or humans. Only the active form diosmetin, is detected after ß-glucoronidase digestion. The phenolic acid products of diosmetin degradation are excreted in the urine, while unabsorbed/unmetabolized diosmin and diosmetin are excreted in the feces.
Alka4-complex
In contrast to sodium bicarbonate, alka4-complex has been shown in acid dissolution studies to release alkalizing salts in two gradual steps. Starting at a pH of 2, alka4-complex takes approximately 15 min to raise the solution pH to 6 whereas this occurs in seconds with bicarbonate. A further increase to a pH of 7 occurs over the next 60-90 min period. This biphasic, slow release characteristic gives alka4-complex seven times more pH neutralizing capacity compared to the same concentration of sodium bicarbonate. The molecular protection afforded by the proprietary matrix allows the alka4-complex to enter the intestinal tract where it is absorbed. Compared to bicarbonate ions, hydroxyl ions present in alka4-complex have been shown to be better absorbed by the intestional lumen.
-
CLINICAL EXPERIENCE
Hepatic and Renal Effects
In multiple clinical studies, the effects of diosmin in VASCULERA on blood chemistry as well as hepatic and renal function were compared to placebo in healthy subjects and patients with CVI. No changes were noted in most studies with the exception of statistically significant decreases in creatinine levels in subjects taking diosmin in several clinical trials.
Cardiovascular Safety
No cardio-vascular adverse effects of the diosmin in VASCULERA have been observed in clinical trials. Small decreases in both systolic and diastolic blood pressure have been noted in several studies for CVI. When used as a pretreatment for coronary artery bypass grafting, troponin I and lactate dehydrogenase levels were significantly lower in the post-cardiopulmonary bypass period.
Blood Glucose, Insulin and Insulin Resistance
No effects of diosmin or alka4-complex have been noted in clinical studies on blood glucose or insulin levels as well as insulin resistance.
- DRUG INTERACTIONS
-
TOXICITY
Diosmin glycoside
In chronic and acute toxicity studies in mice, rats and primates in single oral doses up to 3,000 mg/kg body weight/day or as repeated oral doses up to 583 mg/kg body weight/day (representing approximately 180 and 35 times respectively, the recommended daily human dose) for up to 26 weeks, diosmin showed no effect on body weight, routine blood hematology or chemistry parameters. In animal studies, macroscopic and histologic examination of all major organ systems revealed no pathological changes. Two teratogenicity studies using 50 and 100 mg/kg/day of diosmin against placebo show no differences on fetal formation, number per litter, weight gain, or developmental milestones. Finally, acute dosing of up to 5000 mg/day in humans for 4 days produced no toxic effects.
Effects on Reproductive Tissues
The diosmin in VASCULERA showed no impairment of reproductive function or effects on reproductive tissue in the rat after administration of an oral
dose representing 37 times the recommended daily human dose (600 mg/day). Adverse effects regarding fertility, embryotoxicity and perinatal and
postnatal development of the generation born from treated animals were not observed. Diosmin in VASCULERA was tested in human clinical trials
for hemorrhoids during the third trimester of pregnancy and 4 weeks after birth. Administration of diosmin at 900 mg per day did not affect
pregnancy, fetal development, birth weight or infant growth and feeding.
Alka4-complex
Single doses of alka4-complex granules (0.5, 2 and 5 mg/kg) were administered by oral gavage on day 1 to male and female 6-7 week old Sprague-Dawley rats. Body weight, behavior and food intake were assessed daily showing no effect over a 15 day period. On day 15, animals were sacrificed and gross organ pathology assessed. The No Observable Adverse Effect Level (NOAEL) of alka4-complex granules administered in a single oral dose was 5 g/kg, based on the parameters evaluated, and the maximum tolerated single oral dose is greater than 5 g/kg as shown in this animal study.
- SPECIAL POPULATIONS
-
INDICATIONS AND USAGE
Indications
VASCULERA is indicated for the clinical dietary management of the metabolic processes associated with chronic venous insufficiency (CVI). Conditions which occur due to CVI include varicose veins, spider veins, edema, deep vein thrombosis, stasis dermatitis, venous ulcers and hemorrhoids. Symptoms of CVI include heavy leg syndrome (dull aching discomfort, heaviness, cramping, itching and tingling in legs), pain that gets worse when standing, pain that gets better when legs are raised, swelling of the legs (edema), redness of the legs and ankles, skin color changes around the ankles and thickening of the skin on the legs and ankles (stasis dermatitis). VASCULERA must be used under the supervision of a physician.
Usage
VASCULERA should be taken as directed by a physician. See Dosage and Administration for additional information.
Interactions with Food
VASCULERA can be taken with or without food.
-
PRECAUTIONS AND CONTRAINDICATIONS
General
CVI and its complications should be diagnosed and management monitored by a physician.
Hypersensitivity
VASCULERA is contraindicated for anyone having a hypersensitivity to any ingredient in the product. See “Description and Other Ingredients” for a full list of ingredients.
Patients with Cancer
Diosmin (up to 900 mg/day) has been administered to a small number of breast cancer patients who were experiencing lymphedema following surgical and nodal irradiation treatment with resultant reduction of arm edema but no effect on the cancer. Animal studies and more than 20 years of clinical use in Europe have not found any evidence of carcinogenicity or mutagenicity when the components of Vasculera are used as recommended. As a precaution, VASCULERA (diosmiplex) is not recommended for patients with a history of cancer since no specific studies have been performed in this population.
Pregnancy
Animal studies suggest that diosmin does not produce developmental abnormalities when consumed during pregnancy. A small number of pregnant women have consumed diosmin (up to 900 mg/day) in the third trimester and the first 4 weeks post-partum with no effect on the fetus or baby. Effects in infants of nursing mothers are uncertain for chronic dosing. Therefore, VASCULERA (diosmiplex) is not recommended in pregnant and lactating women. Women capable of becoming pregnant should consult with their physician before taking VASCULERA.
-
ADVERSE EVENTS
Diosmin glycoside
Many clinical trials have been conducted in adults with various manifestations of CVI to assess the efficacy of orally administered diosmin in doses ranging from 400 to 5,000 mg/day for up to a year. No serious adverse events were reported in any of the studies. Commonly reported adverse events included gastrointestinal disturbances and headaches; these were generally mild in severity and did not usually result in patients discontinuing participation in the study. The following adverse events (and approximate percentages) were reported in clinical trials but their frequency did not differ from placebo: rash (1%), cramping in lower limb (2%), phlebitis (2%), venous thrombosis (4%), and skin changes around existing ulcer, swelling of the extremities and body rash (1.6%). Dyspepsia, or non-specific mild stomach upset, occurred in up to 7% of subjects taking diosmin at various doses and was seen with approximately twice the frequency seen in the placebo groups. Rare adverse events include inguinal pain, cystitis, asthenia, metrorrhagia and menometrorrhagia. In clinical trials, the incidence of adverse events in elderly populations (≥70 years of age) was not significantly different from that in younger populations nor were adverse events higher in patients with concomitant hypertension, atherosclerosis, diabetes, neurologic/psychiatric disease or alcoholism.
Alka4-complex
The case histories for 18 subjects who consumed ~3 g/day of alka4-complex for 18 months were analyzed and found to contain no reports of adverse events.
-
PHARMACOKINETICS
Diosmin glycoside
The pharmacokinetic parameters of diosmin have been studied in humans as well as several animal species (i.e., rats, dogs, rabbits, and monkeys). The pharmacokinetics of diosmin administered to 5 healthy subjects (2 men and 3 women), 20 to 45 years of age, at a single oral dose of ~600 mg was determined. Blood samples were taken at 0.5, 1, 2, 4, 8, 12, 24, and 48 hours after administration. Urine was collected at baseline, between 0 and 24 hours, and 24 and 48 hours after administration. All samples were subjected to incubation with ß-glucuronidase prior to assessment. No diosmin was detected in the plasma (detection limit of 20 ng/mL). Only diosmetin, the aglycone of diosmin, was found in the plasma, with a peak concentration (Tmax) of 400 ng/mL after 1 hour. Plasma levels decreased slowly after 2 hours, constantly after 24 hours, and were still detectable after 48 hours. The serum half-life (t1/2) among all subjects was 31.5 ± 8.6 hours. The pharmacokinetic parameters of this study are shown below:
Parameters Mean +/- SD Cmax (ng/mL)
417 +/- 94.1
T1/2 (h)
31.5 +/- 8.6
MRT (h)
36.6 +/- 9.9
Parameters Mean +/- SD AUC (0-48hr) ng/mL.h
5617.1 +/- 1518.4
TCL (L/h)
1.32 +/- 0.42
Vd (L)1
62.1 +/- 7.9
Abbreviations: AUC, area under the curve; Cmax, maximum concentration; MRT, mean residence time; T1/2, half-life; TCL, total clearance; Vd, volume of distribution.
1 Total clearance and volume of distribution were computed assuming complete bioavailability.
-
CLINICAL STUDIES
Dietary Management of Chronic Venous Insufficiency
In addition to more than 2 decades of clinical use in multiple countries, more than 40 clinical trials comprising >15,000 patients have been published. In studies using clinical endpoints of patient reported symptoms, edema and quality of life, diosmin consistently demonstrated 30-60% superiority to placebo or standard of care. The largest of the published CVI studies, a 2-year trial, enrolled 5,052 subjects in 23 countries, all of whom had CVI symptoms. Of those enrolled, 43% had reflux demonstrable with Doppler examination. All subjects were in CEAP (Clinical signs, Etiology, Anatomic distribution, and Pathological dysfunction) class 0-4 corresponding to mild-moderate disease without ulceration. The female/male ratio was 4/1, mean age 45 years and most were physically active. Leg heaviness was the most frequent complaint at baseline followed by aching, swelling and nocturnal cramping. CEAP class correlated with age. After 6 months, there was a significant reduction in CEAP class, edema and quality of life (QOL) score (p=0.0001 for all) irrespective of whether or not reflux was demonstrable at baseline. At the end of 6 months, 79% of subjects and 83% of investigators rated the effectiveness of diosmin as good or excellent irrespective of whether or not subjects regularly wore compression stockings. Studies using objective endpoints such as venous reflux, venous and lymphatic flow measured by techniques such as scintographic Doppler, plethysmography and venous pressure have shown similar statistically significant improvement in all parameters, generally in the range of 20-40% better than placebo. Other studies have confirmed the advantage of the micronized form of diosmin over the unmicronized form with regard to reduction in edema and clinical symptoms.
Dietary Management of Hemorrhoids
Multiple studies involving several hundred patients have been reported. These have routinely shown that diosmin reduces symptoms of discomfort, swelling, inflammation and time to resolution when comapred to standard of care. In general, resolution time of acute flares is shortened from 8 days to 4-5 days. A double-blind, placebo-controlled study (n=120) showed statistically better management of pain, pruritis, discharge, edema, erythema, and bleeding on examination. One study (n=351) compared the effacacy of combining infrared photocoagulation (IRP) and diosmin vs each therapy used alone on bleeding cessation in patients with grades I, II, and III acute internal hemorrhoids. The percentage of patients with no bleeding after 5 days of therapy was higher in the combined group (75%) compared with only diosmin (60%) or with IRP alone (56%). Patients with grades I and II hemorrhoids responded significantly better (83% and 62%, respectively) to either therapy than those with grade III hemorrhoids (23%). Patients experiencing an acute hemorrhoidal episodes for less then 48 h were enrolled in a randomized, double-blind, placebo-controlled study. One group received diosmin (n=49) and the other a placebo (n=41). During the 7 day administration, there was a significant difference in favor of diosmin in the dietary management of discomfort, edema and bleeding. A study has compared the results of the conservative management of hemorrhoids between diosmin and sclerotherapy (SCL), in terms of the subjective and objective outcome of patients after a follow-up period of 2 years. Average Symptoms Score (ASS) and Average Anoscopy Scores (AAS) decreased in both of the groups in the first three visits. At the end of the second visit, ASS and AAS significantly declined in the diosmin group. In addition, ASS and AAS decreased to the nadir level in both groups at the end of the 26th week. In another study of 100 patients with endoscopically confirmed hemorrhoids, patients receiving diosmin showed an improvement in discomfort, anal discharge, proctitis and time to resolution compared to placebo (p<0.001).
Dietary Management of Venous Ulcers
More than 20 clinical trials involving >1000 patients have been published. In general, these compare treatment using diosmin with standard of care (compression and dressing) and tend to be open label. Most show improvement within 2-4 months. Diosmin uniformly increased the number of resolved ulcers and the time to resolution for ulcers <10 cm but had little benefit for ulcers >10 cm. Diosmin also decreased the length, but not the number, of hospital stays. Several randomized trials (n=107-150) compared ulcer resolution and time on diosmin plus standard of care (SOC) vs SOC alone or with placebo. In each case, the patients administered diosmin had a better outcome than the comparators. Ulcer resolution varied with the size of the ulcer with about 70% resolution of ulcers <3 cm vs 50% in the SOC group while ulcers between 3-6 cm showed resolution rates of 60% and 30%, respectively. A meta-analysis of seven comparable prospective studies (n=723) found a significant difference in overall ulcer resolution between diosmin administered groups and controls at 2 months (p=0.0088). By 6 months, the difference, while still significant, had narrowed to 61% vs 48% for placebo (OR=2.02,p=0.035), the difference representing the fact that persistent ulcers tend to be larger and deeper and have more vascular compromise. A meta-analysis of 5 European trials (n=723) found that for ulcers between 5-10 cm, diosmin, together with SOC, significantly reduced the time to clearance, the percent of ulcers resolved and was especially valuable for ulcers that had been present for 6-12 months.
Concomitant Use
No evidence of drug incompatibility, drug interaction, or photosensitizing action of diosmin was observed when combined with drugs used to treat a wide variety of clinical disorders. Although grapefruit juice is known to affect metabolism of many drugs by the CYP450 enzyme system, this effect has not been ascribed specifically to diosmin or hesperidin. Hesperidin, the flavonoid from which diosmin is derived, is thought to increase bioavailability of diltiazem by a combination of CYP450 3A4 inhibition and increased enteric absorption. In single dose PK studies, diosmin significantly delayed the T1/2 and increased the AUC of chlorzoxazone, possibly by interfering with CYP2E1 metabolism. Similar effects have been noted with diclofenac and metronidiazole, both of which are metabolized by the CYP2C9 enzyme system.
-
OVER USAGE
Diosmin glycoside
There are no known cases of over usage of the diosmiplex in VASCULERA. Animal studies have shown that consuming the equivalent of 56 VASCULERA tablets containing 600 mg of diosmin or 30 mg of alka4-complex at one time did not produce adverse events. However, as in most over usage situations, symptoms following an over usage of VASCULERA could vary according to the patient. If an over usage occurs, patients should be managed by systematic and supportive care as soon as possible following product consumption.
-
PRODUCT ADMINISTRATION
The recommended intake of VASCULERA (diosmiplex) is 1 tablet per day for the dietary management of Chronic Venous Insufficiency (CVI), manifested as: varicose/spider veins, edema, stasis dermatitis and or venous ulcers. Results may not be seen for 4 to 8 weeks. For venous ulcers, results may not be seen for several months.
For CVI manifested as hemorrhoidal disease, the recommended initial intake is 1 tablet 3 times daily for 4 days followed by 1 tablet twice daily for 9 days, or as directed by a physician. For hemorrhoidal discomfort persisting after resolution of the acute flare or for recurrent hemorrhoidal flares irrespective of persisting discomfort, a maintenance intake of 600 mg of Vasculera daily is recommended.
-
HOW SUPPLIED
VASCULERA is a grayish-yellow oblong tablet with white flecks on the surface.
The tablets are packaged in unit-of-use blister packages :
68040-610-14 contents 30 tablets per carton (30-day supply)
68040-610-10 contents 15 tablets per carton (15-day supply sample)
68040-610-25 contents 3 tablets per carton (3-day supply sample)Storage
Store at room temperature 59° – 86°F (15° – 30°C). Protect from light and moisture. Store tablets in original carton until usage. Keep out of reach of children.
- Vasculera Carton
-
INGREDIENTS AND APPEARANCE
VASCULERA
diosmiplex tabletProduct Information Product Type MEDICAL FOOD Item Code (Source) NHRIC:68040-610 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIOSMIN (UNII: 7QM776WJ5N) (DIOSMIN - UNII:7QM776WJ5N) DIOSMIN 600 mg CALCIUM MAGNESIUM POTASSIUM CARBONATE CHLORIDE HYDROXIDE (UNII: Z7R65IFU98) (CALCIUM MAGNESIUM POTASSIUM CARBONATE CHLORIDE HYDROXIDE - UNII:Z7R65IFU98) CALCIUM MAGNESIUM POTASSIUM CARBONATE CHLORIDE HYDROXIDE 30 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POVIDONE (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) WATER (UNII: 059QF0KO0R) Product Characteristics Color brown (Greyish-yellow) Score no score Shape OVAL (Symmetrical oblong tablet) Size 16mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:68040-610-14 30 in 1 CARTON Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date medical food 04/02/2012 Labeler - Primus Pharmaceuticals, Inc. (130834745) Establishment Name Address ID/FEI Business Operations Cornerstone Research & Development, Inc. 825319767 manufacture