Label: MAYBELLINE NEW YORK FIT ME FOUNDATION DEWY PLUS SMOOTH SPF 18 SUNSCREEN- octinoxate lotion
- NDC Code(s): 49967-969-01
- Packager: L'Oreal USA Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
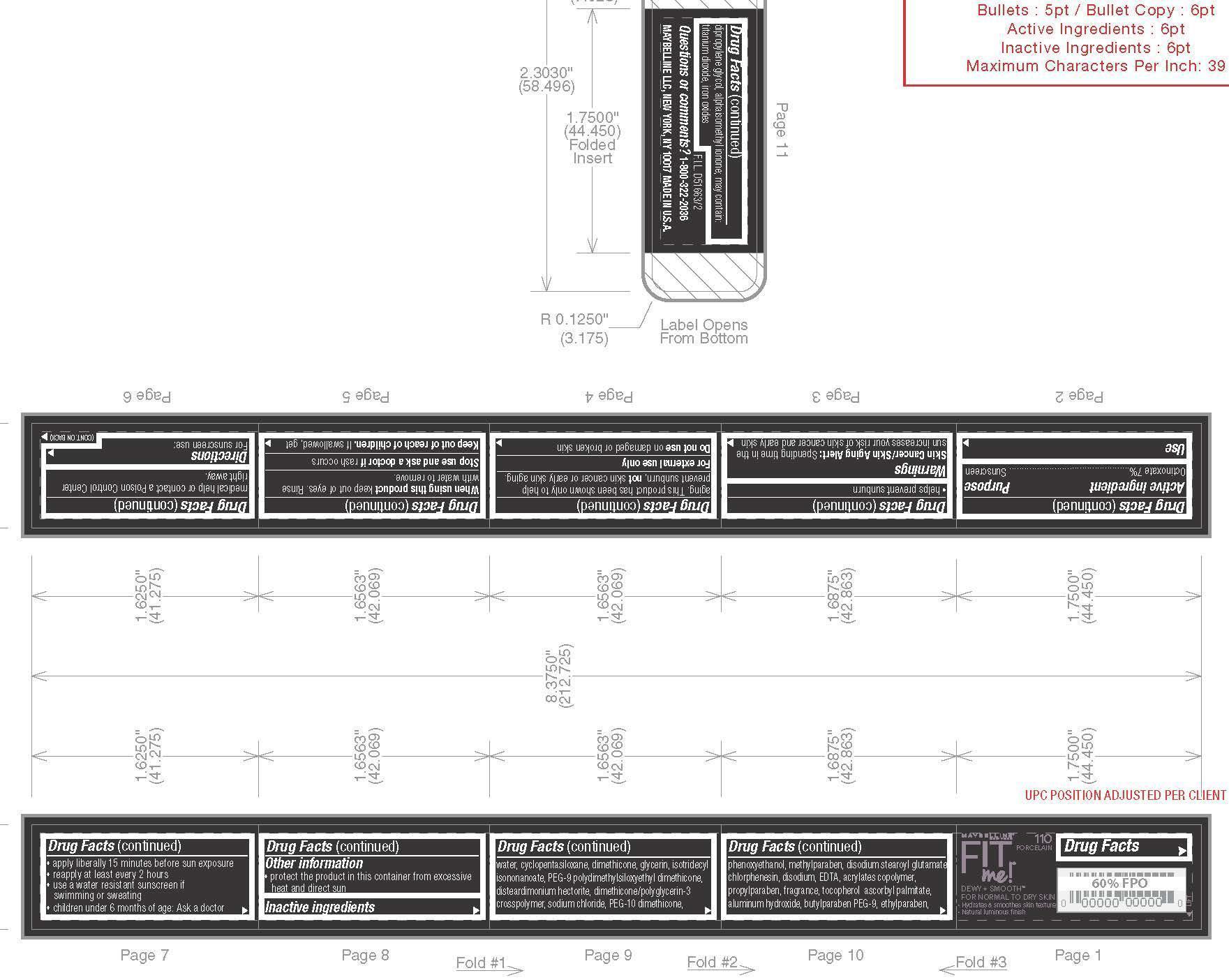
- Active ingredient
- Purpose
- Use
- Skin Cancer/Skin Aging Alert:
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
- Directions
- Other information
-
Inactive ingredients
water, cyclopentasiloxane, dimethicone, glycerin, isotridecyl isononanoate, PEG-9 polydimethylsiloxyethyl dimethicone, disteardimonium hectorite, dimethicone/polyglycerin-3 crosspolymner, sodium chloride, PEG-10 dimethicone, phenoxyethanol, methylparaben, disodium stearoyl glutamate chlorphenesin, disodium, EDTA, acrylates copolymer, propylparaben, fragrance, tocopherol ascorbyl palmitate, aluminum hydroxide, butylparaben PEG-9, ethylparaben, dipropylene glycol, alphaisomethyl ionone; may contain: titanium dioxide, iron oxides
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MAYBELLINE NEW YORK FIT ME FOUNDATION DEWY PLUS SMOOTH SPF 18 SUNSCREEN
octinoxate lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49967-969 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 70 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49967-969-01 30 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 09/01/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 09/01/2010 Labeler - L'Oreal USA Products Inc (002136794) Establishment Name Address ID/FEI Business Operations L'Oreal USA, Inc. 624244349 manufacture(49967-969)