SORE THROAT- phenol spray
HyVee Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Hy-Vee, Inc. Sore Throat Spray Drug Facts
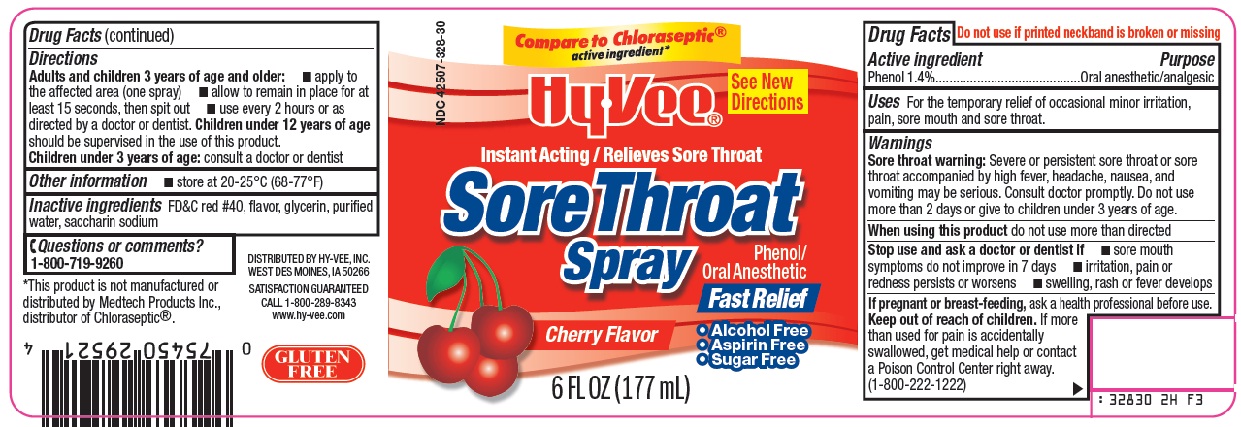
Warnings
Sore throat warning: Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea, or vomiting may be serious. Consult doctor promptly. Do not use more than 2 days or give to children under 3 years of age.
Stop use and ask a doctor or dentist if
- •
- sore mouth symptoms do not improve in 7 days
- •
- irritation, pain, or redness persists or worsens
- •
- swelling, rash or fever develops
Keep out of reach of children.
If more than used for pain is accidentally swallowed, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Directions
Adults and children 3 years of age and older:
- •
- apply to the affected are (one spray)
- •
- allow to remain in place for at least 15 seconds, then spit out
- •
- use every 2 hours or as directed by a doctor or dentist.
Children under 12 years of age should be supervised in the use of this product.
Children under 3 years of age: consult a doctor or dentist
| SORE THROAT
phenol spray |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - HyVee Inc (006925671) |