8 HOUR PAIN RELIEF- acetaminophen tablet, film coated, extended release
HyVee Inc
----------
Hy-Vee, Inc. 8 Hour Pain Relief Drug Facts
Uses
- •
- temporarily relieves minor aches and pains due to:
- •
- muscular aches
- •
- backache
- •
- headache
- •
- minor pain of arthritis
- •
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- •
- more than 6 caplets in 24 hours, which is the maximum daily amount
- •
- with other drugs containing acetaminophen
- •
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- •
- skin reddening
- •
- blisters
- •
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Directions
- •
- do not take more than directed (see overdose warning)
|
adults and children 12 years and over |
|
|
children under 12 years |
|
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, FD&C red #40 aluminum lake, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polysorbate 80, povidone, pregelatinized starch, stearic acid, titanium dioxide
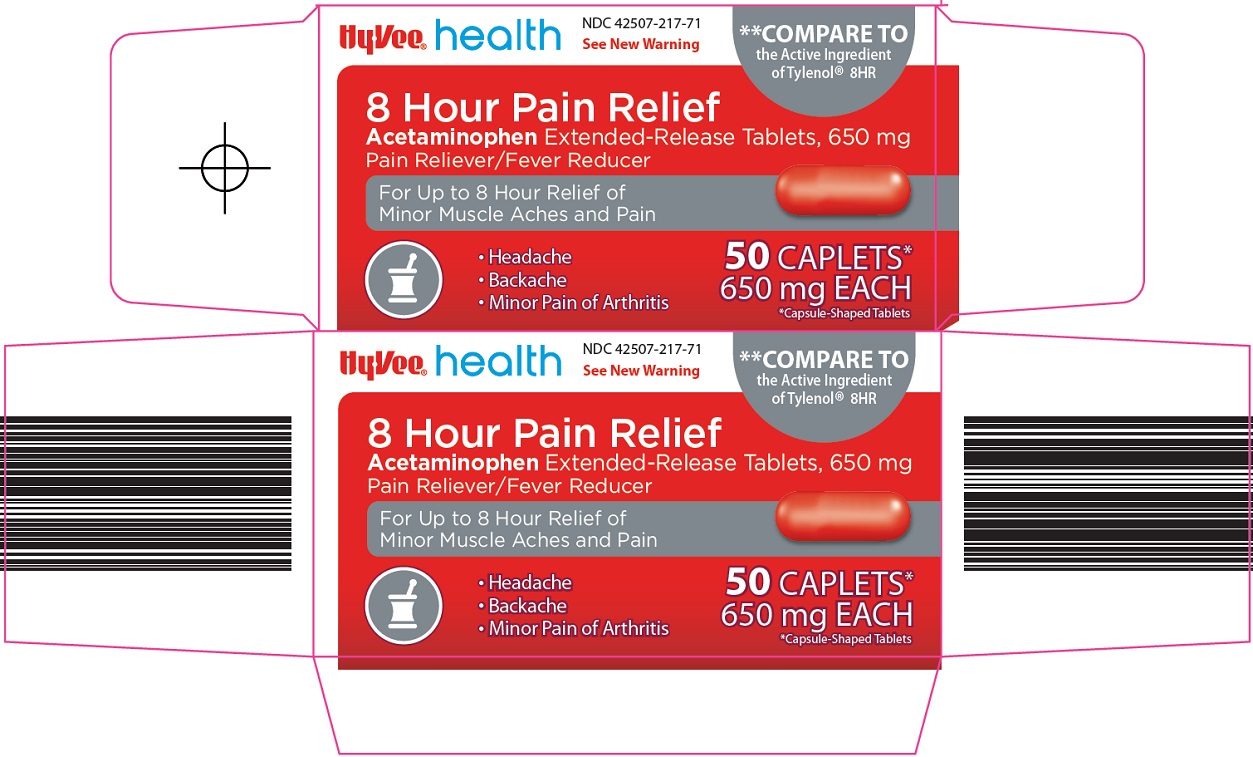
Principal Display Panel
See New Warning
COMPARE TO the Active Ingredient of Tylenol® 8HR
8 Hour Pain Relief
Acetaminophen Extended-Release Tablets, 650 mg
Pain Reliever / Fever Reducer
For Up to 8 Hour Relief of Minor Muscle Aches and Pain
Headache
Backache
Minor Pain of Arthritis
50 CAPLETS*
650 mg EACH
*Capsule-Shaped Tablets
| 8 HOUR PAIN RELIEF
acetaminophen tablet, film coated, extended release |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - HyVee Inc (006925671) |