ROBAXIN- methocarbamol tablet
REMEDYREPACK INC.
----------
DESCRIPTION
robaxin(methocarbamol tablets, USP), a carbamate derivative of guaifenesin, is a central nervous system (CNS) depressant with sedative and musculoskeletal relaxant properties.
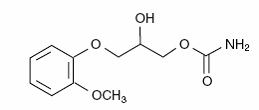
The chemical name of methocarbamol is 3-(2-methoxyphenoxy)-1,2-propanediol 1-carbamate and has the empirical formula C11H15NO5. Its molecular weight is 241.24. The structural formula is shown below.
Methocarbamol is a white powder, sparingly soluble in water and chloroform, soluble in alcohol (only with heating) and propylene glycol, and insoluble in benzene and n-hexane.
robaxinis available as a light orange, round, film-coated tablet containing 500 mg of methocarbamol, USP for oral administration. The inactive ingredients present are corn starch, FD&C Yellow 6, hydroxypropyl cellulose, hypromellose, magnesium stearate, polysorbate 20, povidone, propylene glycol, saccharin sodium, sodium lauryl sulfate, sodium starch glycolate, stearic acid, titanium dioxide.
robaxinis available as an orange capsule-shaped, film-coated tablet containing 750 mg of methocarbamol, USP for oral administration. In addition to the inactive ingredients present in robaxinrobaxinalso contains D&C Yellow 10.
CLINICAL PHARMACOLOGY
The mechanism of action of methocarbamol in humans has not been established, but may be due to general central nervous system (CNS) depression. It has no direct action on the contractile mechanism of striated muscle, the motor end plate or the nerve fiber.
Pharmacokinetics
In healthy volunteers, the plasma clearance of methocarbamol ranges between 0.20 and 0.80 L/h/kg, the mean plasma elimination half-life ranges between 1 and 2 hours, and the plasma protein binding ranges between 46% and 50%.
Methocarbamol is metabolized via dealkylation and hydroxylation. Conjugation of methocarbamol also is likely. Essentially all methocarbamol metabolites are eliminated in the urine. Small amounts of unchanged methocarbamol also are excreted in the urine.
Special populations
Elderly
The mean (SD) elimination half-life of methocarbamol in elderly healthy volunteers (mean (SD) age, 69 (4) years) was slightly prolonged compared to a younger (mean (SD) age, 53.3 (8.8) years), healthy population (1.5 (0.4) hours versus 1.1 (hours, respectively). The fraction of bound methocarbamol was slightly decreased in the elderly versus younger volunteers (41 to 43% versus 46 to 50%, respectively).
Renally impaired
The clearance of methocarbamol in 8 renally-impaired patients on maintenance hemodialysis was reduced about 40% compared to 17 normal subjects, although the mean (SD) elimination half-life in these two groups was similar: 1.2 (0.6) versus 1.1 (hours, respectively.
Hepatically impaired
In 8 patients with cirrhosis secondary to alcohol abuse, the mean total clearance of methocarbamol was reduced approximately 70% compared to that obtained in 8 age- and weight-matched normal subjects. The mean (SD) elimination half-life in the cirrhotic patients and the normal subjects was 3.38 (1.62) hours and 1.11 (0.27) hours, respectively. The percent of methocarbamol bound to plasma proteins was decreased to approximately 40 to 45% compared to 46 to 50% in the normal subjects.
INDICATIONS & USAGE
robaxinand robaxinare indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions. The mode of action of methocarbamol has not been clearly identified, but may be related to its sedative properties. Methocarbamol does not directly relax tense skeletal muscles in man.
CONTRAINDICATIONS
robaxinand robaxinare contraindicated in patients hypersensitive to methocarbamol or to any of the tablet components.
WARNINGS
Since methocarbamol may possess a general CNS depressant effect, patients receiving robaxinor robaxinshould be cautioned about combined effects with alcohol and other CNS depressants.
Safe use of robaxinand robaxinhas not been established with regard to possible adverse effects upon fetal development. There have been reports of fetal and congenital abnormalities following in utero exposure to methocarbamol. Therefore, robaxinand robaxinshould not be used in women who are or may become pregnant and particularly during early pregnancy unless in the judgment of the physician the potential benefits outweigh the possible hazards (see
PRECAUTIONS, Pregnancy).
Use In Activities Requiring Mental Alertness
Methocarbamol may impair mental and/or physical abilities required for performance of hazardous tasks, such as operating machinery or driving a motor vehicle. Patients should be cautioned about operating machinery, including automobiles, until they are reasonably certain that methocarbamol therapy does not adversely affect their ability to engage in such activities.
PRECAUTIONS
Information for Patients
Patients should be cautioned that methocarbamol may cause drowsiness or dizziness, which may impair their ability to operate motor vehicles or machinery.
Because methocarbamol may possess a general CNS-depressant effect, patients should be cautioned about combined effects with alcohol and other CNS depressants.
DRUG INTERACTIONS
See
WARNINGSand PRECAUTIONSfor interaction with CNS drugs and alcohol.
Methocarbamol may inhibit the effect of pyridostigmine bromide. Therefore, methocarbamol should be used with caution in patients with myasthenia gravis receiving anticholinesterase agents.
DRUG & OR LABORATORY TEST INTERACTIONS
Methocarbamol may cause a color interference in certain screening tests for 5-hydroxyindoleacetic acid (5-HIAA) using nitrosonaphthol reagent and in screening tests for urinary vanillylmandelic acid (VMA) using the Gitlow method.
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
Long-term studies to evaluate the carcinogenic potential of methocarbamol have not been performed. No studies have been conducted to assess the effect of methocarbamol on mutagenesis or its potential to impair fertility.
PREGNANCY
Teratogenic Effects
Pregnancy Category C
Animal reproduction studies have not been conducted with methocarbamol. It is also not known whether methocarbamol can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. robaxinand robaxinshould be given to a pregnant woman only if clearly needed.
Safe use of robaxinand robaxinhas not been established with regard to possible adverse effects upon fetal development. There have been reports of fetal and congenital abnormalities following in utero exposure to methocarbamol. Therefore, robaxinand robaxinshould not be used in women who are or may become pregnant and particularly during early pregnancy unless in the judgment of the physician the potential benefits outweigh the possible hazards (see
WARNINGS).
NURSING MOTHERS
Methocarbamol and/or its metabolites are excreted in the milk of dogs; however, it is not known whether methocarbamol or its metabolites are excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when robaxin or robaxin is administered to a nursing woman.
PEDIATRIC USE
Safety and effectiveness of robaxinand robaxinin pediatric patients below the age of 16 have not been established.
ADVERSE REACTIONS
Adverse reactions reported coincident with the administration of methocarbamol include:
Body as a whole: Anaphylactic reaction, angioneurotic edema, fever, headache
Cardiovascular system: Bradycardia, flushing, hypotension, syncope, thrombophlebitis
Digestive system: Dyspepsia, jaundice (including cholestatic jaundice), nausea and vomiting
Hemic and lymphatic system: Leukopenia
Immune system: Hypersensitivity reactions
Nervous system: Amnesia, confusion, diplopia, dizziness or lightheadedness, drowsiness, insomnia, mild muscular incoordination, nystagmus, sedation, seizures (including grand mal), vertigo
Skin and special senses: Blurred vision, conjunctivitis, nasal congestion, metallic taste, pruritus, rash, urticaria
OVERDOSAGE
Management of overdose includes symptomatic and supportive treatment. Supportive measures include maintenance of an adequate airway, monitoring urinary output and vital signs, and administration of intravenous fluids if necessary. The usefulness of hemodialysis in managing overdose is unknown.
DOSAGE & ADMINISTRATION
robaxin(methocarbamol), 500 mgAdults:
Initial dosage: 3 tablets q.i.d.
Maintenance dosage: 2 tablets q.i.d.
robaxin(methocarbamol): 750 mgAdults:
Initial dosage: 2 tablets q.i.d.
Maintenance dosage: 1 tablet q.4h. or 2 tablets t.i.d.
Six grams a day are recommended for the first 48 to 72 hours of treatment. (For severe conditions 8 grams a day may be administered). Thereafter, the dosage can usually be reduced to approximately 4 grams a day.
HOW SUPPLIED
robaxin(methocarbamol tablets, USP)
500 mg tablets are light orange, round, film-coated tablets engraved with ROBAXIN 500 on the unscored side and SP above the score on the other side. They are supplied as follows:
Bottles of 100NDC 52244-429-10robaxin(methocarbamol tablets, USP)
750 mg tablets are orange, capsule-shaped, film-coated tablets engraved with ROBAXIN 750 on one side and SP on the other. They are supplied as follows:
Bottles of 100NDC 52244-449-10
| ROBAXIN
methocarbamol tablet |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - REMEDYREPACK INC. (829572556) |